Recently, several studies investigating insect biological resources have been performed in Korea. Some of these cases involved insects that are used as traditional medicines with proven efficacy (Chung et al. 2013a; Chung et al. 2013b; Kwon et al. 2013). The distribution and sales of certain edible insects have accelerated the industrialization of insects; however, the mass breeding of insects has also caused an increase in diseases (Kwak et al. 2014; Lee et al. 2015). Accordingly, increased monitoring has been initiated for diseases occurring in farms of Protaetia brevitarsis seulensis (Coleoptera: Cetoniidae), an edible insect that is mass-reared in Korea, as well as the Japanese rhinoceros beetle Allomyrina dichotoma (Coleoptera: Scarabaeidae), and diseases with a high incidence were identified. Insects are mainly infected by fungi, bacteria, viruses, and protozoa, and mostly show cross-infection of fungi-bacteria, fungi-viruses, or fungi-bacteria-viruses. Statistical analysis of entomopathogenic microorganisms discovered at P. b. seulensis farms from February 2013 to July 2014 showed that the most common bacterium was Serratia marcescens (Enterobacteriales: Enterobacteriaceae), with an infection rate of 60% revealed by polymerase chain reaction. Furthermore, there was no mortality observed in 3rd stage larvae after 4 wk of peroral infection due to intake of S. marcescens. However, when a peroral infection experiment was performed at an elevated bacterium concentration of 1 × 108/mL in 2nd stage P. b. seulensis larvae, S. marcescens was found to be fatal.
S. marcescens is an entomopathogenic bacterium that is commonly used as an insecticide (Carol R. Lauzon 2003; Ishii et al. 2014). Several investigations have shown that S. marcescens is lethal to many kinds of insects, including Bombyx mori (Lepidoptera: Bombycidae), P. b. seulensis (Coleoptera: Cetoniidae), and Rhagoletis pomonella (Diptera: Tephritidae) (Ishii et al. 2014; Kwak et al. 2014; Lauzon C. R. 2003). Moreover, S. marcescens is also considered to be an opportunistic pathogen and can cause antibiotic-resistant hospital-acquired infections (Murdoch et al. 2011). S. marcescens can outcompete other bacteria and survive to express antibacterial-resistant activity (Murdoch et al. 2011). Paenibacillus popilliae (Eubacteriales: Bacillaceae) causes milky disease and is known as an obligate pathogen in Japanese beetle and other scarab larvae (F. E. El-Borai 2005; Harrison et al. 2000). In addition to P. popilliae, a novel Paenibacillus sp. was found to be an obligate entomological pathogen (F. E. El-Borai 2005). Bacillus thuringiensis (Bacillales: Bacillaceae) is a soil bacterium that has been used as a safe and environmentally friendly biopesticide (Gao et al. 2012), and its main toxins include Cry4Aa, Cry4Ba, Cry11Aa, and Cyt1Aa (Noguera and Ibarra 2010; Stalinski et al. 2014). In particular, the Cry and Cyt toxins are proteins that are used worldwide for insect control, and have been shown to interact with receptors on the host cell surface (Bravo et al. 2007; Stalinski et al. 2014).
P. b. seulensis (Kolbe) was reared in the plastic cages (60 × 33 × 38 cm) at 25℃, 40–50% relative humidity with autoclaved sawdust. For the experiments, only 2nd and 3rd stage larvae of P. b. seulensis were collected. The insects were starved for 24 h before entomopathogen treatment.
Bacillus thuringiensis (KACC10169) and Serratia marcescens (KACC11892) were purchased from the Korean Agricultural Culture Collection (KACC, Wanju, Korea). Paenibacillus popilliae (KCTC3806) was purchased from the Korean Collection for Type Cultures (KCTC, Daejeon, Korea). Lyophilized bacteria were incubated on nutrient agar plates, grown for 18–24 h, and colonies were isolated. The cultured bacteria were diluted to a total concentration of 1 × 108 pfu/mL using a hemocytometer.
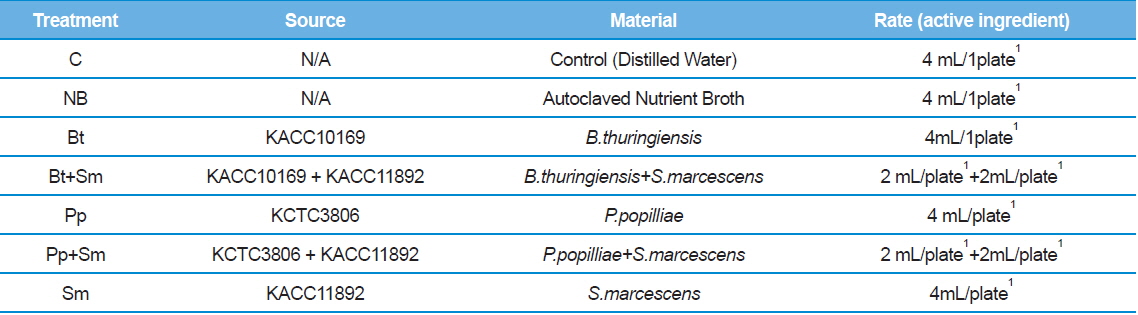
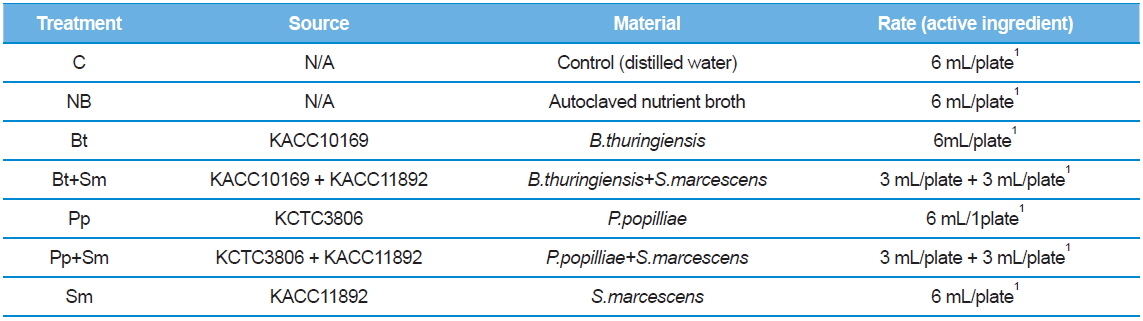
We placed seven 2nd stage P. b. seulensis larvae onto separate plates 4.5 cm in diameter and 1 cm in height. We treated 2 g of fermented sawdust per insect with 4 mL (4 × 108 pfu/mL) of each bacterium: B. thuringiensis (Bt), P. popilliae (Pp) and S. marcescens (Sm). The bacteria mixtures (Bt+Sm, Pp+Sm) were prepared with 2 mL Bt, 2 mL Sm, or 2 mL Pp, depending on the combination, to achieve the same total bacterial concentration (Table 1). Similarly, we placed 3rd stage P.b. seulensis larvae individually onto plates 4.5 cm in diameter and 3 cm in height, and treated 4 g of fermented sawdust per insect with 6 mL (6 × 108 pfu/mL) of each bacterium (Bt, Pp, Sm). The bacteria mixtures (Bt+Sm, Pp+Sm) were prepared with 3 mL Bt, 3 mL Sm, or 3 mL Pp to achieve the same total bacterial concentration (Table 2). We used the same volumes of distilled water as a negative control, and the same volumes of the nutrient broth from the bacterial culture as a positive control. The experiment was repeated three times, and the P. b. seulensis treated with bacteria were examined for 4 wk.
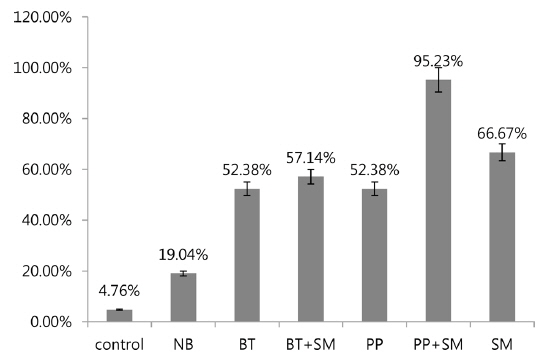
A previous study showed high mortality of 3rd stage P. b. seulensis larvae via hemolymph injection of S. marcescens (Kwak et al. 2014), but showed 0% mortality when 3rd stage P. b. seulensis larvae ingested S. marcescens (1 × 102, 1 × 104, 1 × 107 pfu/mL) together with sawdust. In this study, we investigated the mortality rate resulting from cross-infection with S. marcescens and entomopathogenic bacteria (B. thuringiensis and P. popilliae) that occur commonly in P. b. seulensis with a high fatality rate. We also confirmed the effect of S. marcescens on mortality, which is diagnosed very frequently in P. b. seulensis breeding farms and is known as an opportunistic bacterium. The results of this study confirmed that mortality increased in insects with peroral infections of S. marcescens, similar to other bacteria. We observed mortality after 4 wk when insect sawdust was treated with Sm together with Bt and Pp, which are known to be pathogenic in Coleoptera. In 2nd stage P. b. seulensis larvae, Bt treatment at 4 × 108 pfu/mL per insect resulted in 52.38% mortality. The Bt+Sm combination treatment resulted in 57.14% mortality, which was 4.74% higher than Bt treatment alone. Pp treatment resulted in 52.38% mortality, whereas the combination of Pp+Sm resulted in a mortality rate of 95.23%, which was 42.85% higher than Pp treatment alone (Fig. 1). Sm treatment resulted in a 14.29% higher mortality rate than Bt and Pp.
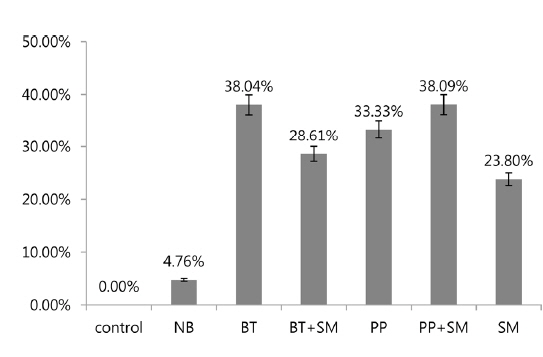
In 3rd stage P. b. seulensis larvae, Bt treatment at 6 × 108 pfu/mL per insect resulted in 38.04% mortality, and the Bt+Sm combination resulted in a mortality rate of 28.61%. Pp treatment resulted in 33.33% mortality, whereas mortality for the Pp+Sm combination was 38.09%, which was 4.76% higher. Sm treatment resulted in a rate of 23.80%, which was lower than those of Bt and Pp. These results confirmed that mortality is dependent on the insect developmental stage. In 2nd stage larvae, mortality due to S. marcescens was higher than that caused by B. thuringiensis and P. popilliae, whereas it was lower in 3rd stage larvae (Fig. 2). In other words, S. marcescens induced stronger mortality in 2nd stage larvae than 3rd stage larvae, and showed mortality with higher-efficiency in 2nd stage larvae compared to B. thuringiensis and P. popilliae. The Pp+Sm combination treatment revealed a very strong synergistic effect in 2nd stage P. b. seulensis larvae compared to the Pp treatment alone, and its mortality rate after 4 wk was 95.23%, which was 42.85% higher than the Pp treatment alone. Since mortality is maximal when P. popilliae and S. marcescens are detected together in a breeding environment or in sawdust, the results of this study can be utilized in the prevention and diagnosis of such cases. S. marcescens, an entomopathogen, has been reported to be an opportunistic bacterium (Murdoch et al. 2011), but the results of this study confirmed that it can also show higher mortality than B. thuringiensis and P. popilliae, beyond a certain concentration, which proves that its fatal effects are stronger at younger larval stages. Furthermore, this study provides valuable information with respect to predicting the effect of these bacteria on mortality when they are detected together at insect breeding sites, which will make prevention possible. In future studies, it will be necessary to investigate the mechanisms of how S. marcescens interacts with P. popilliae to maximize mortality and to examine the specific mortality rate of each bacterium for each stage of insect development.