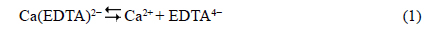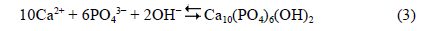Graphene (Gr) is a one atom thick two-dimensional lattice where sp2-hybridized carbon atoms are arranged in a flat honeycomb manner. It has drawn attention in the composite materials field as a useful reinforcement for composites due to its unique qualities: excellent thermal and mechanical properties, large specific surface area and electrical conductivity [1-3]. Graphene has been utilized as a new material in applications including field-effect transistors, memory devices, photovoltaic devices, electron acceptors, various sensors [3], and composites [4-7].
Even though graphene was originally developed for nanoelectronics applications [8], recent research has reported that graphene also has a significant potential in biomedical applications [9]. It has shown great prospects as a filler in bone tissue engineering, while interaction with biomaterials has rarely been reported [2]. In order to minimize direct metal-body fluid contact and to limit the undesired release of metallic ions into the body, biocompatible coatings on the metallic substrate, such as hydroxyapatite (Ca10(PO4)2(OH)2, HAP), have been suggested by many researchers [10-13]. As a major component of natural bone tissues, hydroxyapatite is chemically similar to the natural mineral in bone and has excellent biocompatibility; therefore it is very suitable as an orthopedic biomaterial in various forms and shapes for bone growth and integration [1].
To overcome the poor mechanical properties of hydroxyapatite, our previous research focused on applying graphene in HAP/Gr composite coatings on titanium substrates [14,15]. Recent studies employing graphene as an ideal reinforcement material for hydroxyapatite have demonstrated the significant toughness and strength of the composites, which retain their biocompatibility without any offsetting bioactivity [1,14-16]. It is worth mentioning that, for biomedical purposes, graphene has been used along with chitosan in composites that reportedly show a 200 times increase in elasticity modulus compared to undoped chitosan [17]. Also, it has been reported that the superior electrical conductivity of graphene stimulates the adhesion and proliferation of osteoblasts during new tissue growth [2]. Crack deflection is more effective when using sheet-like reinforcement, as compared with tubular-like reinforcement, suggesting that graphene nano-sheets (GNSs) exhibit a more pronounced toughening effect on brittle materials than do carbon nanotubes (CNTs) [18]. Unlike CNTs, Gr has been synthesized in relatively pure ways and is expected to exhibit low cytotoxicity, since few metallic catalyst particles are associated with its production [19,20]. Therefore, the investigation of graphene-based nanomaterials, and their biomedical application in particular, is an exciting new field of research [21].
The presence of implant materials inside the human body can interfere with the host immune response, and microorganisms have opportunity to adhere to the implant surface, forming socalled biofilms [22]. The antimicrobial properties of graphenebased materials have been thoroughly evaluated by many researchers [22-25]. The antibacterial activity of graphene oxide and graphene is attributed to the significant membrane stress induced by the sharp edges of their nanosheets, which may result in physical damage to cell membranes, leading to the loss of bacterial membrane integrity and the leakage of RNA [25,26]. GNSs serve as “cutters” to disrupt and damage bacteria cell membranes, consisting of phospholipid molecules, leading to the release of bacterial intracellular contents into the environment and eventually bacterial death [25].
The aim of this work was to electrochemically synthesize HAP/Gr composite powder under constant current density for medical aplications as a hard tissue implant (scaffold), and to investigate its antibacterial effect against the growth of bacterial strains, including
2.1. Materials and sample preparation
A homogeneous aqueous dispersion with a final volume of 200 mL, containing Na2H2EDTA·2H2O, NaH2PO4 and CaCl2 (Ca/EDTA/PO43− = 0.25/0.25/0.15 M) and 0.01 wt% graphene was used. The Na2H2EDTA·2H2O, NaH2PO4 and CaCl2 chemicals were all reagent grade (Sigma Aldrich Chemie, Munich, Germany) and used without further purification. The graphene nanopowder (AO-3) was purchased from Graphene Supermarket, USA. The average thickness of the graphene nanoflakes was 12 nm. Approximately 30-50 layers of graphene monolayers were overlapped together. The pH value of the as-received dispersion was 2.0, subsequently titrated with 10 M NaOH to pH 9.0 with an appropriate amount of NaOH.
Subsequently, HAP/Gr powder was synthesized galvanostatically in a two-electrode cell (two rectangular Pt electrodes, 10 mm × 10 mm) at an applied current density of 137 mA cm−2, with constant stirring at room temperature for 3 h, using Reference 600 Potentiostat/Galvanostat/ZRA (Gamry Instruments Inc., Warminster, PA, USA). After that the dispersion was kept at 40℃ until a dense precipitate was obtained. The precipitate was rinsed with deionized water and spun down several times in a centrifuge (10,000 rpm) until a negative reaction for Cl− ions with AgNO3 was observed. The final HAP/Gr powder was obtained by drying for 48 h at room temperature.
The morphology of the HAP/Gr powder was analyzed by field emission scanning electron microscopy (FE-SEM), conducted on a LEO SUPRA 55 (Carl Zeiss, Oberkochen, Germany) operated at an acceleration voltage of 200 kV. Transmission electron microscopy (TEM) measurements of the HAP/Gr powder were carried out using a JEOL 100CX microscope instrument at an operating voltage of 100 kV. Samples were prepared by placing a drop of diluted aqueous HAP/Gr suspension onto a carbon-coated copper grid, and dried samples were examined by TEM. HR-Raman analysis was used to verify graphene incorporation into the composite HAP/Gr powder. This analysis was carried out using a inVia Raman spectrophotometer (Renishaw) equipped with a 514 nm argon laser with 10% intensity of total power (50 mW) and the spectral range of 100 to 3500 cm−1. X-ray diffraction analysis (XRD) was used to assess the phase composition of the HAP/Gr powder. XRD intensity was measured by Philips PW 1051 powder diffractometer, with Ni filtered CuKα radiation (λ = 1.5418 Å), using a scan-step technique (2
The antibacterial activity of the HAP/Gr composite powder was tested against the Gram-positive pathogenic bacteria strain
Overnight cultures (
3.1. Morphological characterization
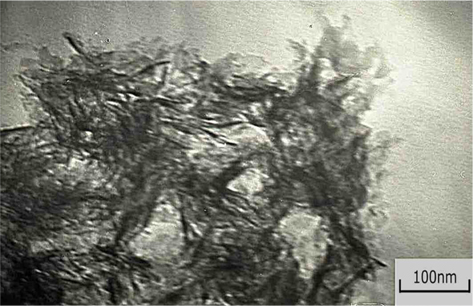
The surface morphology, size and quality of the prepared composite HAP/Gr powder was investigated by TEM and FE-SEM analysis. It can be clearly seen from the TEM results (Fig. 1) that the Gr surface is highly modified, which confirms the EDTA functionalization on its surface. The FE-SEM image (Fig. 2, under high magnification of ×100,000) displays the formation of thick nano-sheets with wrinkled morphology, which is in good agreement with the TEM image.
The results are a solid indication that Gr acts effectively as a nano-reinforcement filler, by providing frictional pull out, crack deflection, and crack bridging as the major toughening mechanisms to resist crack propagation [1]. The high specific surface area of the graphene nanosheets creates an increased contact area with the hydroxyapatite matrix, significantly enhancing the bonding strength between the graphene and hydroxyapatite grains. Moreover, the rough and wrinkled surface texture of the graphene nanosheets plays an important role in enhancing mechanical interlocking, leading to increased load-transfer efficiency between the hydroxyapatite matrix and graphene nanosheets. Finally, it was shown that the two ends of the graphene nanosheets are well bonded to the adjacent hydroxyapatite grains with the plane of the graphene nanosheets nearly parallel to the fracture surface. Thus, it is hypothesized that the grain bridging by graphene nanosheets has a fundamental role in inhibiting crack propagation along the grain boundary (grain-boundary toughening mechanism).
The FE-SEM microphotographs also revealed the surface homogenicity of the electrochemically prepared HAP/Gr powder, which consisted of big agglomerates composed of fine plate-like crystallites of ~40 nm. A similar morphology was observed in the case of electrochemically pure hydroxyapatite powder obtained at the same current density and pH value [29]. The agglomerates of hydroxyapatite nanoparticles, evident in the TEM images shown in Fig. 1, are embedded in a transparent overlay of graphene sheets.
Additionaly, the FE-SEM image (Fig. 2) of prepared composite powder showed that the graphene was surrounded by clusters of plate-like hydroxyapatite nanoparticles (marked with an arow in the high magnification image). In our previous work we have established that hydroxyapatite nanoparticles have a strong tendency to agglomerate due to their high surface area and energy [29].
3.2. XRD, Raman and FT-IR analysis
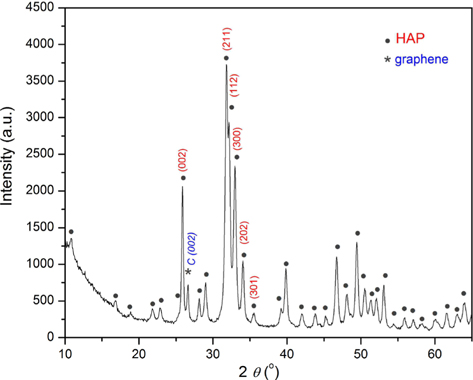
The XRD pattern of the HAP/Gr composite powder (Fig. 3) demonstrates that the predominately detected crystalline phase is assigned to hydroxyapatite. High intensity hydroxyapatite peaks at crystal planes (002), (211), (112), and (300) at 2
According to Ju et al. [30], thermally-reduced graphenes showed a broad (002) peak at 2
Using the X-ray line profile fitting program with a fundamental parameters convolution approach for generating line profiles [32], we calculated the coherent domain sizes and microstrain of HAP/Gr using the (002), (211), (112), (300), (202), and (301) crystal planes. The crystallite domain size, calculated to be 28.9 nm, indicates that the agglomerates observed by FE-SEM analysis were formed through the drying process. This is in good agreement with the proposed nucleation-aggregation-agglomeration growth mechanism [33]. According to this mechanism, particle formation occurs in the following steps: (1) nucleation and growth to form crystallites in nanometric size range, (2) aggregation of elemental nanocrystals by physical attractions, and (3) further crystal growth to form stable agglomerates. However, the presence of Gr increased the crystalline domain size of the hydroxyapatite grains, since the reported value for pure hydroxyapatite electrochemically synthesized at the same pH and current density was 21.6 nm [29], indicating that there were interactions between the hydroxyapatite and graphene sheets.
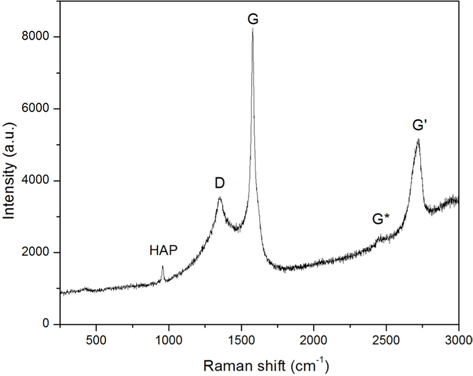
Raman measurement was employed to identify the existence of Gr in the composite powder. The Raman spectrum of the HAP/Gr powder (Fig. 4) depicts both the typical D peak at 1350 cm−1 and the G peak at 1578 cm−1, as well as the very weak G* peak at 2434 cm−1 and G' peak at 2715 cm−1, denoted as a major fingerprint of graphene structure [34]. The shape, position and intensity of the G' band depends markedly on the number of layers. The G-band corresponds to the ordered sp2-bonded carbon atoms and the D-band represents defects originating from the disordered aromatic structure on the Gr edges [35]. The characteristic sharp Raman peak at 961 cm−1 is characteristic of the nanosized crystalline hydroxyapatite phase, which is attributed to the symmetric stretching mode ν1(PO43−) [36]. The Raman study therefore indicates the interaction between Gr and hydroxyapatite, confirming the integration of graphene in the novel electrochemically prepared HAP/Gr composite powder.
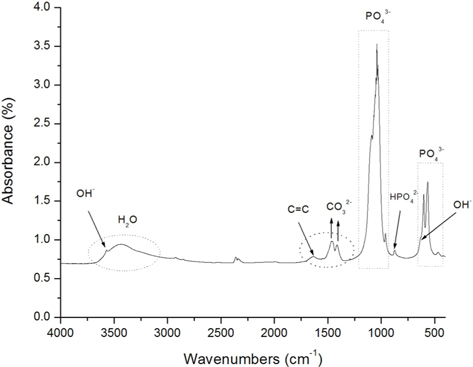
The Fourier transform infrared spectroscopy (FT-IR) spectrum of the HAP/Gr powder is depicted in Fig. 5 indicating the characteristic absorption bands of natural hydroxyapatite at 463, 569 and 603 cm−1, which are attributed to the P−O bending of phosphate groups [37]. The three absorption bands of the PO43− group were clearly distinguished in the composite HAP/Gr nanopowder at 1092, 1038 and 960 cm−1, which corresponds to the O−P−O phosphate ions of the hydroxyl site [38,39]. The characteristic band at 3572 cm−1 and weak broad band at 634 cm−1 can be attributed to the stretching and vibrational modes of the structural OH− groups of the hydroxyapatite lattice [40]. The absorbance bands at 1420 and 1460 cm−1 correspond to ν3 asymmetrical stretching vibrations of the CO32− ions, indicating predominatly B-type hydroxyapatite [41]. It is preferential substitution in the human bone known for its excellent bioactivity and osteoinductivity [41]. A vibration band located at 1640 cm−1 corresponds to the skeletal vibration of Gr due to aromatic sp2 hybridized C=C vibration stretching [38,39,42], confirming its presence in the synthesized composite.
The mechanism of hydroxyapatite formation requires the liberation of Ca2+ ions from EDTA [29,43]:
the generation of OH− ions through water reduction:
and ionic migration towards the electrodes, where precipitation of hydroxyapatite occurs:
As previously emphasized, the role of EDTA is to provide a functionalized surface on the Gr sheets, where the nucleation of hydroxyapatite grains is facilitated. However, hydroxyapatite nanorods grow with a preferred orientation along the graphene sheets. According to Liu et al. [21], the hydroxyapatite and graphene sheets are connected by van der Waals bonding. Nucleation of the hydroxyapatite crystals probably occurs on either the graphene wall or the cross-section of graphene multi-sheets, followed by subsequent crystal growth along or perpendicular to the surface of the graphene sheet. They proved that the (300) plane of the hydroxyapatite crystals is parallel to the surface of the graphene walls.
Keeping in mind the atomic structure of hydroxyapatite, the (300) plane contains Ca atoms at each corner of the rectangle and the distance between each pair of Ca atoms is 0.9418 nm and 0.6884 nm, whereas the distance between two neighboring Ca atoms in the (100) plane is 0.9418 nm and 0.3442 nm, respectively, while the distance between adjacent graphene sheets is 0.347 nm. Single layer graphene is constituted by carbon atoms arranged periodically in a hexagonal manner, and the nearest distance between two carbon atoms is 0.142 nm. Multilayer graphene sheets contain several graphene monolayers with the inter-wall distance of 0.340 nm. On the other hand, the lattice spacing of the (002) plane of hydroxyapatite is 0.344 nm. Since the (300) plane takes priority over the (100) plane to match with the surface of the graphene sheets, and the open ends of the graphene multisheets form relatively stronger interfaces with the (002) plane of the hydroxyapatite crystals than other planes, it can be considered that the (300) plane of the hydroxyapatite forms a strong interfacial bond with the surface of the graphene wall, and the cross-section of graphene builds a strong interface with the (002) plane of the HAP crystals due to the smaller lattice mismatch.
Therefore, all of the above analyses―XRD, FT-IR and Raman spectroscopy―confirm the incorporation of graphene in the prepared HAP/Gr powder.
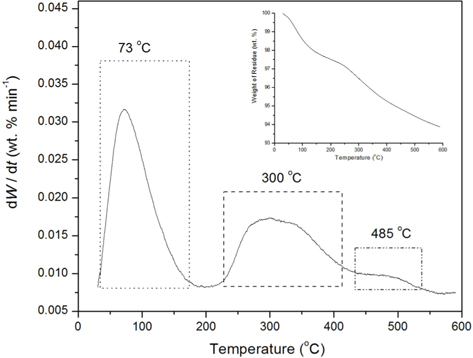
TGA was conducted in order to study the decomposition behavior and phase transformation of the HAP/Gr composite. Fig. 6 depicts the differential thermogravimetry (DTG) and TG curves of nanosized HAP/Gr powder in the temperature range 25℃-600℃, exhibiting three weight loss stages. The first stage from 25℃ to 170℃, with 2.32 wt% mass loss and a sharp peak in the DTG curve at 73℃, corresponds to the desorption of adsorbed water molecules from the crystallite surface. The second stage (170℃-420℃), with 2.60 wt% mass loss and a broad peak at around 300℃ in the DTG curve, corresponds to the combustion of the organic material, EDTA [44] and the unstable carbon in the graphene structure [45], as well as the beginning of hydroxyapatite dehydroxylation [14,15]. The third stage, observed between 420℃ and 600℃, with 1.22 wt. % mass loss and a peak at 485℃ in the DTG curve, is attributed to the complete decomposition of graphene [46] and the dehydroxylation or early slow decomposition of hydroxyapatite [29]. The total mass loss at 600℃ was 6.14 wt%, confirming the greater thermal stability of the nanosized HAP/Gr powder, compared to 9.50 wt% mass loss for pure hydroxyapatite synthesized under the same conditions.
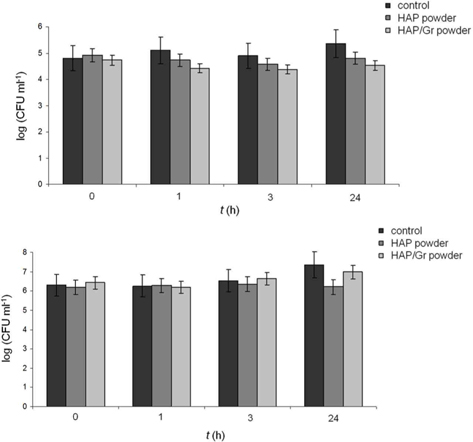
The antibacterial activity of the HAP/Gr powder was investigated quantitatively against two bacterial strains, Gram-positive
A slight decrease in the total number of
The antibacterial mechanism of the HAP/Gr powder can be understood as a result of the graphene-based materials causing direct damage to cell membranes and/or by oxidative stress. Namely, bacterial viability decreases as a result of graphene causing direct physical damage to bacterial membranes and finally resulting in the release of intracellular contents. This breach in the cell wall is caused by the blade-like action of low thickness graphene with sharp edges [25,47]. In addition, graphenes may chemically increase cellular oxidative stress, which could disrupt a specific microbial process. Oxidative stress mediated by the graphene-based materials could have several different routes. The first one is through formation of reactive oxygen species (ROS), whereby oxidative stress is induced by ROS generated by graphene and related species, e.g. graphene oxide or reduced graphene oxide. The other possibility is ROS-independent oxidative stress. Graphene-based materials may target a specific microbial process or a vital cellular structure or component by disturbing them or oxidizing, without ROS production [25].
Both of these effects are enhanced in the reduced form of the materials (pure graphene), due to the stronger interaction between sharp sheet edges and the bacteria’s cell membrane and/or better charge transfer between the bacteria and the graphene-based material itself.
Based on our antimicrobial results, the investigated HAP/Gr composite powder could be considered a selective antibacterial material. Selectivity and specificity, targeting specific bacterial strains, are mandatory requirements for any type of antibiotic medication. Even though its present efficacy needs improving, the HAP/Gr composite powder is a candidate for a selective biomaterial for bone tissue engineering. However, since the antibacterial activity of graphene remains a controversial issue [47], further insights into the mechanism of graphene antibacterial action are needed and should be further pursued. To ensure the strong antibacterial activity of the HAP/Gr composite, the addition of small quantities of silver nanoparticles is necessary, as we proved in our previous work [15].
In summary, we demonstrated the succesful electrochemical synthesis of nanosized composite HAP/Gr powder at constant current density. Agglomerates of HAP/Gr nanoparticles in the graphene overlayer were observed by FE-SEM and TEM analysis. Raman spectra provided evidence of the characteristic D, G, G* and fingerprint G' (2715 cm−1) bands, indicating that the graphene retained its original structure in the HAP/Gr powder. XRD analysis proved Gr incorporation, and the increase in the crystalline domain size of HAP/Gr in comparison to pure hydroxyapatite powder, due to established interactions between the hydroxyapatite and graphene sheets, which was also confirmed by the composite's greater thermal stability. The FT-IR spectrum revealed the vibration band located at 1640 cm−1 which corresponds to aromatic sp2 hybridized C=C vibration stretching, as expected for HAP/Gr. The absorbance bands indicated predominatly carbonate B-type hydroxyapatite, identical to the human bone and therefore an ideal substitution material with excellent bioactivity and osteoinductivity. The electrochemically synthesized HAP/Gr powder exhibited a slight antibacterial effect against