At present, 5 synthetic phosphodiesterase type 5 (PDE-5) inhibitors (sildenafil, tadalafil, vardenafil, mirodenafil and udenafil) are used legally for the treatment of penile erectile dysfunction. The market for this medication has grown rapidly, and has become a target of illegal trade. It is necessary to monitor drugs with illicit origins1 because they can cause problems including adverse effects on cardiovascular function such as arterial systemic blood pressure reduction.2 Some of the counterfeit drugs for the treatment of penile erectile dysfunction contain analogues of the five approved compounds, and they are found in various forms of tablets, capsules, herbal medicines, health foods or drinks. Administration of these illegal products may result in severe damage due to improper application or overdose.
Generally, PDE-5 inhibitors and their analogues are analyzed using HPLC3,4 or LC-MS.5 Application of GC-MS is limited mainly for the high molecular weight and low volatility of the compounds, which lead to lower sensitivity and resolution than the HPLC method. However, the conventional HPLC method cannot supply characteristic mass spectral data which can help to discriminate between lots of analogues and chemicals. Even though analysis yielding high sensitivity and selectivity of target compounds is possible by LC-MS, many laboratories cannot apply this method due to the expensive instruments required and high maintenance costs. Recently, a GC-MS method coupled with a short capillary column has been proposed to overcome these difficulties,6 and application of trimethylsilyl (TMS) derivatization has been reported as a way to improve sensitivity for analysis of sildenafil and its N-desmetyl metabolite in hair samples.7 These studies strongly support the practicality of GC-MS for the analysis of the drugs in biological samples. We used GC-MS to analyze 18 kinds of the drugs used for treatment of penile erectile dysfunction that are often applied improperly, and TMS derivatization was applied for the hydroxylated analogues which require further process to increase volatility and sensitivity for GC-MS analysis.
Standards of 18 kinds of PDE-5 inhibitors and their analogues were provided by the Korea Food & Drug Administration (KFDA). The TMS derivatizing reagents, N,O-bis(trimethylsilyl)trifluoroacetamide (BSTFA), N-trimethylsilylimidazole (TMSI) and N-methyl-N-(tertbutyldimethylsilyl) trifluoroacetamide (MTBSTFA), were purchased from Sigma-Aldrich (St. Louis, MO, USA).
>
Sample preparation and derivatization
Each standard solution of the PDE-5 inhibitors and their analogues (100 mg/L, in ethyl acetate) was transferred into a GC vial, and 1 μL of the sample was injected into GC-
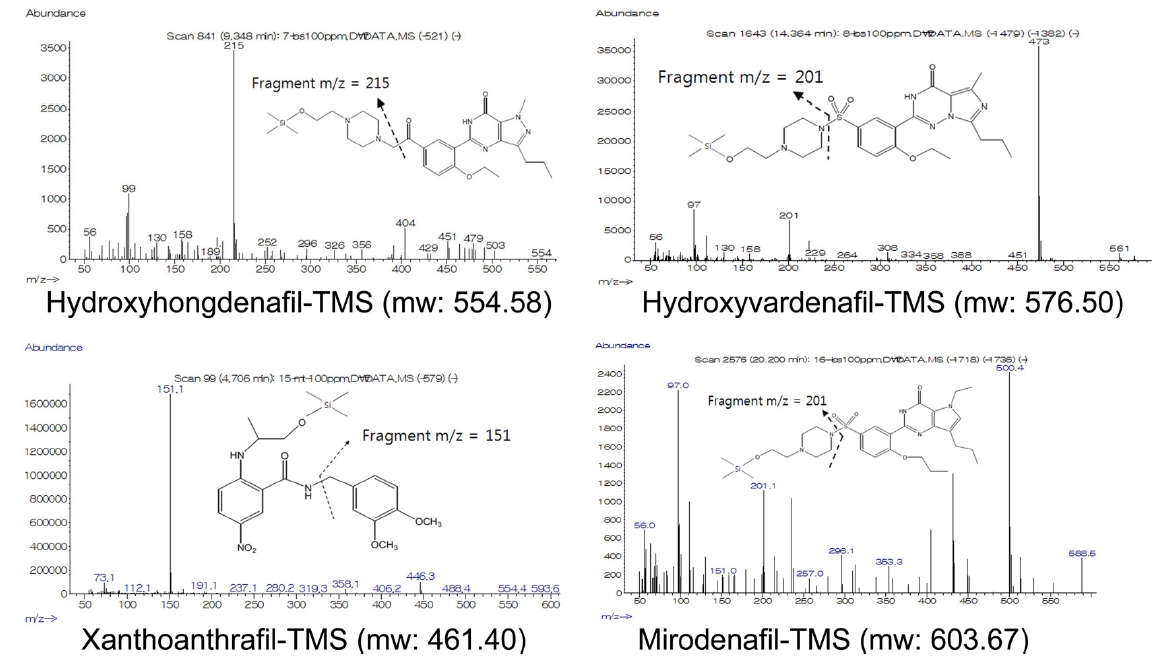
MS. Most of the compounds were detected by this process, and some of them were identified by comparison with the published report7. For some compounds that were not detected by the simple process, sensitivity was improved by TMS derivitization. Among the generally used silylation reagents, BSTFA and MTBSTFA were employed for derivatization of the hydroxylated analogues as they showed better derivatizing efficiency than TMSI. Referring to the previous report8, the derivatization process was established as follows: 30 μL of derivatizing reagent (BSTFA or MTBSTFA) was added to a 10 mL test tube containing 30 μL of the standard solution (100 mg/L, in ethyl acetate). The test tube was capped and derivatized at 90℃ for 40 min. After derivatization, the solvent was removed under nitrogen stream for 3 min. And then, the residue was dissolved with 50 μL of ethyl acetate, the sample solution
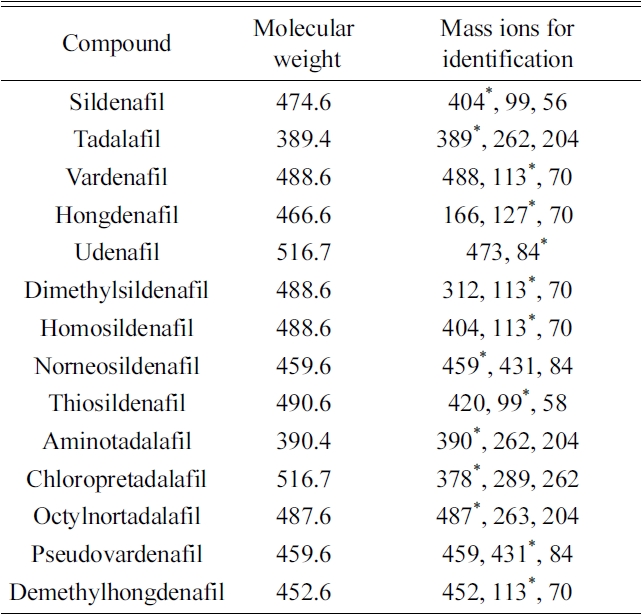
[Table 1.] GC-EI-MS spectra of the 14 PDE-5 inhibitors and their analogues.
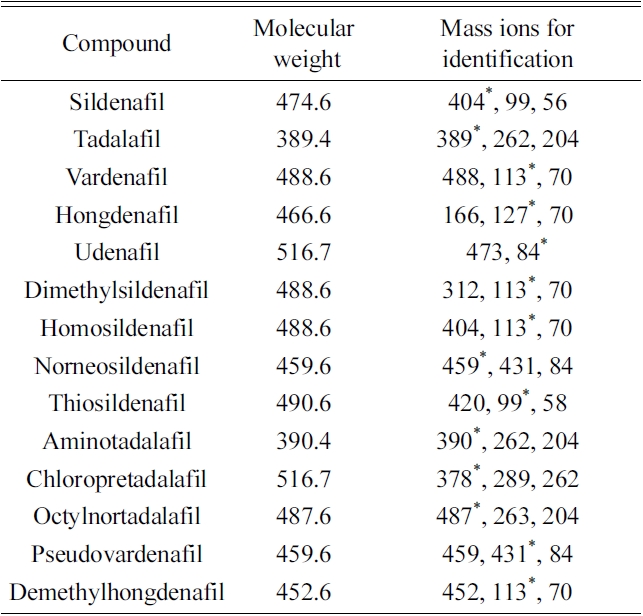
GC-EI-MS spectra of the 14 PDE-5 inhibitors and their analogues.
was moved to a GC vial, and 1 μL of the sample was injected into GC-MS.
GC-MS analyses were performed on an MSD5975C GC/MS system (Agilent Technologies Co., USA) equipped with HP7890A GC, HP7693 autosampler and shortened HP-
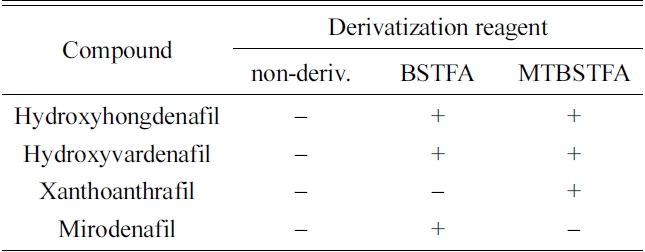
[Table 2.] Availability of TMS derivatization for the hydroxylated compounds.
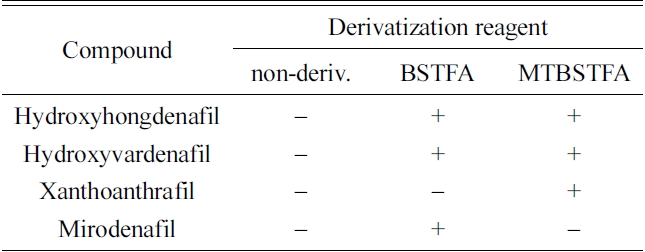
Availability of TMS derivatization for the hydroxylated compounds.
5MS capillary column (0.25 mm i.d. × 15 m length, 0.25 μm film thickness). Oven temperature was set to 290℃ for 25 min, the same temperature as the injector and the transfer line. High purity helium was used as a carrier gas, and the flow rate was 1.2 mL/min. The initial injection mode was splitless, and was then purged after 0.3 min with a split ratio of 50:1.
The electron ionization (EI) mode was used for ionization of the samples, and the electron voltage was set as 70 eV. Acquisition was conducted in scan mode, and the mass range was set as m/z 50-600.
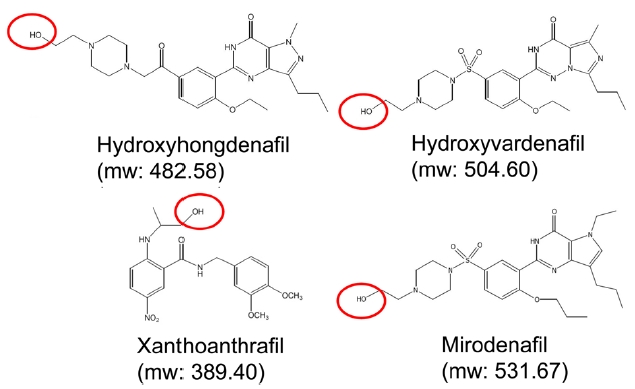
Fourteen compounds, including sildenafil, were identified by GC-MS without derivatiziation (Table 1). However, four compounds with hydroxyl functional groups, mirodenafil, hydroxyhongdenafil, hydroxyvardenafil and xanthoanthrafil (Figure 1), were not detected by this procedure. Additional derivatization processes were performed for the four compounds to improve sensitivity. Hydroxyhongdenafil and hydroxyvardenafil were detected by GC-MS with both reagents, but xanthoanthrafil and mirodenafil were detected selectively with MTBSTFA and BSTFA, respectively (Figure 2 and Table 2). The reactivity of MTBSTFA is similar to BSTFA. But, MTBSTFA can enhance the stability of the derivative reactive to hydrolysis,9 and it may be why xanthoanthrafil was detected only by MTBSTFA derivatization. Whilst, analysis of mirodenafil can be hindered by greater bulkiness of MTBSTFA, and it may explain the selectivity for the TMS reagents.
TMS derivatization of the hydroxylated analogues of the PDE-5 inhibitors significantly improved the sensitivities for the GC-MS analysis, and it also indicates the existence of polar functional groups. Since GC-EI-MS is a highly selective and effective tool for the identification of chemical structure, the method suggested in this study may be used to confirm the results obtained by conventional HPLC method and for the identification of new analogues.
Eighteen of the legally or illegally traded PDE-5 inhibitors and their analogues were analyzed by GC-MS, and a TMS derivatization method was developed to ascertain four compounds with hydroxyl functional groups in their structures. GC-MS method can complement the conventional HPLC method, and it will contribute to prevent the illegal trade of the counterfeit drugs for the treatment of penile erectile dysfunction.