It was first recorded in
Ju
Therefore, this experiment was conducted not only to study the single-dose toxicity of intramuscular administration of
The substances used in the experimental groups of this study were prepared by making
Six-week-old SD rats were used in this experiment. Twenty male rats (body weights: 180.4-199.7 g) and 20 female rats (body weights: 144.0-162.3 g) were assigned to the
All animals in both the
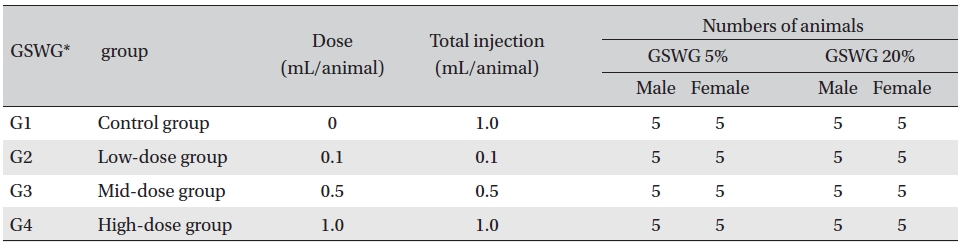
[Table. 1] Composition of the Guseonwangdo-go glucose group
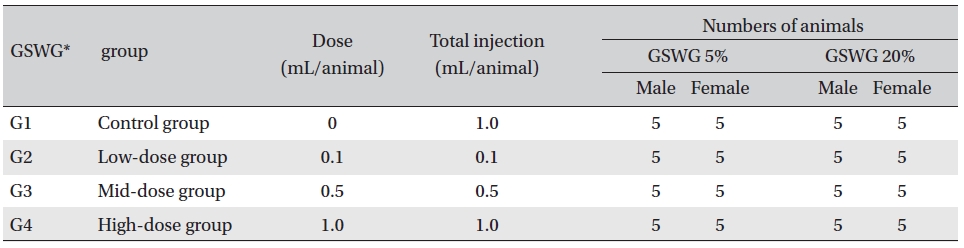
Composition of the Guseonwangdo-go glucose group
In a pilot test (Biotoxtech Study No.: B12878P), 1.0 mL/ animal, referring to 1.0 mL/time of clinically applied dose, was administered by intramuscular (IM) injection (left thigh) to one male and one female rat and resulted in no deaths. From this result, the doses for
All experiments were conducted at Biotoxtech, an authorized institution for non-clinical studies, under the regulation of Good Laboratory Practice of KFDA Notification No. 2012-61 (Test Guidelines for Non-clinical Studies, Aug 24, 2012) [7].
The general symptoms (types of toxic symptoms, expression time, recovery time, etc.) and mortality were observed after 30 minutes, and then after 1, 2, 4, and 6 hours on the day of injection (day 0). From the next day to the 14th day after the injection, the general symptoms were examined once a day.
The body weights were measured on the day of the injection (immediately before the injection) and then on the 3rd, 7th, and 14th days after the injection.
All animals were fasted during the 18 hours before autopsy. They were then anesthetized with isoflurane, after which blood was collected from the abdominal aorta on the day of autopsy (15 days after the injection). For the hematological test, approximately 1 mL of the collected blood was placed in an ethylene-diamine-tetra-acetic acid tube and was analyzed using a hematology analyzer (ADVIA ® 120, Siemens, Germany). For the coagulation test, approximately 2 mL of the collected blood was placed in a tube with 3.2% sodium citrate and centrifuged at 3,000 rpm for 10 minutes. Blood plasma was then collected. Different laboratory tests were conducted using a coagulation time analyzer (Coapresta® 2000, Sekisui, Japan).
For the biochemical test, the blood remaining after carrying out the hematological tests was centrifuged at 3,000 rpm for 10 minutes, and the serum was collected. Tests were done using a biochemistry analyzer (7180, Hitachi, Japan) and an electrolyte analyzer (AVL9181, Roche, Germany).
During the necropsy performed on all the animals, organs and tissues from the entire body were checked thoroughly by visual inspection.
Tissues at the injection sites for all the animals were sampled and fixed in 10% neutral buffered formalin. Routine histological methods, such as trimming, dehydration, and paraffin embedding, were conducted on the fixed organs and tissues. These were then sliced using a microtome and stained with hematoxylin & eosin (H&E).
All the results obtained were analyzed by using STATA/SE 9.2 for Windows (StataCorp LP, College Station, TX, USA). The equal variance was tested using Bartlett’s test. If the sample variances were equal, significant results were obtained using the one-way analysis of variance. Dunnett’s multiple range
During the observation, no mortalities or clinical signs were observed in any of the experimental or control groups.
Weight gains were observed in both the experimental and the control groups, but comparison of the experimental and the control groups showed no significant changes in body weight.
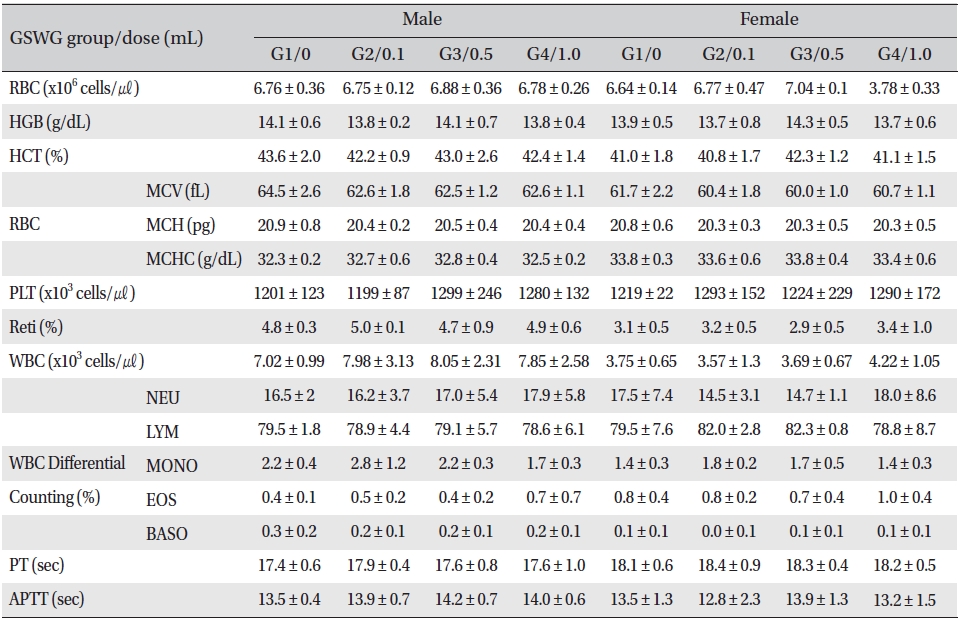
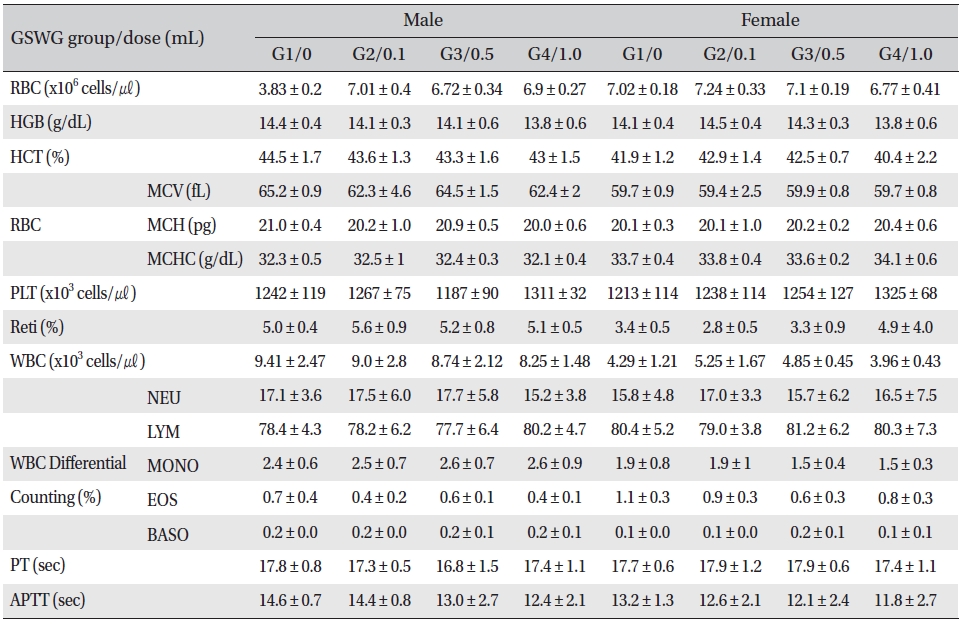
No significant changes were noted in the hematological test results, comparison of the experimental and the control groups (Tables 2,3).
[Table. 2] Mean hematology parameters of Sprague-Dawley rats in the Guseonwangdo-go glucose 5% group
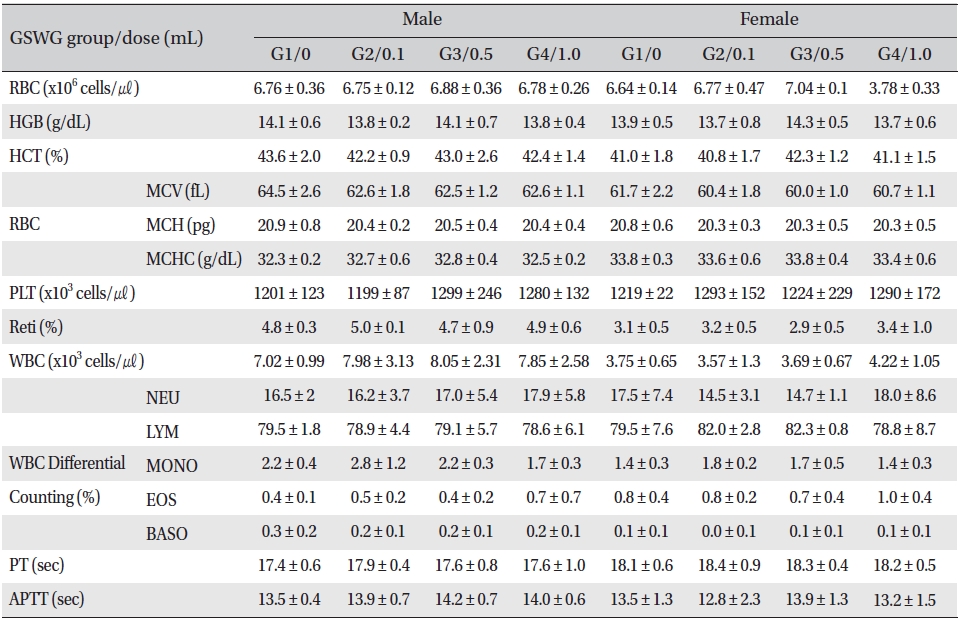
Mean hematology parameters of Sprague-Dawley rats in the Guseonwangdo-go glucose 5% group
[Table. 3] Mean hematology parameters of SD rats in the Guseonwangdo-go glucose 20% group
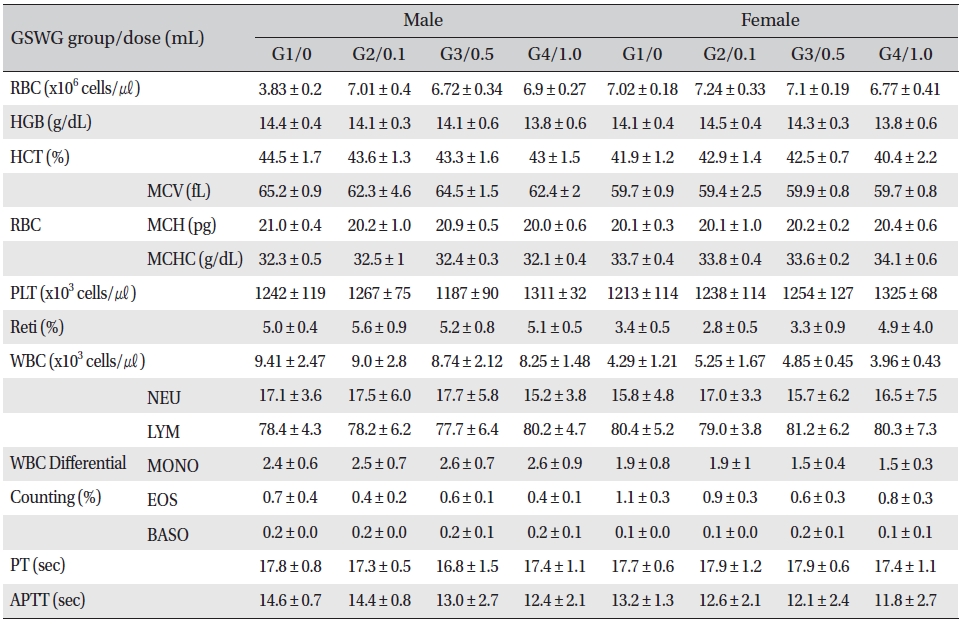
Mean hematology parameters of SD rats in the Guseonwangdo-go glucose 20% group
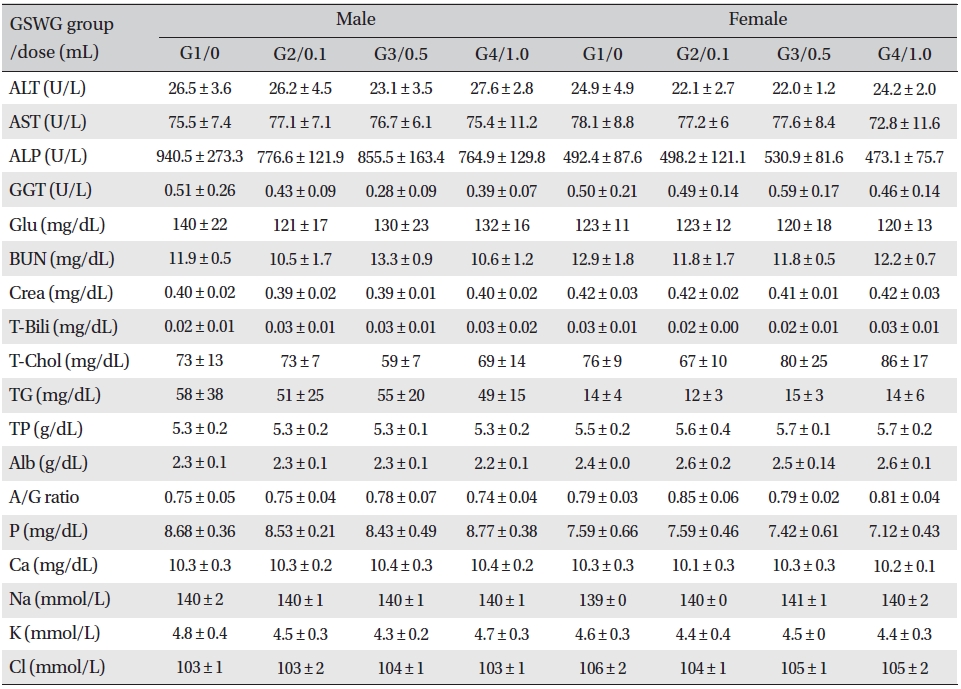
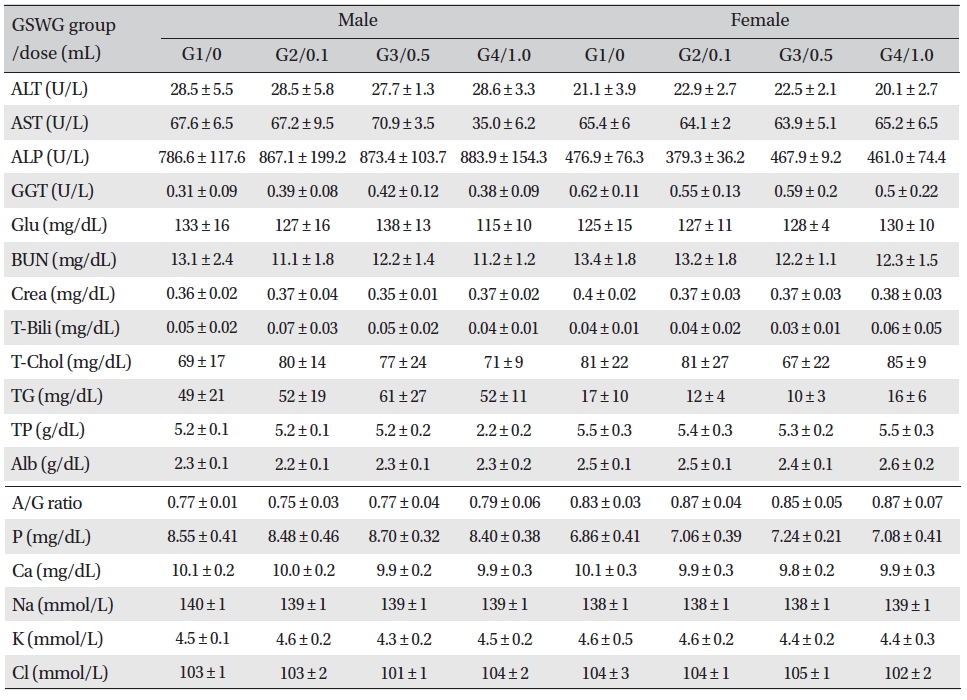
No significant biochemical changes in the experimental groups were observed when the blood was analyzed (Tables 4,5).
[Table. 4] Mean clinical chemistry of SD rats in the Guseonwangdo-go Glucose 5% group
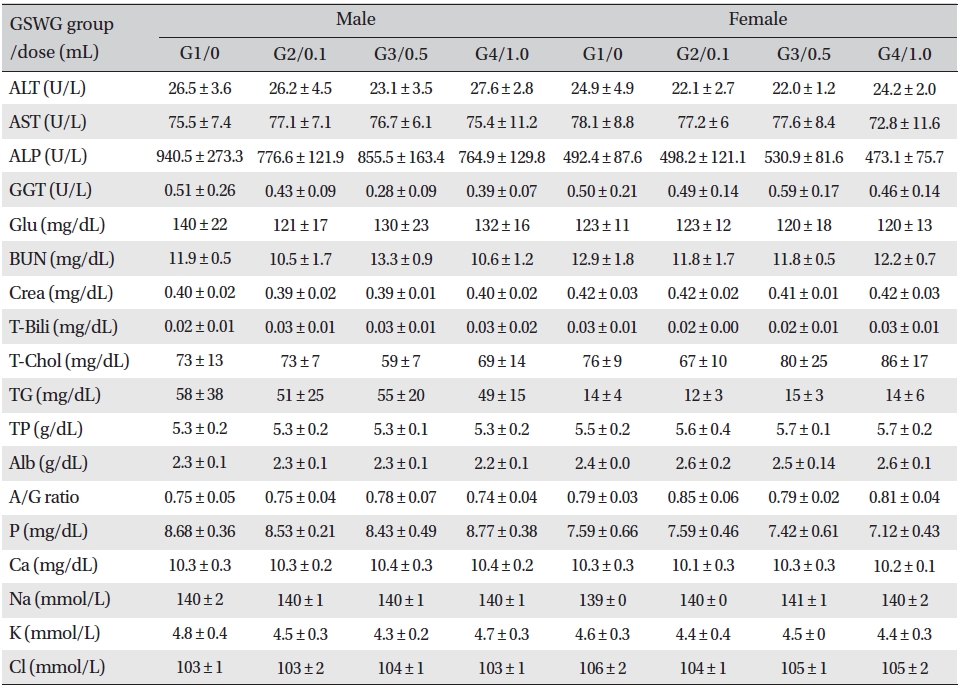
Mean clinical chemistry of SD rats in the Guseonwangdo-go Glucose 5% group
[Table. 5] Mean clinical chemistry of SD rats in the Guseonwangdo-go glucose 20% group
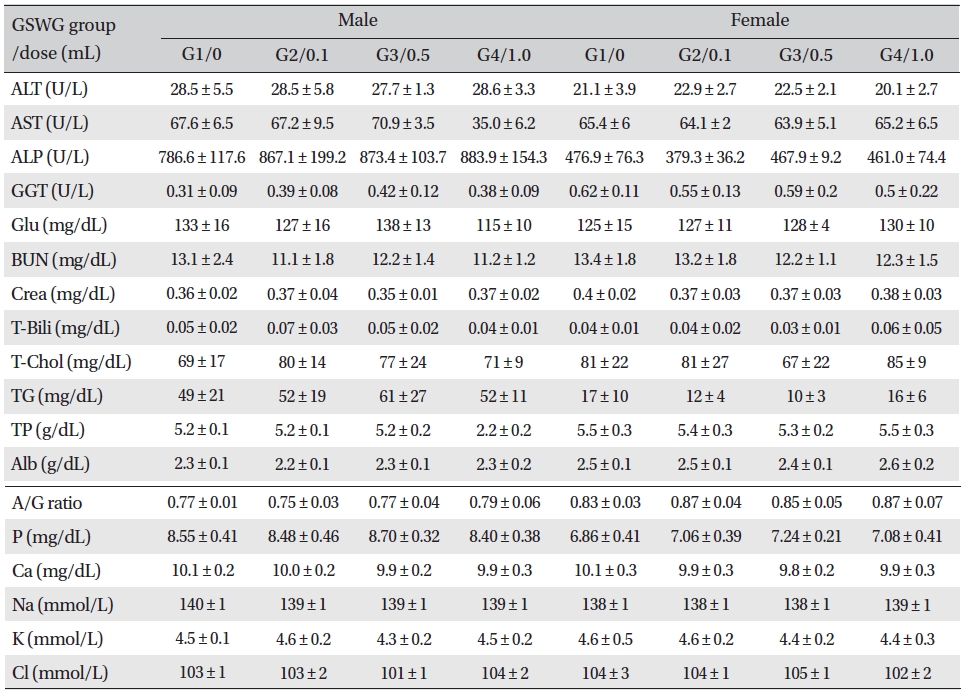
Mean clinical chemistry of SD rats in the Guseonwangdo-go glucose 20% group
On necropsy, no abnormalities were observed when the visual inspection was conducted on all of the animals in all groups.
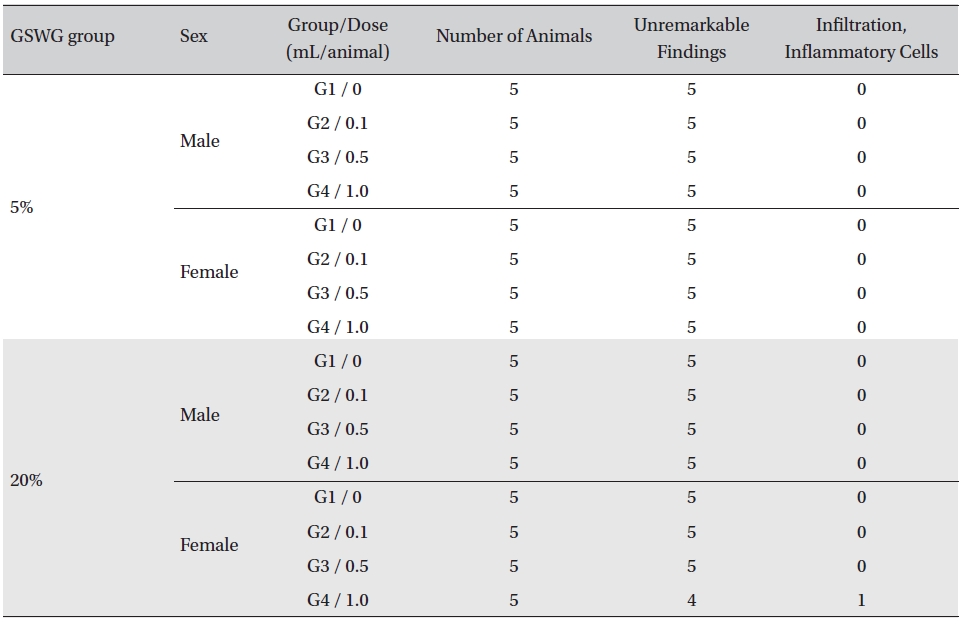
No histopathological abnormalities were observed on the local tolerance tests on the injection sites of all but one out of the 80 animals. Infiltration of inflammatory cells was observed in one animal of the
[Table. 6] Summary of histopathological findings
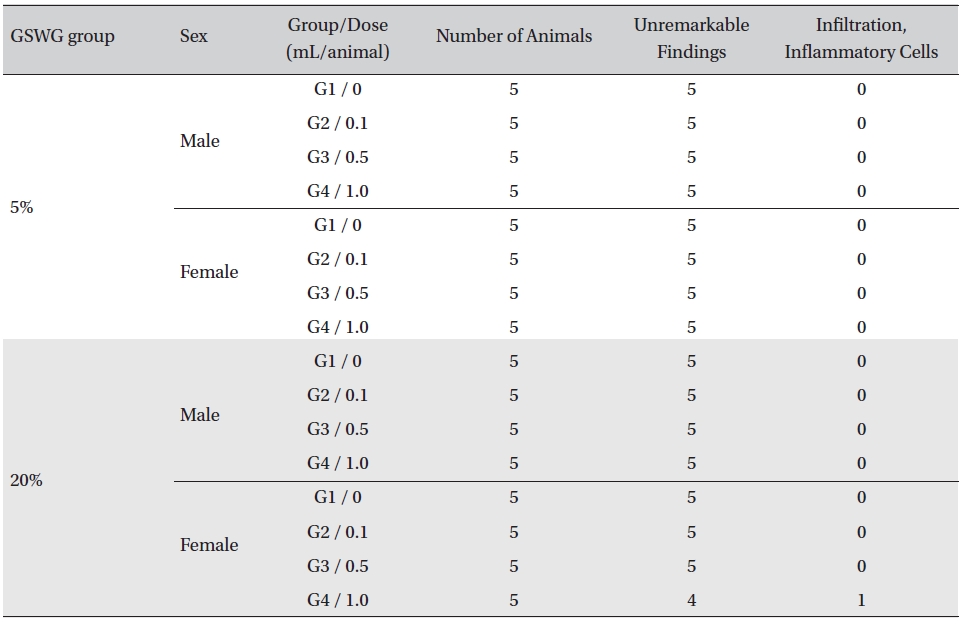
Summary of histopathological findings
The main goal of a toxicity study is to ensure the safety of clinical medicine by evaluating the stability of the new drug. Toxicity can be classified mainly into acute toxicity, sub-acute toxicity and chronic toxicity study [8].
Acute toxicity test results have been reported in Korea for Scolopendra [10], Juglandis Semen [11], Ginseng Radix (wild) [8], Carthami Tinctor-Fructus [12], Triglii Semen [13], Armeniacaeamarum Semen [14], Scutellariae Baicalensis Radix [15], Houttuyniae Herba [16], Salviae Radix [17], Vermilionum [18], Ephedrae Herba [19] and Ginseng Radix [20] as single drugs; and for Bovis calculus + Felursi [21], Bovis calculus + Felursi + Moschus [22] and Hwangryeonhaedoktang [23] as combination drugs. Sweet Bee venom pharmacopuncture [24] has also been tested for toxicity.
From the experimental data obtained in this study, the following conclusions can be drawn: First, the LD50 value could not be obtained as no deaths or abnormalities were observed in any of the groups. In previous studies, Sweet Bee venom [24] had an LD50 value of 0.17 mg/kg and showed abnormalities such as hyperemia and abnormal gait. Ephedrae Herba [19] had an LD50 value of 3885.112 mg/kg, and Triglii Semen [13] had an LD50 value of 0.30mL/kg. Based on these results,
Second, no significant changes in body weights between the experimental groups and the control group were observed during the observation period. In previous studies involving Carthami Tinctor-Fructus [12] and Triglii Semen [13] weight gains were observed.
Third, the variance between measurements from the hematological test was not accepted. This represents a similarity in the toxicity results for pharmacopuncture formulations mentioned above, so
Fourth, some statistically significant results were found on some parts of the biochemical test. However, they were not meaningful in terms of toxicity because of the small variance and the lack of a dose dependency. In the toxicity test for herbs above, Scolopendra [10], Juglandis Semen [11], and Ginseng Radix (wild) [8] showed a significant increase in the total cholesterol, and Armeniacaeamarum Semen [14] and Scutellariae Baicalensis Radix [15] showed increases in glucose.
Fifth, no effects of
A single-dose of
This study was designed to investigate the safety of Guseonwangdo- go glucose 5% and 20% pharmacopuncture. A single-dose IM injection was administered to six-weekold SD rats of both sexes in order to analyze its toxicity and determine a lethal dose. The following results were found:
Therefore, the approximate lethal dose of