Conventional activated sludge plants encounter difficulties including inefficient sludge settling and excess formation of scum-foam when operating at a higher sludge retention time. Application of fixed-film bioreactors (FFBs) for biological wastewater treatment has many advantages over suspended-growth bioreactors.1) Fixed-film bioreactors exhibit characteristics such as simple operation, shock load handling, less solid sludge waste,2) and less power requirement for operation.3) Advanced FFBs can utilize a variety of media, and therefore provide high surface area-to-volume ratios allowing enhanced attachment and growth of microorganisms.1,4)
In previous studies, it has been demonstrated that FFBs may provide a very efficient means of nitrification.4) Furthermore, uniform distribution of heterotrophic, nitrifying, and denitrifying microorganisms throughout biofilms have already been observed in FFBs.5-7) In a well developed biofilm, oxidation of organics and nitrification occur simultaneously with denitrification in the same biofilm as denitrifying microorganism develops deep inside the biofilm, where oxygen transfer becomes limited and conditions become anoxic.8,9) Masuda et al.9) reported loss of nitrogen in an rotating biological contractor (RBC) plant treating the leachate. Halling-Sorensen and Hjuler10) have observed the same phenomena in their study using a submerged filter. In the denitrifying biofilm, the pH gradient can be increased inside the biofilm to the point at which precipitation of phosphate can occur,11) and hence phosphorus removal is also possible to some extent.
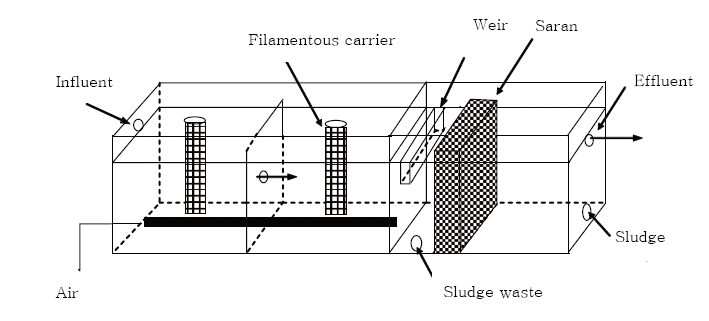
In this study, a FFB combined with a settling tank was used to investigate the effects of organic loading on simultaneous carbon and nitrogen removal. The FFB was composed of a two-compartment aeration tank and a settling tank. In the aeration tank, a nylon filamentous carrier support media was used due to its high area/volume ratio, good mechanical resistance, and suitability for microorganism attachment. In the settling tank, a three-dimensional fibrous assembled polyvinylidene monofilaments media (Saran media) was used as a filter media to aid removal of suspended solids after biological treatment. The Saran media does not absorb water and remains completely unaffected when immersed in water, making it therefore well-suited for use as a filtering media in wastewater treatment. The main features of the bioreactor used in this study, besides the numerous advantages in a fixed-film, are: (i) stationary nature of the support media, which reduces energy expenditure in comparison to moving bed bioreactors, (ii) elimination of short circuiting and dampening shock loading in the bioreactor due to division of the aeration tank into two compartments, and (iii) utilization of Saran media in the settling tank which can enhance the treatability of suspended solids in a short period of time.
The objectives of this study were to evaluate the process performance of a FFB in terms of organic removal, nitrification, and denitrification at different loading rates by varying the influent flow rate, and to determine the efficiency of the settling tank in the presence of submerged Saran media.
The schematic of the FFB used in this study is shown in Fig. 1. The bioreactor was composed of two aeration reactors of equal size, and one settling tank equipped with a baffle. The total volume of the aeration tank was 4 L. The filamentous-carrier support media used for attached microbial growth was a rope-shaped fixed biological contractor (BC Plus; Hyosung, Jakarta, Indonesia) consisting of Y-shaped nylon multifilaments and nylon monofilaments having specific surface area of about 880?1,000 m2/m3 (Fig. 2). The supporting media of each aerobic reactor had a length of 21.5 cm, diameter of 40-45 mm, and packing ratio of 13.5%. The porosity (void) of the filamentous carrier was 95?96%.
The volume of the settling tank was 0.82 L. In this settling tank, a net-type Saran media (Asahi Chemical Co., Ltd., Tokyo, Japan) was immersed, having a porosity of 96.3%, a packing ratio of 17% in occupied volume, and a specific surface area of 400 m2/m3. The filamentous carriers and Saran media used are shown in Fig. 2. The temperature of system was controlled at 20±1℃ during the entire experiment. Compressed air was introduced into aeration tanks using diffusers at the rate of 4 standard liters per minute. The dissolved oxygen (DO) concentration in the aerobic tank was maintained at about 2.5 mg/L.
[Table 1.] Operating conditions adopted in this study

Operating conditions adopted in this study
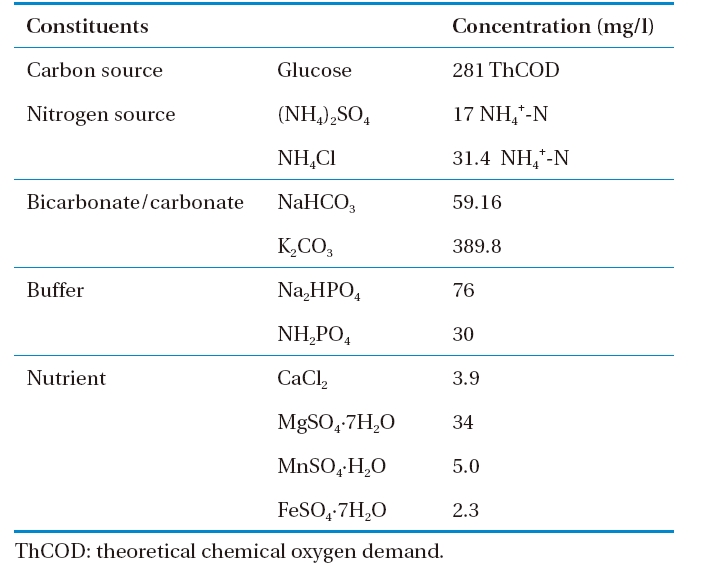
The operational characteristics of the system adopted in this study are given in Table 1. The reactor was operated at three levels of organic loading rates (Runs 1, 2, and 3). The applied organic loadings were varied by changing the flow rate using constant organic (soluble total organic carbon [STOC] concentration=100 mg C/L) and nitrogen strength in the feed solution. The composition of the synthetic feed solution introduced in the system is given in Table 2.12) Glucose was used as a carbon source to prepare synthetic wastewater simulating high strength domestic wastewater (281 mg/L as theoretical chemical oxygen demand [COD]) and a combination of NH4Cl and (NH4)2SO4 was used as a nitrogen source in the synthetic feed solution.
[Table 2.] Composition of synthetic feed solution
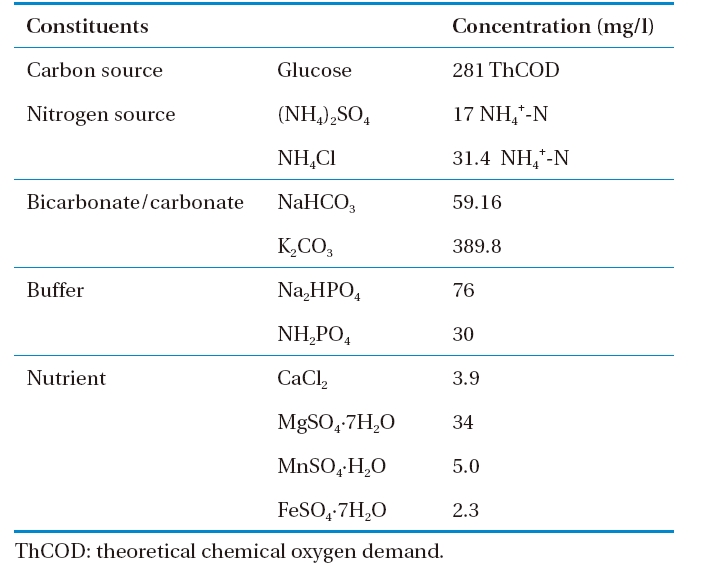
Composition of synthetic feed solution
The reactor was inoculated during the startup period with a mixed microbial culture taken from the return sludge line of a local sewage treatment plant. This culture was acclimated with the synthetic wastewater for a month in a batch mode in order to develop the biofilm on the fibrous media. Air was continuously supplied to the reactor through a diffuser. Consequently, a layer of biofilm developed after two weeks of continuous operation. Thereafter, feed solution was continuously pumped into the reactor. The consistency of developed biofilm on media was monitored visually throughout the trial period.
When the influent flow rate was 13.1, 13.6 and 20 mL/min, the resulting hydraulic retention time (HRT) in the settling tank of each Run was 1.5, 1.0, and 0.68 h, respectively (Table 1). In each case, entrapment of suspended solids was examined by measuring the effluent mixed liquor suspended solids (MLSS) with and without Saran media in the settling tank. The experimental conditions when Saran media was not used in the settling tank served as a control test. In two aeration reactors, the HRT was 7.3, 4.9, and 3.3 h corresponding respectively to Runs 1, 2, and 3. In each Run, the biomass on the fibrous carrier was measured by determining the dry weights of the known length of the media.
The effluents of Compartments 1 and 2, and the settling tank were analyzed every two days for temperature, pH, STOC, ammonia, nitrite, nitrate, total suspended solid, and biomass. Analysis of nitrate and nitrite concentrations was carried out by an ion chromatograph (Dionex model DX-120; Dionex, Sunnyvale, CA, USA). A TOC analyzer (Rosemount Dohrmann DC-180; Mason, Ohio, USA) was used for STOC analysis. All analytical methods were done according to the Standard Method.13)
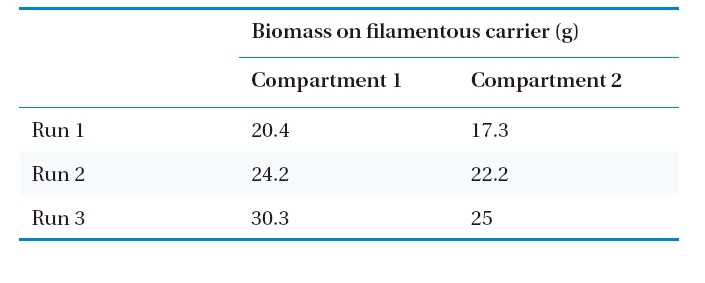
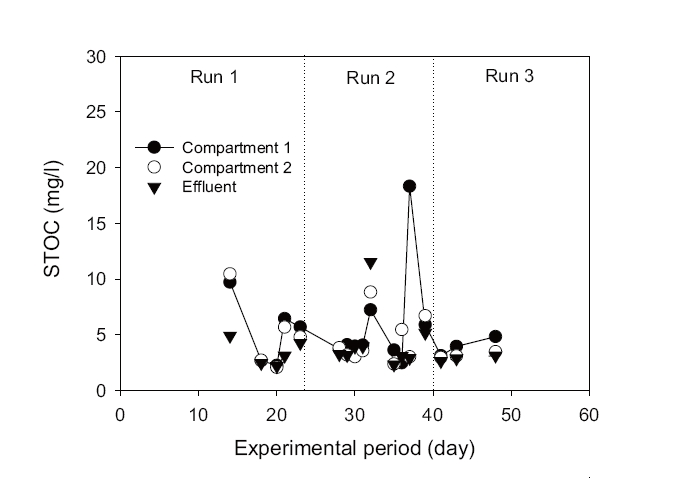
The average organic removal efficiency was about 96% during the entire operation of the bioreactor. The STOC in the influent, Compartments 1 and 2 of the aeration tank, and settling tank at different hydraulic loading rates (Runs 1, 2, and 3) are shown in Fig. 3. It is evident that more than 90% of STOC removal efficiency was achieved in Compartment 1 of the aeration tank during Runs 1, 2, and 3, with applied organic loading rates of 0.92-2.02 kg COD/m3/day. A higher amount of biomass was attached to the filamentous carrier in Compartment 2 than that of Compartment 1 (Table 3), particularly when the organic loading was highest (Run 3). This observation suggests that a high volumetric and a higher surface organic loading rate in Compartment 1 of the aeration tank resulted in large yields of biomass. Therefore, good removal efficiency of organics was observed in Compartment 1, as shown in Fig. 3. The comparatively lower organic loading in Compartment 2 obviously caused a high removal efficiency of STOC in Compartment 1. The STOC concentration remained below 5 mg/L throughout the trial period in the Compartment 2 (Fig. 3), which was suitable for an autotrophic nitrification process in Compartment 2.
The treatment efficiency was found to be stable during the three experimental Runs, even when the inflow rate was increased. The media’s availability of large surface area for microbial growth was one of the major reasons for the stable treatment performance of the system. The applied low HRT (or, in other words, high organic loading) favored microbial growth and produced a high concentration of biomass. Consequently, a higher treatment capacity in Compartments 1 and 2 of the aeration tank, particularly in Compartment 1, was observed. On the other hand, an increased inflow rate induced both normal and shear stress on the biofilm, which enhanced sloughing. The purpose of the introduction of Saran media in the settling tank was to prevent deterioration of effluent quality due to this increased sloughing.
[Table 3.] Amount of biomass on filamentous carrier in the aeration tank
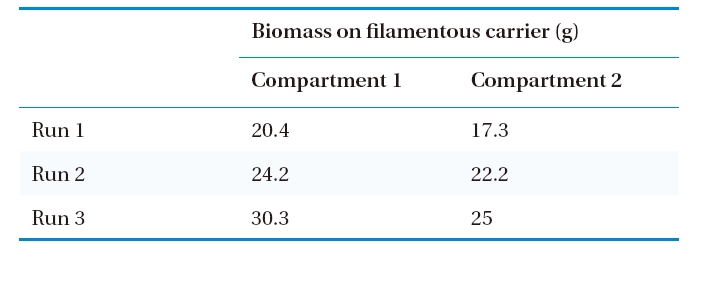
Amount of biomass on filamentous carrier in the aeration tank
3.2. Nitrification and Denitrification
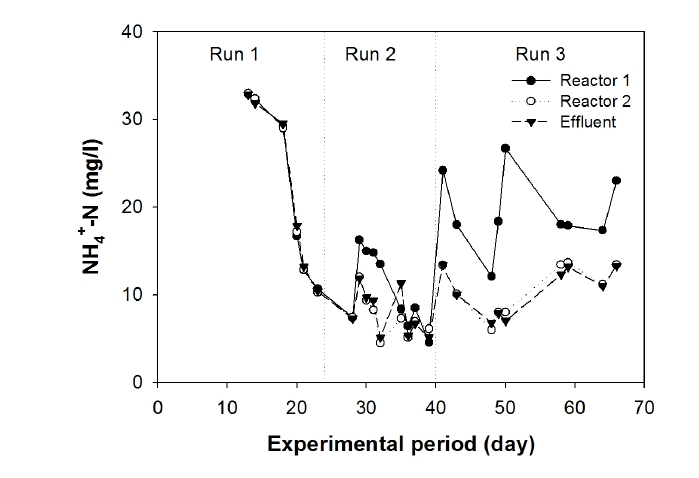
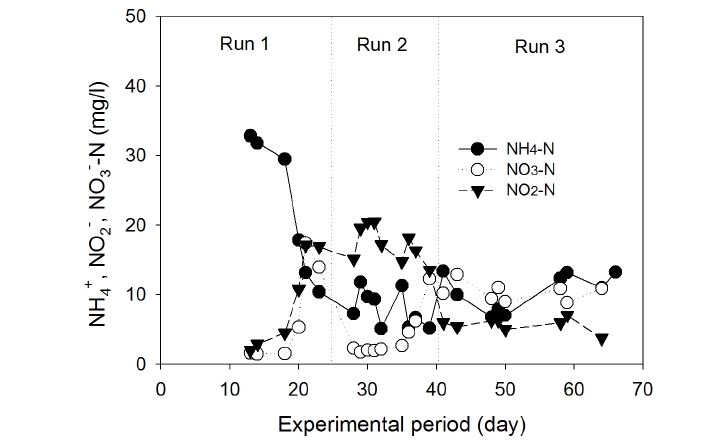
To observe the nitrification and denitrification processes, ammonium-nitrogen concentrations in Compartments 1 and 2 and the settling tank were monitored, as shown in Fig. 4. During Run 1, a decrease in ammonium-nitrogen concentration was observed in Compartments 1 and 2 of the aeration tank, probably due to the dominant nitrification process at a low applied volumetric organic loading rate (i.e. 0.92 kg COD/m3/day). When the volumetric organic loading rate was increased to 1.38 kg COD/m3/day, nitrification initially suppressed in Compartment 1, and a sudden increase in ammonium-nitrogen concentration was observed. However, the ammonium-nitrogen concentration reached below 10 mg/L at day 40 of operation. Further increase in volumetric organic loading rate to 2.02 kg COD/m3/day suppressed nitrification in Compartment 1 and, consequently, fluctuating ammonium- nitrogen concentrations between 10 mg/L to 30 mg/L were observed. However, ammonium nitrogen concentrations in Compartment 2 and effluent were between 10 mg/L to 15 mg/L (Fig. 4). It suggests that nitrification process was dominant in Compartment 2 over that of Compartment 1 of the aeration tank. This observation was clear, and consistent with those reported earlier. For example, Canziani et al.14) used two-stage fixed-bed biofilters for nitrogen removal and found that if the organic loading exceeded 2.5 g COD/m2/day, the nitrification rate was reduced by 50%.
Ammonium-, nitrite-, and nitrate-nitrogen concentration profiles in the effluent of the settling tank are shown in Fig. 5. These results demonstrate that the concentrations of NH4+-N decreased with time during Runs 1, 2, and 3 and then leveled off at 7-14 mg/L. The removal efficiency of ammonia-nitrogen was 70-84% during Run 3 (Fig. 5) despite the higher NH4+-N loading. Total nitrogen (the sum of ammonia, nitrite, and nitrate) removal efficiency during Run 3 was 40?45%, indicating that the denitrification process occurred simultaneously inside the biofilm attached on the fibrous media in the aeration tank. Chui et al.15) studied nitrogen and organic matter in an anoxic/aerobic upflow fixed bed filter and found that nitrogen removal was possible when simultaneous denitrification took place inside the support media where DO was absent.
3.3. Mixed Liquor Suspended Solids Concentration in the Aeration Tank
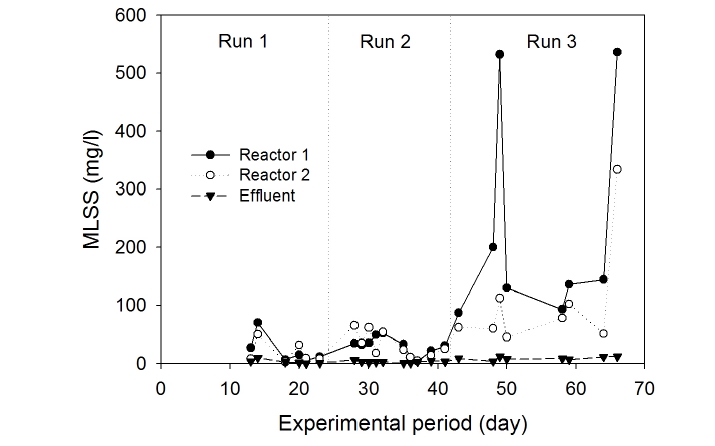
The average MLSS concentrations of bulk solution in Compartments 1 and 2 were low, in the range of 40-60 mg/L, as shown in Fig. 6. Mixed liquor suspended solids increased abruptly in Run 3 compared with Runs 1 and 2. In Compartment 1 in particular, MLSS concentrations fluctuated in the range of 100-500 mg/L. The MLSS concentration in Compartment 2, however, was lower than that in Compartment 1. The fluctuation in MLSS concentration observed in Compartment 1 was due to two reasons: (i) increased applied inflow rate, which caused mechanical detachment of microorganisms from the media, and (ii) higher organic loading rate, which caused suspended microbial growth. The MLSS value during Run 3 was found to be very low compared to conventional activated sludge process (2,000-3,000 mg MLSS/L). It is likely that microorganisms had a tendency to be attached on the supporting media rather than to be in the suspension.
3.4. Effect of Saran Media on Settling Performance
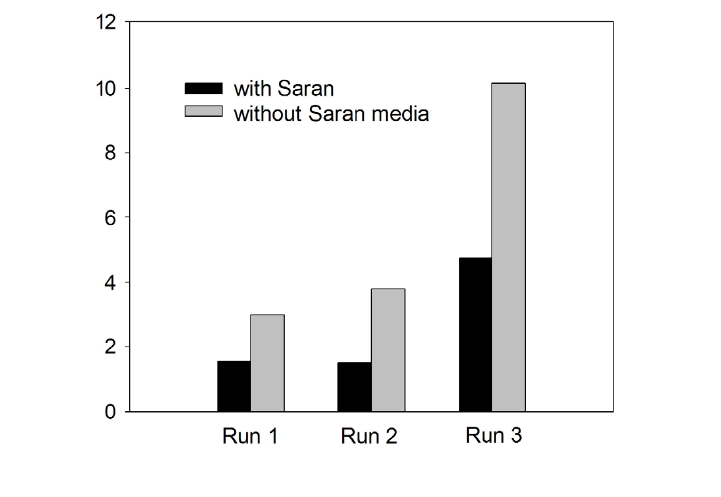
The changes in MLSS concentrations observed in the settling tank during three experimental Runs with and without Saran media are shown in Fig. 7. During Run 1 (HRT of settling tank: 1.5 h) and Run 2 (HRT of settling tank: 1.0 h), suspended solids
removal efficiency in the settling tank with Saran media was found to be slightly higher than that of the control (without Saran media). However, suspended solids removal efficiency with Saran media was about two times better than that without Saran media even during a shorter HRT (i.e. 0.68 h).
The FFB used in this study demonstrated very efficient simultaneous soluble organic and nitrogen removal efficiencies. Due to the presence of a filamentous carrier as the biofilm support media, it was possible to increase the organic loading rate by varying the HRT without biomass washout from the bioreactor. The configuration used in the FFB prevented short circuiting in a short HRT (3.3 h). Stable treatment efficiencies were observed throughout the trial period. A removal efficiency of NH4+-N of up to 84% was achieved at a higher applied organic loading rate (2.02 kg/m3/day). Moreover, the total nitrogen removal efficiency, i.e. of the sum of ammonium-nitrogen, nitrate-nitrogen, and nitrite-nitrogen, was in the range of 40?45%. This removal of total nitrogen leads to the conclusion that the denitrification reaction occurred simultaneously in the biofilm attached to the fibrous media in the aeration tank. The settleablity of suspended solids in the settling tank was enhanced when Saran was used as a supporting media. For identical applied HRTs (0.68 h), the settleability of suspended solid with Saran media in the settling tank was found to be twice as good as when it was not used. In conclusion, the FFB used in this study can be utilized for treatment of domestic wastewater for a short retention time while keeping good effluent quality.