LDL cholesterol has been known as bad cholesterol because its high level is closely associated with an increased risk of heart attack and stroke [1]. Recently, a large number of bio-assays for screening high level LDL cholesterol have been reported in the literature since it has been recognized that those assays can greatly increase the chances for successful treatment. They can be classified into three categories according to the technology used: immunosensor utilizing surface plasmon resonance phenomenon on immobilized self-assembled monolayer (SAM) [2,3], amperometric and potentiometric detector to measure electrochemical potential or current generated from the enzymatic reaction of cholesterol [4-8] and spectrophotometer to measure the absorbance of light by the sample at a given wavelength [9,10]. Among these sensors the spectrophotometer is expected to be the most easily applicable to LOD, commonly known as a compact disc (CD) based centrifugal microfluidic platform, because of its noncontact measurement. The spectroscopy technique has been most frequently employed in life science laboratories as a method to determine the concentration of a certain substance in a solution. Spectrophotometry approach to LOD allows the automation of complex fluidic tasks by centrifugal force as well as providing contact-free measurement of bio-chemical reaction. That’s the reason why many biochemical analyzers have been focused on developing spectrophotometry-based LOD [11-13]. Despite such advantages, however, there are still some difficulties in obtaining LOD as thin as a compact disc because of Lambert-Beer’s law that requires a long path length, typically 10 mm that is the distance that light travels through a sample, to offer enough sensitivity as a spectrometer. It consequently led to the development of LOD with a 10 mm thick detection chamber [14,15]. To solve the thickness issue, J. Steigert and M. Grumann [16,17] integrated two V-grooved total internal reflections (TIR) into LOD and thereby achieved the 10 mm path length with no more than about 1.4 mm thick detection cell. However, this approach requires inevitably more designing area to be available on LOD than is traditionally occupied since it needs a detection cell and two V-grooves azimuthally arranged on the LOD.
In this paper, these problems are overcome through the use of a serial reflected face-to-face mirror (SRM) that offers longer path length and at the same time eliminates the necessity of those V-grooves.
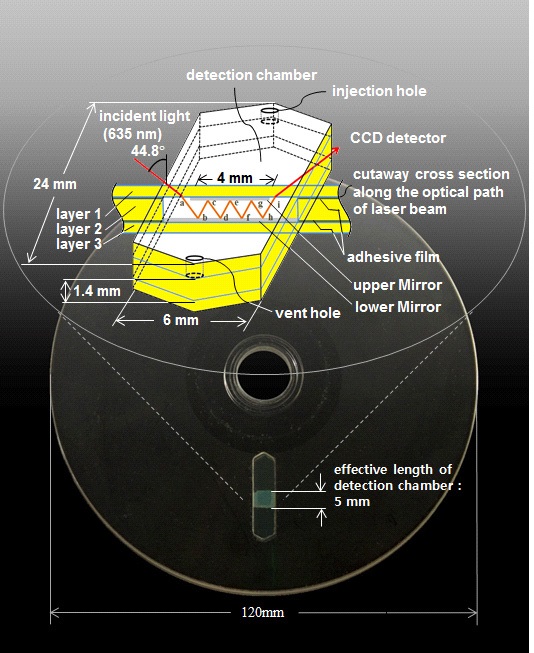
The conventional CD was modified to suit our SRM system made from three polycarbonate layers by an injection molding machine (FA-DRA1F, Panasonic, Japan). Those layers were combined with double sided adhesive films (Tesa 4928, Germany, thickness: 0.125 mm), consisting of a transparent PET film backing and a modified acrylic adhesive, under pressure to finally construct a detection chamber for storing biological samples to be analyzed and to form aface-to face mirror consisting of lower and upper mirrors. Polycarbonate has good properties such as high light transmittance in the visible light range (over 85%), easy molding and low water absorption (0.16~0.35%). The two mirrors were coated with aluminum to a thickness of about 200 nm and were set to face each other, causing multiple reflections between them and in turn making the effective path length of light longer. Fig. 1 shows the whole schematic diagram of the proposed SRM system designed on a compact disc with the diameter of 120 mm where the test sample is contained within a detection chamber with a volume of approximately 50 ㎕. The chamber has a dimension of width 6 mm depth 1.4 mm effective length 5 mm. 99% pure aluminum film was deposited on one side of the polycarbonate layers 1 and 3, by a vacuum decomposition process called cathode-sputtering (SWIVEL 1.8, UNAXIS, USA). Then the layers, with appropriate masks, were placed below
cathode and at some distance from the material being evaporated. The masks placed on the layers allow a specific pattern to be created. The layers 1 and 3 were, respectively, masked by patterns with a rectangular opening of 4 mm×5 mm and 6 mm×5 mm. These opening patterns leave upper and lower mirrors on each surface of the layers after the sputtering cycle.
Our SRM system is designed to have the upper mirror slightly smaller in size compared to the lower one in order to allow the incident light to easily enter the chamber and exit from the chamber at the final reflection point without any interruption. A potential of about 3 kilovolts is applied between the anode and cathode of the sputterer. When the sputtering cycle is completed, the vacuum is released and the mask is removed from the polycarbonate layer, leaving the thin aluminum film for the mirrors on all parts of the layers exposed by the open portions of the mask.
III. SRM (SERIAL REFLECTED FACE-TO-FACE MIRROR) SYSTEM
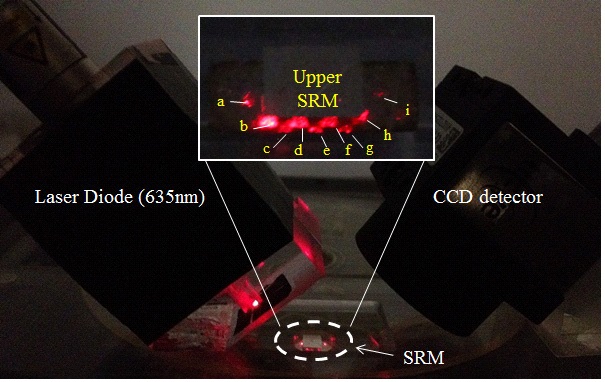
In this experiment, a two dimensional-charge coupled device (2D-CCD) detector is used as a sensor of a spectrophotometer to detect the spectral intensity of LDL cholesterol. The test sample in the chamber is illuminated by laser beam (LM-6305MR, Lanics co., Korea) with wavelength of 635 nm and then the 2D-CCD detector (UI-1220-M, uEye co., German) with 752×480 pixels at 8bit dynamics measures the light that passes through it. First, the incident light passes through layer 1 and starts reflecting when it reaches the surface of the lower mirror. The reflected beam keeps traveling along the face-to-face mirror formed between layers 1 and 3 making multiple reflections and finally arrives at the CCD detector via layer 1 with the absorbance of the light proportional to the concentration of the chemical species that the light passes through (See Fig. 2).
The absorbance (A) can be given according to the Lambert- Beer’s law,
IV. EXPERIMENTAL RESULTS AND DISCUSSION
In this experiment, an enzymatic reaction called COD-POD method was used to determine the concentration of LDL cholesterol dissolved in a standard cholesterol solution. This involves hydrolyzing cholesterol esters by cholesterol esterase (ChE) into LDL cholesterol, and then the LDL cholesterol is oxidased by cholesterol oxidase (COD) to produce 4-Cholestenone and an equimolar amount of hydrogen peroxide (
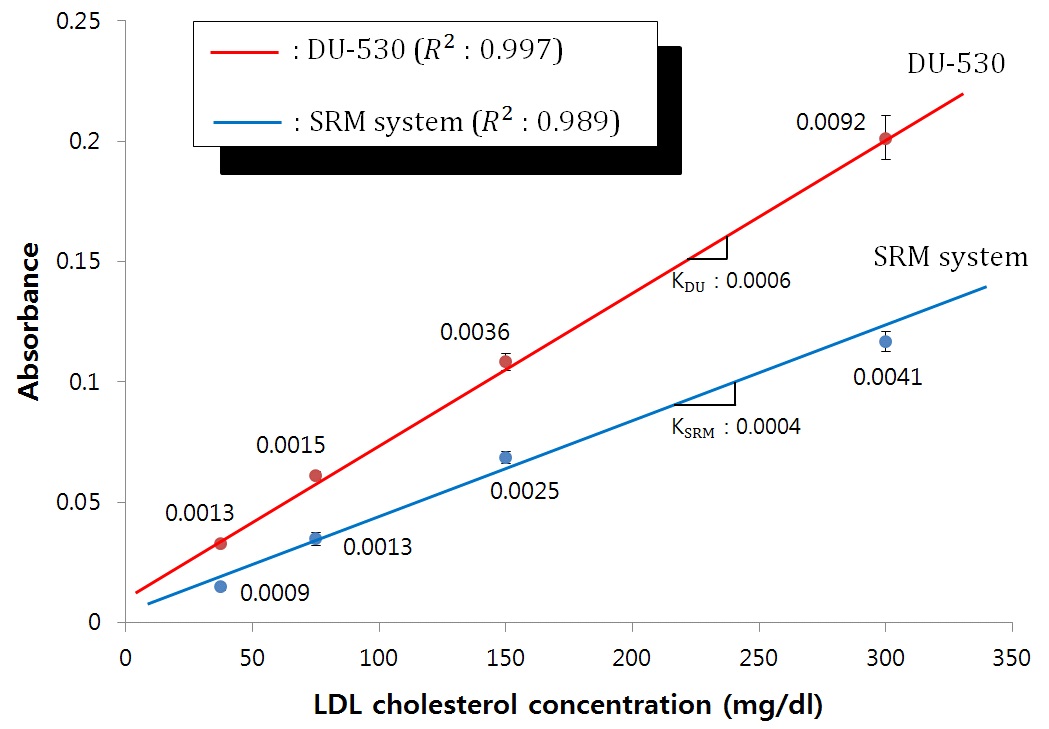
mg/dl are regarded to be borderline high. The curve shows a good linear relationship (
We presented a novel detection device for determining the concentration of LDL cholesterol which uses a SRM buried in the detection chamber. Multiple beam reflections formed during the travel of the light through the chamber gave a path length long enough to determine the LDL cholesterol concentration with almost the same level of sensitivity as the spectrophotometer DU-530 (Beckman, USA). The experimental results show that our proposed SRM system is not only comparable to already commercialized spectrometers in terms of coefficient of variation and coefficients of determination (