Gastritis appears to result from reproduction of gastric epithelial cells after the gastric mucositis, which is a comm-on disease among digestive diseases [1]. In medicine, there are many drugs for gastritis [2]. Recently developed drugs for gastritis are mainly H2 receptor blockers, because histamine plays an important role in the production of stom-ach acid. In Korean medicine, Atractylodis Rhizoma, roots of
Many gastritis animal models exist, and the H2 receptor blocking effect via compound 48/80 can be evaluated using the acute gastritis model. Compound 48/80 is known to cause degranulation of connective tissue mast cells, but not mucosal mast cells, with the release of serotonin and histamine from the cells [4, 5].
The reactive oxygen species (ROS) is toxic oxygen free radi-cals that cause the oxidative damage involved in cell injury and inflammation [6, 7]. Gastric mucin is known to interact with ROS [8].
So far few reports about the effects of Atractylodis Rhizoma and Atractylodis Rhizoma pharmacopuncture (ARP) on gast-ritis have been reported [9-12]. However, no reports on the protective effects of ARP for acute gastritis, such as the lat-est research trends for acute gastritis induced by compound 48/80, to the best of our knowledge, exist. Then, in the present study, we investigated the protective effects of ARP against acute gastric mucosal lesions induced by compound 48/80. Gastric mucosal thiobarbituric acid reactive substances (TBARS, an index of lipid peroxidation), xanthine oxidase (XO) and superoxide dismutase (SOD, an enzyme to scavenge O2 to H2O2 and O2) were measured for investigating the antioxidative activity as well.
Atractylodis Rhizoma was purchased from Omniherb (Daegu, Korea). ARP was prepared as follows: One-hundred gram (100 g) of Atractylodis Rhizoma in 2,000 ml of distilled water was heated in a heating extractor for 3 hrs. The extracts were filter-ed and concentrated by using a rotary evaporator. The extracts were lyophilized by using a freeze dryer (14.2%). The lyophilized extracts were dissolved in water and filtered three times through microfilter paper (Whatman no. 2, 0.45 - 0.2 ㎛). The ex-tracts were then placed in disinfected vials, which were sealed for further study.
Sodium chloride was purchased from Duksan (Ansan, Gyeon-ggido, Korea). Saccharose and perchloric acid were purchased from Daejung (Siheung, Gyeonggido, Korea). Alcian blue was purchased from BDH Laboratory Supplies (Poole, UK). Sodium phosphate monobasic and sodium phosphate dibasic were purchased from Jin Chemical (Tokyo, Japan). All other reagents were purchased from Sigma-Aldrich (St. Louis, MO, USA).
Male Wistar rats, aged six weeks (weights: about 225 - 235 g), were purchased from Samtaco Co. (Osan, Gyeonggido, Korea). The animals were housed in cages in a ventilated animal room with controlled temperature (23 ± 2°C) and relative humidity (55 ± 5%) and with 12 hrs of light (7:00 to 19:00). The animals had free access to rat chow (Samtaco Co., Osan, Gyeonggido, Korea) and tap water ad libitum for one week. All animals rec-eived humane care in compliance with the guidelines of the Animal Care and Use Committee.
2.4. Gastric mucosal lesion induced by compound 48/80
For the experiment, the rats were divided into 4 groups: normal group, control group, positive control group and experimental group. The number of rats in each group was eight. All rats were 7 weeks old, had fasted for 24 hrs, and had free access to water, but not food, during the experiment.
The normal group and the positive control group were respectively administrated distilled water and famotidine (4 mg/kg) orally. The control group and the experimental group were injected with distilled water and ARP, respectively. The injected acupoints were Joksamni (ST36) on the left and the right legs and Jung-wan (CV12) on the abdomen. The anatomi-cal locations of the stimulated acupoints in the rats were determined in accordance with other reports [13,14]. For the injections, 26 gauge safti syringes (Becton Dickinson Korea, Inc., Seoul, Korea) were inserted into a depth of 2 ? 3 ㎜.
Compound 48/80 (0.75 mg/kg body weight), dissolved in distilled water, was intraperitoneally injected into the control group, the positive control group and the experimental group. In these groups, acute gastric mucosal lesions were induced whithin 1 hr of the above treatment. The normal group received an intraperitoneal injection of an equal volume of distilled water.
The rats were sacrificed under ether anesthesia within in 3 hrs of compound 48/80 injection. The stomachs were removed, inflated with 10 ml of 0.9% NaCl, and put into 10% formalin for 10 mins. The stomachs were then opened along the greater curvature.
2.5. Determinations of gastric adherent mucus and gastric mucosal hexosamine, TBARS, XO, SOD
For the assays of gastric adherent mucus and gastric mucosal hexosamine, TBARS, XO, SOD, gastric mucosal tissues were homogenized in 9 volumes of ice-cold 0.05 M Tris-HCl buffer (pH 7.4). The homogenate was centrifuged at 4°C (10,000 x g, 20 mins), and the resultant supernatant was dialyzed against 100 volumes of the same buffer at 4°C for 24 hrs. Gastric mucosal SOD was assayed using the manufacturer|s protocol (Dojindo, Kumamoto, Japan). XO activity was assessed by measuring the increase in absorbance at 292 nm following the formation of uric acid at 30°C. One unit of this enzyme is defined as the amo-unt of enzyme forming 1 ㎛ of uric acid per min (manufacture-r|s protocol, Cayman chemical, Michigan, USA). Gastric mucosal TBARS was spectrophotometrically determined by using the manufacturer|s protocol (Zeptometrix, New York, USA). Hexosamine obtained from the hydrolyzed mucin was assayed using acetylacetone and Ehrlich|s reagent. Gastric adherent mucus was assayed by using the method of Kitagawa et al. [15] as follows: The removed stomach was cut open along the greater curvature and rinsed with 10 mL of ice-cold 0.25 M sucrose. Then, 50 ㎟ (approx. 8 ㎜ in diameter) of the glandular portion of the stomach was excised with a scalpel, and the excised part was weighed. The excised stomach was soaked in 2㎖ of 0.1% alcian blue, which was dissolved in 0.16 M sucrose buffered with 0.05 M sodium acetate (pH 5.8) for 2 hrs. Uncomp-lexed dye was removed by using two successive washes in 2 ml of 0.25M sucrose for 15 and 45 mins, respectively then, the dye complex with mucus was extracted with 30% DSS for 2 hrs. After centrifugation (3,000 rpm for 10 mins), the optical density of the solution of alcian blue extracted with DSS was read at 620 nm, and the concentration of the extracted alcian blue was calculated in comparison with a calibration curve obtained with alcian blue solutions of known concentrations. The concentr-ation of gastric mucosal adherent mucus is expressed as that of alcian blue adhered to the gastric mucosal surface (㎍/g tissue).
Samples of the corpus were excised, transformed to 10% fresh formalin and later processed by using routine techniques before embedding in paraffin. Sections (2 ㎜ thickness) were mounted on glass slides and stained with successive stainings using alcian blue, hematoxylin, and eosin. Coded slides were exami-ned with a light microscope (BX60, Olympus, Tokyo, Japan) by an experienced pathologist blinded to the treatment.
The results were expressed as means ± standard errors of the mean (SEM). Significances of changes were evaluated using the one-way ANOVA and Dunnett|s post-hoc test (SPSS ver.10.0). Values of p〈 0.05 were considered significant.
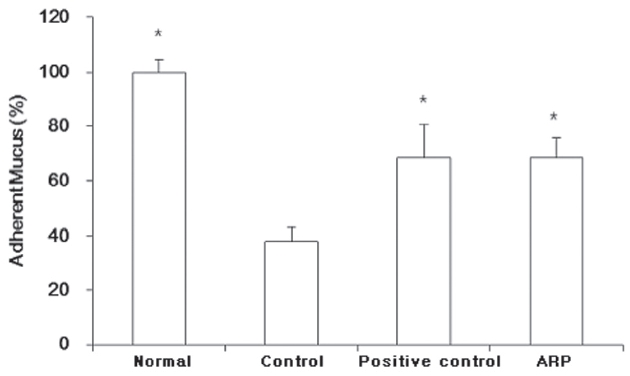
3.1. Effect of ARP on the gastric adherent mucus co-ntent
Gastric adherent mucus in the control group was reduced to 38.2 ± 0.9%. Both the positive control and the ARP groups were significantly protected against the loss of mucus (adherent mucus: 68.5 ± 12.3% and 68.5 ± 7.2%, respectively) (Fig.1 ).
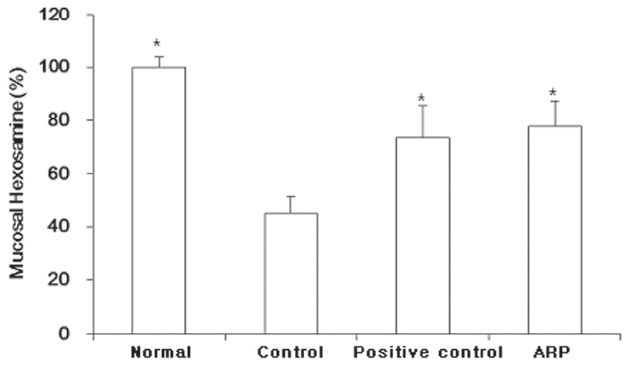
3.2. Effect of ARP on the gastric mucosal hexosami-ne content
Gastric mucosal hexosamine content showed patterns similar to those of gastric adherent mucus. Gastric mucosal hexosami- ne content in the control group was reduced to 45.2 ± 6.2%. Both the positive control and the ARP groups were signifi-cantly protected against the loss of mucus (mucosal hexosam-ine:73.6 ± 11.9% and 77.9 ± 9.6%, respectively) (Fig.2 ).
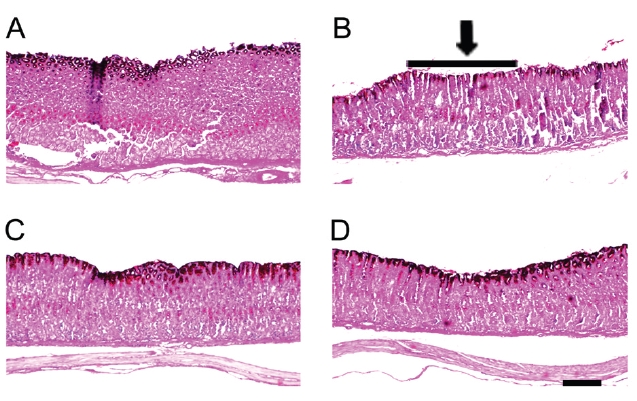
3.3. Effect of ARP on the gastric mucosal lesion
As shown in Fig. 3, gastric mucosal lesions appeared 0.5 hr after treatment with compound 48/80 (Fig.3 B). Alcian blue stained the mucosal layer to dark blue (upper layer in Fig.3 ).The mucosal layer was thinned after compound 48/80 treatm-ent. Both famotidine and ARP treatments protected the mucosal layer (Figs.3 C and D).
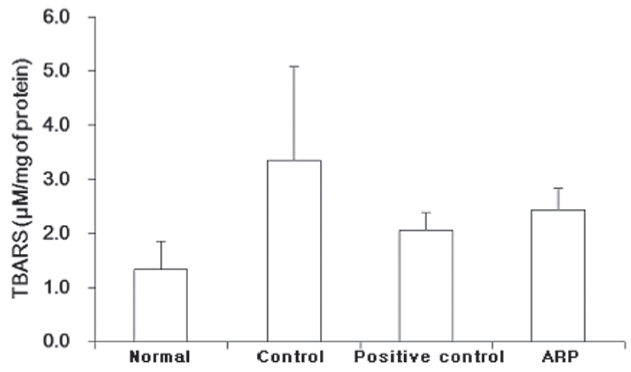
3.4. Effect of ARP on the gastric mucosal TBARS content
TBARS in the normal group were 1.3 ± 0.5
were increased, to 3.4 ± 1.7
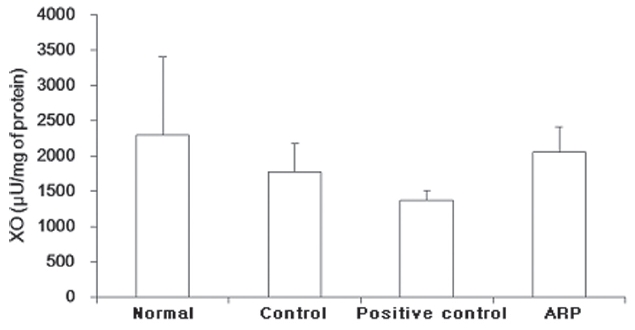
3.5. Effect of ARP on the gastric mucosal XO activity
XO in the normal group was 2288.1 ± 1105.5
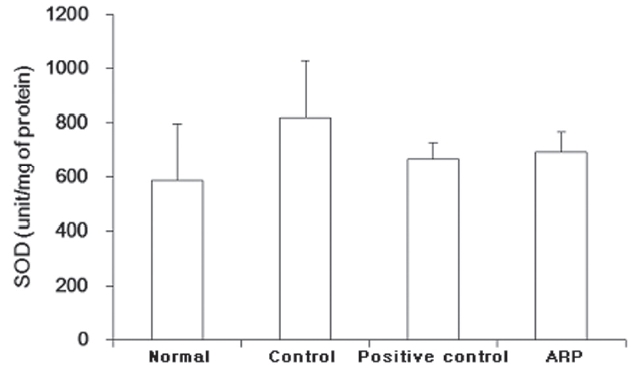
3.6. Effect of ARP on the gastric mucosal SOD activity
SOD in the normal group was 585.7 ± 213.9 U/protein. SOD in the control group was higher, 820.9 ± 209.4 U/protein. The positive control and the ARP groups showed SOD activities of 667.4 ± 62.2% and 694.3 ± 73.7%, respectively (Fig.6 ).
Gastritis should be confirmed based on a diagnosis of infla-mmatory cell in fitbrating the gastric mucosa, but can be identi-fied by flares, edema and ulceration on the gastric mucosa acc-ompanying heartburn, epigastric pain, nausea and vomiting [1].
Experimental studies have investigated the use of Atractylodis Rhizoma for protecting the gastric mucosa and supporting the intestinal immune system, and for its anti-inflammatory effect [9-11]. Treatments used Atractylodis Rhizoma also have effects on mast-cell-mediated inflammatory responses. There effects proceed from a mechanism for regulating mast cells through histamine and H2 receptors [16]. Accordingly, ARP might be thought to have H2 blocking effects. In this study, famotidine (H2 receptor blockers) was used as a positive control for compar-ison with ARP [17].
So far, most reports have studied the use of orally-administe-red Artractylodis Rhizoma for protection of the gastric mucosa and for anti-inflammation in cases of gastritis [9-11]. Kim et al. [12] reported the effects of ARP on acute gastritis. However, some differences, method and acupoints, exist between both Kim et al.|s [12] report and this one. In this study, compound 48/80 was used to induce acute gastric mucosal lesions. Many studies revealed that a single treatment of compound 48/80 in rats could cause gastric mucosal lesions to occur [15, 18, 19]. ARP was injected at Joksamni (ST36) and Jung-wan (CV12) in this study. Both ST36 and CV12 are recognized as the most important acupoints for stomach disorders. ST36 is simultaneously the acupoint of the Stomach meridians, which has an excellent effect on stomach disorders, and the main acupoint of all diseases. CV12, which is the acupoint of the Conception vessel, can be utilized in the treatment of gastric disease due to the relation with Stomach meridians [20].
In the present study, ARP could significantly protect against the loss of gastric adherent mucus. Gastric adherent mucus in control group was 38.2 ± 0.9%, Both the positive control and the ARP groups were significantly protected against the loss of mucus (68.5 ± 12.3% and 68.5 ± 7.2%, respectively). The effect was almost equal to that for the positive control drug, famotidine (Fig.1 ). ARP could also significantly protect against the loss of gastric mucosal hexosamine. The gastric mucosal hexosamine content in the control group was 45.2 ± 6.2%. Both the positive control and the ARP groups were significantly protected against the loss of mucus (73.6 ± 11.9% and 77.9 ±9.6%, respectively).
Increasing the gastric mucosal hexosamine contributes to both the prevention and the healing of gastric mucosal lesions [21]. The protection against the loss of gastric mucosal hexosamine was almost equal to that for the positive control drug, famo-tidine (Fig.2 ). The mucosal lesions caused by the compound 48/80 showed thin mucosal layers (darkly-stained region of gastric tissue) in photomicrographs. ARP and famotidine produced almost the same protection of the mucosal layer as was observed in the normal group (Fig.3 ).
Gastric mucin is well known to interact with ROS [22].
ROS is toxic oxygen free radicals that cause the oxidative damage involved in cell injury and inflammation [6, 7]. Gastric mucin is known to interact with ROS, especially the hydroxyl radical in vitro [8]. The accumulation of ROS can modulate many processes in the gastric epithelium [23].
Concentrations of TBARS are an index of lipid peroxidation and oxidative stress, TBARS being increased by oxidation [24]. In this study, TBARS contents were higher in the control group and were lower in both the famotidine- and ARP-treated groups. TBARS contents in the control group were 3.4 ± 1.7
XO has an increasing tendency for ROS-oxidation [25, 26]. In this study, XO in the control group was slightly decreased to 1769.5 ± 399.4
SOD has an increasing tendency for neutralizing ROS that generate oxidation, there forming TBARS in human organism [7, 24]. In this study, SOD in the control group was increased to 820.9 ± 209.4 U/protein. The normal group showed 585.7 ± 213.9 U/protein. Both the positive control and the experimental (ARP-treated) groups showed lower levels of SOD activities (667.4 ± 62.2% and 694.3 ± 73.7%, respectively). SOD on the contrary was increased after compound 48/80 treatment. Both famotidine and ARP treatments reduced the elevation of SOD activities (Fig.6 ).
In conclusion, ARP showed protective effects of the gastric mucosa against acute gastritis induced by compound 48/80. Although the famotidine treatment group were oral administration as contrasted with the ARP treatment group, which were injection, ARP has coordinative effects on H2 receptor blockers in comparison with famotidine. These results suggest that ARP may have potential as an anti-gastric mucosal lesion treatment. Further studies investigating the mechanis-ms, more effective acupoints, and differences in ARP concent-rations are needed.
The Atractylodis Rhizoma pharmacopuncture (ARP) was injected at Joksamni (ST36) and Jung-wan (CV12) 1 hr before compound 48/80 treatment. The amounts of gastric adherent mucus, gastric mucosal hexosamine, TBARS, XO, and SOD were measured, and histological examinations were performed. The results were as follow;.
1. ARP could significantly protect against the loss of gastric adherent mucus.
2. ARP could significantly protect against the loss of gastric mucosal hexosamine content.
3. ARP could protect the mucosal layer on histological examination.
4. ARP reduced the elevation of TBARS content, but this result had no statistical significance.
5. ARP did not change the XO and the SOD activities.
The results showed that ARP had protective effects against acute gastric mucosal lesions induced by compound 48/80 in rats. Further studies investigating the mechanisms, more effective acupoints, and differences in ARP concentrations are needed.