Production of finfish culture increased gradually these years in Korea. Aquaculture of olive flounder
The present study investigated the effects of dietary microbial phytase supplementation on the nutrient digestibility, growth performance and body composition in the juvenile olive flounder fed soybean meal-based diets.
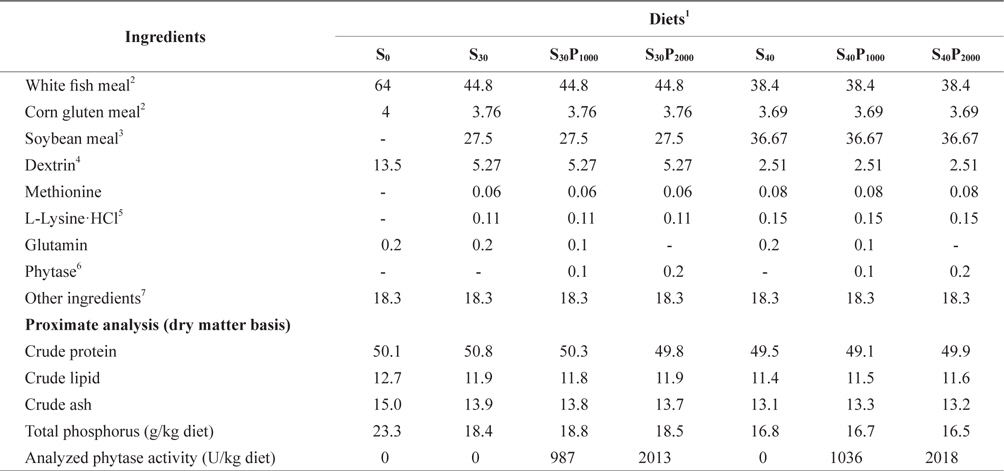
Composition of the experimental diets is shown in Table 1. Seven experimental diets were formulated to be isonitrogenous and isocaloric to contain 50% crude protein (CP) and 16.7 kJ of available energy/g with or without dietary phytase supplementation (Natuphos-5000TM, BASF, NJ, USA). Estimated available energy of the experimental diets was calculated as 16.7, 16.7 and 37.7 kJ/g for protein, carbohydrate and lipid, respectively (NRC, 1993). White fish meal (FM) provided 89.1% of the total protein in the basal diet (S0), in the other 6 diets 30% or 40% FM protein was replaced by soybean meal as: 70% FM+30% soybean meal (S30); 70% FM+30% SM+1000 U phytase/kg diet (S30P1000); 70% FM+30% SM+2000 U phytase/kg diet (S30P2000); 60% FM+40% SM (S40); 60% FM+40% SM+1000 U phytase/kg diet (S40P1000); and 60% FM+40% SM + 2000 U phytase/kg diet (S40P2000). All the ingredients were mixed completely and then pelleted by using a 2 mm diameter module. After processing, all the diets were packed into small bags and kept at -80˚C until use.
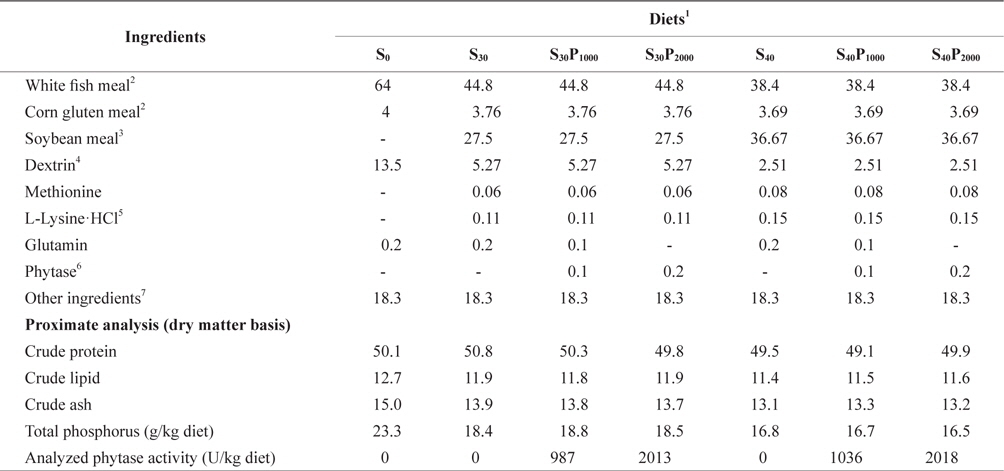
Composition and proximate analysis of experimental diets for juvenile olive flounder Paralichthys olivaceus
Juvenile olive flounder were transferred from Pukyong Aquaculture Company, Tongyoung, Korea to the Department of Fisheries Biology, Pukyong National University, Korea. Before the start of the experiment, all the fish were reared in a 3000-L round tank and were fed a basal diet (S0) for 2 weeks to acclimated with the experimental diet and conditions. The feeding trial was conducted by using a flow-through system with 60-L aquaria receiving filtered sea water at a rate of 1 L/min. Supplemental aeration was provided to maintain dissolved oxygen near saturation. Water temperature was maintained at 15 ± 1˚C. Fish averaging 6.15 ± 0.04 g (mean ± SD) were distributed randomly to each aquarium as a group of 25 fish and were fed one of seven experimental diets in triplicate groups at a rate of 3% of wet body weight for 8 weeks. Total fish weight in each aquarium was measured every 2 weeks, and the amount of diet fed to fish was adjusted accordingly.
>
Sample collection and analysis
At the end of the feeding trial, all fish were weighed and counted to calculate percent weight gain (WG), feed efficiency ratio (FER), specific growth rate (SGR), protein efficiency ratio (PER), hepatosomatic index (HSI), condition factor (CF), and survival. Blood samples were obtained from the caudal vein by using a heparinized syringe. Hematocrit (PCV) was determined from five individual fish per aquarium by the microhematocrit method (Brown, 1980) and hemoglobin (Hb) was measured in the same five fish by the cyanmethemoglobin procedure using Drabkin’s reagent. A hemoglobin standard prepared from human blood (Sigma Chemical, St. Louis, MO) was used. Five fish per aquarium were used for proximate analysis. Crude protein, moisture and ash of whole-body samples were analyzed by AOAC methods (1995). Crude lipid was determined by using an ether extraction procedure (Soxtec System 1046, Foss, Sweden) after freeze-drying samples for 12 h. Fecal collections and digestibility determination were conducted according to the method described by Lim et al. (2004).
All data were subjected to one way ANOVA using SPSS for Windows (release 14.0). When a significant treatment effect was observed, an Honestly Significant Difference (HSD) test was used to compare means. Treatment effects were considered significant at
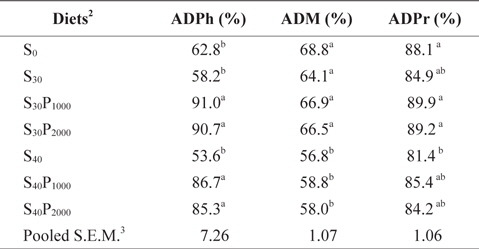
Apparent digestibility coefficients (ADCs) of phosphorus, dry matter and crude protein of diets containing different levels of soybean meal and phytase fed to juvenile olive flounder for 8 weeks are summarized in Table 2. ADCs of phosphorus in diets S30P1000, S30P2000, S40P1000 and S40P2000 were significantly higher than those in diets S0, S30 and S40 (
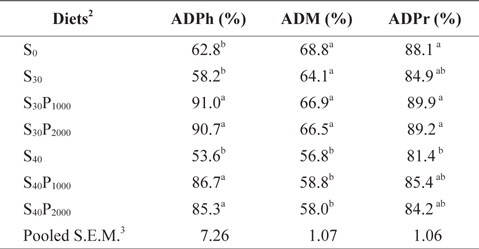
Apparent digestibility coefficients of phosphorus (ADPh), dry matter (ADM) and crude protein (ADPr) of the diets containing different levels of soybean meal and phytase in juvenile olive flounder Paralichthys olivaceus1
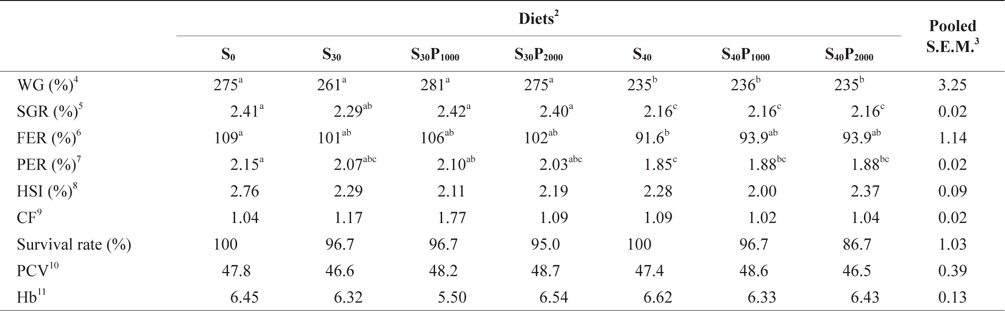
Growth performance, hepatosomatic index (HSI) and survival rate of juvenile olive flounder fed seven experimental diets for 8 weeks were shown in Table 3. There was no significant difference in weight gain (WG), specific growth rate (SGR), feed efficiency ratio (FE) and protein efficiency ratio (PER) of fish fed S0, S30, S30P1000 and S30P2000. However, WG, SGR, FE and PER of 40% soybean meal groups were significantly lower than those of fish fed S0 and 30% soybean meal. The reduced growth of 40% soybean meal groups could be because of the effect of difference in amino acid profiles and amount of antinutritional factors in the diets. Soybean contains numerous antinutritional factors like proteinase inhibitors and Suragglutinating lectins, as reported by many researchers (Refstie et al., 2001; Sun et al., 2007). In this study, supplementation of 1000 or 2000 U phytase did not improve WG. Similar results were observed in Korean rockfish, red sea bream and parrot fish (Yoo et al., 2005; Biswas et al., 2007; Lim and Lee, 2009). However, growth improvement was also observed in salmon, rainbow trout and Nile tilapia fed the diets with phytase pretreated ingredients (Cain and Garling 1995; Vielma et al., 2002; Cao et al., 2008). According to Cao et al., (2008), pretreatment of plant ingredients using microbial phytase significantly increased nutrient digestibility and growth. To improve the nutritive value of plant products for fish, ingredients are being modified by chemical, mechanical and biological methods (Cao et al., 2007). While differences in dietary formulations could be one of the reasons contributing to differences in feeding response of fish to phytase, species differences is another possible factor (Venou et al., 2006). Therefore, further studies should be conducted to compare the economic benefits among using chemical, mechanical and biological methods to the plant protein sources. There was no significant difference in HSI, condition factor (CF), hematocrit (PCV), hemoglobin (Hb) and survival rate among all the dietary treatments. Survival rate of fish ranged from 94.6 to 100 with flounder fed S40P2000 showed the lowest value of survival.
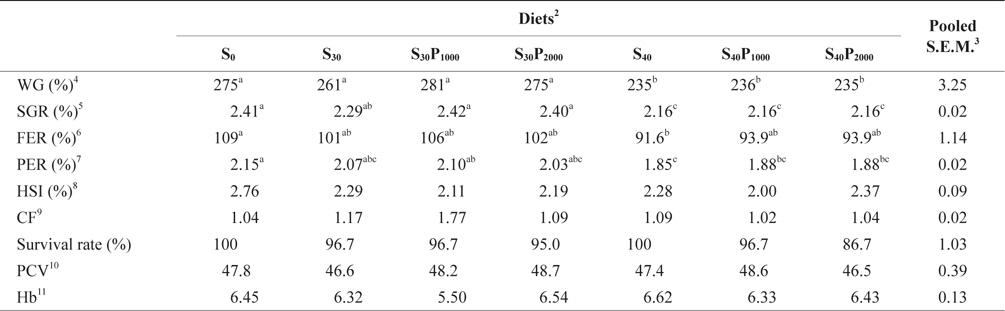
Growth performance, hepatosomatic index (HSI), and survival rate of juvenile olive flounder Paralichthys olivaceus fed seven experimental diets for 8 weeks1
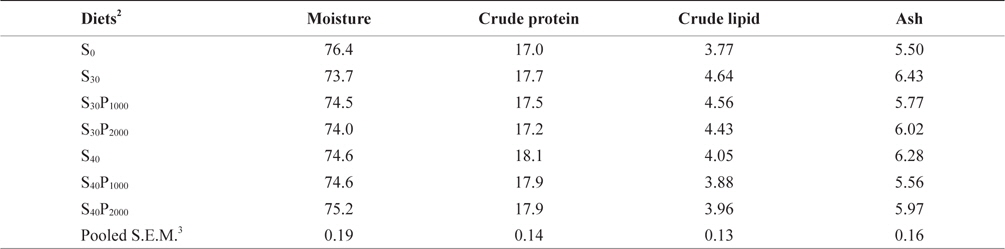
Whole-body proximate compositions were shown in Table 4. In this study, whole body composition resulted to be not affected by soybean meal and phytase level. This result is in agreement with several studies on other species (Kim et al., 2009). Whole body protein slightly increased with increasing soybean meal in diets regardless the supplementation levels of phytase. There was a negative correlation to whole body lipid and ash content a slight decrease was observed when soybean meal inclusion increased. Similar results were also reported by Lin et al. (2012) and Bonaldo et al. (2008). Based on the experimental results, we conclude that supplementation of phytase could significantly increase available P content in feed, indicating that the indigestible phytate P successfully converted to available P by phytase.
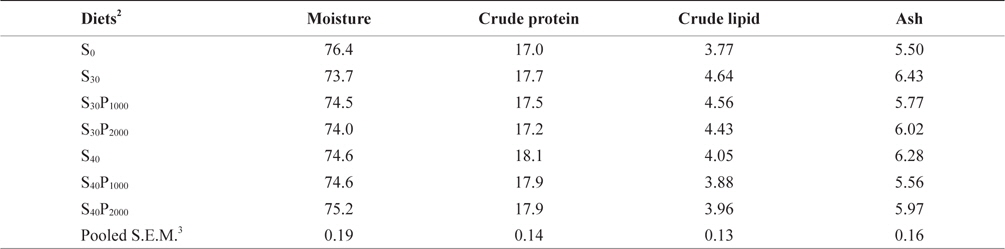
Whole body proximate composition of juvenile olive flounder Paralichthys olivaceus fed experimental diets for 8 weeks (% of as-is basis)1