Simultaneous determination of three corticosteroids (clobetasol propionate, betamethasone dipropionate, fluticasone propionate) in moisturizers was performed by using liquid chromatography (LC) coupled with tandem mass spectrometry (MS/MS). Sample preparation was conducted by the liquid-liquid extraction (LLE). Moisturizers include emulsifying agent and it forms micelles. In order to improve the extraction efficiency of corticosteroids trapped in micelle, newly developed-optimized extraction conditions which can remove the matrix effect from moisturizers was applied with various pH conditions in LLE extraction stage of sample preparation. Thus, the addition of 10 μL of 1 M HCl into moisturizers sample before extraction could improve the extraction efficiency. For the quantitative analysis, SRM table that contained specific transition of all of target corticosteroids was created. The developed method was validated for linearity, accuracy, precision, limit of detection (LOD), limit of quantization (LOQ) and recovery. Over the 0.99 r2 value was obtained in calibration standard range. Effective accuracy and precision were also obtained. LODs were below 31 ng/mL and LOQs were estimated below 94 ng/mL for all corticosteroids tested.
Atopic dermatitis is an inflammatory skin disease with chronic and recurrent. Common symptoms of atopic dermatitis can be exacerbated by stress, low humidity, sweat, dust and cigarette smoke.1,2 Thus a high-quality dermatology approved moisturizer should be used daily to avoid the severe dryness, and it should be free of ingredients which may cause side effects.
In general, moisturizers contain a humectant having an excellent moisturizing ability to prevent dryness of the skin and to alleviate itching, and also contain some corticosteroid for being the therapeutic treatment.1 The most used corticosteroids for the treatment of patients with dermatitis3,4 are clobetasol propionate, betamethasone dipropionate and fluticasone propionate corticosteroid. However, there are some reports that moisturizer products do not display the corticosteroids as the ingredients.2 In fact, it was confirmed that corticosteroids have been used as the main active ingredient in ointments intended for the treatment of patients with dermatitis.
As a result, many pediatric patients with atopic dermatitis are constantly using the moisturizer including the corticosteroids whether or not it is marked. It can lead to greater side effects, especially, in the case of children. Thus, it is essential to development of an analytical method for determination of trace amounts of corticosteroids in moisturizers such as lotion, cream and ointment.
There are several methods to detect corticosteroids using a variety of techniques, including HPLC-UV,3 immunoassay,5 GC-MS,4,6,7 LC-MS and LC-MS/MS.8-12 These corticosteroids analysis were performed in human plasma, urine, drug targets or cosmetic products. In order to extract corticosteroids from the real samples, SPE or LLE sample preparation methods with addition of NaOH for hydrolysis of sample matrices were mainly used.13
Here, we present corticosteroids analysis in moisturizers with LLE extraction method and LC-MS/MS. However, moisturizers include emulsifying agent in order to mix the water-soluble constituent and fat-soluble constituent, and it forms micelles. Due to their trapping of the corticosteroids, the complete extraction of corticosteroids from moisturizers can be interfered.13 Thus it is necessary to develop an extraction method to improve the extraction efficiency of corticosteroids from moisturizer matrices (lotion, cream and ointment) as disorganizing the micelle.
In this study, we have developed the simultaneous determination method for the trace amount of corticosteroids with newly developed-optimized extraction conditions which can remove the matrix effect from moisturizers, with applying various pH conditions for LLE extraction stage of sample preparation. From those, this article come to a conclusion that this method was successfully applied to real samples with good validation results.
All reagents were analytical grade unless otherwise specified and used without further purification. Sample dilution was with 50% acetonitrile [acetonitrile and water (50:50 v/v)]. The prepared solutions were stored at 4℃.
We employed the LLE solvent extraction method to extract corticosteroids from moisturizers. 10 μL of 1 MHCl were added to 1 mL of moisturizers.
Mass spectrometry used in this experiment was Quattro Premier triple quadruple analyzer model of Waters equipped with an electrospray ionization interface. Mass scan range was (m/z 150-600). Ion source temperature was 130℃, N2 desolvation gas flow was 800 L/h, and cone gas flow was 50 L/h. Cone voltage was 20 V and capillary voltage was 4.4 kV. Extractor voltage was 4 V and RF Lens voltage was 0.1 V. Ar gas was used as collision gas, and collision cell pressure was 1.00 e-4 mbar. Collision cell entrance potential was 15 V and collision cell exit potential was 1.0 V.
>
Optimization of Tandem MS Detection Conditions
In this experiment, the MS detection optimization experiments were performed using 1 μg/mL standard solutions for each compound by directly infused into ionization source via a syringe pump. We employed electrospray ionization (ESI) and positive ion mode was applied. Clobetasol propionate had an (m/z 467), betamethasone dipropionate had an m/z 505, fluticasone propionate had an (m/z 501) and fluticasone propionate-d3 had an (m/z 504) as protonated molecules ([M+H]+ ion). In the betamethasone dipropionate mass spectrum, metal (Na+) adduct ion peak was showed at (m/z 527, [M+23]).
The fragment ion of [M+H]+ ion of clobetasol propionate was (m/z 373). The fragment ion of [M+H]+ ion of betamethasone dipropionate was (m/z 411), the fragment ion of [M+H]+ ion of fluticasone propionate was (m/z 313) and the fragment ion of [M+H]+ ion of fluticasone propionate-d3 was also (m/z 313).8
>
Optimization of LC Separation Conditions
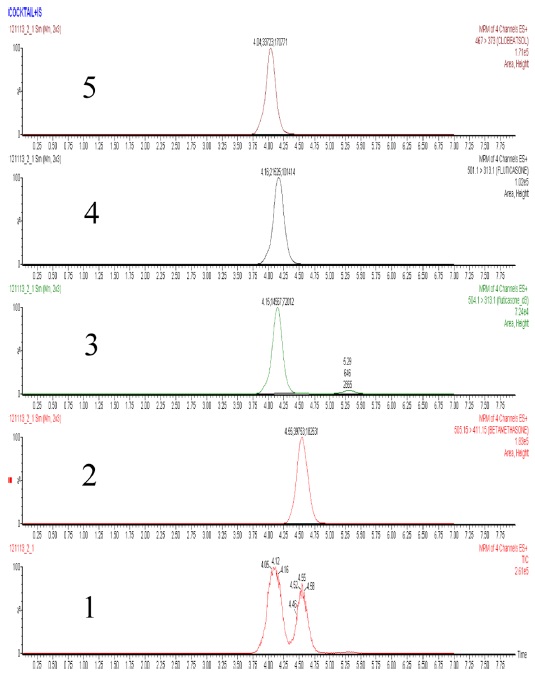
Liquid chromatography analyses were performed with Alliance HT system from Waters. The chromatographic separation was achieved with XBridge C18 column (3.5 μm, 2.1 × 150mm) from Waters. For the aforementioned SRM conditions, LC separation conditions were optimized using acetonitrile and water as mobile phases and isocratic elution. Using 3 μg/mL mixed standard solution of internal standard and analytes, composition of the mobile phases was selected for the optimal separation of the 4 compounds by changing mixed ratio of acetonitrile and water. We chose a mixed solvent composition of 0.1% formic acid in 59% acetonitrile, as the separation of the retention times of the 4 compounds was maximized (no entire separation) and same time the total separation time was less than 8 min at 0.3 mL/min flow rate. LC-MS/MS peaks from clobetasol propionate, betamethasone dipropionate, fluticasone propionate and fluticasone propionate-d3 were observed at 4.0, 4.6, 4.2 and 4.2 min respectively. The peaks for 4 compounds were somewhat overlapped, however, we were able to analyze clobetasol propionate, betamethasone dipropionate and fluticasone propionate without complete separation of SRM (Selected Reaction Monitoring) chromatogram obtained from SRM transition pattern of mass spectrometry. The total ion chromatogram and SRM chromatogram were shown in Figure 1.
Formic acid, an additive to mobile phase, could decrease the pH of solvent and enhance the efficiency of ionization of analyte. Thus we added a small amount of that additive to the mobile phase.
>
Optimization of sample preparation conditions
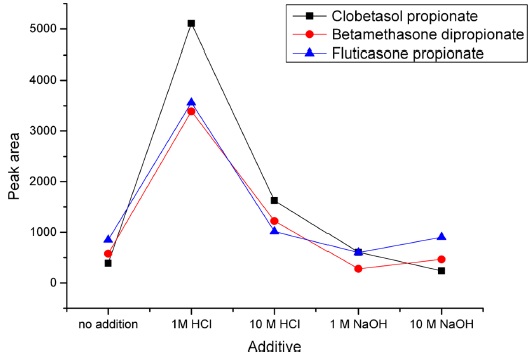
To optimize the extraction conditions of additive, solvent type, solvent volume and extraction time of LLE, 50 μL of mixed standard solution (10 μg/mL) was spiked to base moisturizer and various extraction conditions were experimented. The first, we investigated the effect of additives for acidic or basic solution conditions in order to maximize extractions. Ten microliters of 1 M HCl, 10 MHCl, 1 M NaOH and 10 M NaOH were added into moisturizers before extraction of the corticosteroids and the extraction efficiency was compared (n=3). As a result, the best extraction of corticosteroids was obtained when 1 MHCl was added (Figure 2).
The reason is that an anionic micelle was usually employed as emulsifier in moisturizers. It is expected that corticosteroid compounds are trapped in the anionic micelle, so acid additive could make the micelle disorganized and corticosteroid compounds get free to extract. Second, in order to select the specific extraction organic solvent, the various solvents of methanol, ethanol, acetonitrile and ethyl acetate were tested and compared (n=4). The effective extraction solvent chosen was ethanol. Third, the extraction solvent volume of LLE was optimized. 3, 5, 7 and 9 mL of ethanol was experimented (n=4). As a result, 5 mL of ethanol showed good extraction for all corticosteroids. Next, extraction time (5 min, 10 min and 20 min) of LLE was optimized (n=3). The extraction time was defined as centrifugation time after extraction solvent was added into moisturizer in this study. The difference of extraction efficiency of 10 min and 20 min was not found, thus 20 min of extraction time was selected to extract the corticosteroids.
>
Validation of a Developed Analysis Method
Validation of a Developed Analysis Method The calibration curves were formed for the three corticosteroids. The calibration curves were obtained with standard concentrations of each corticosteroid. For all analytes, the coefficient of correlation (r2) of the calibration curve was greater than 0.99, which indicates a good linearity (n=6).
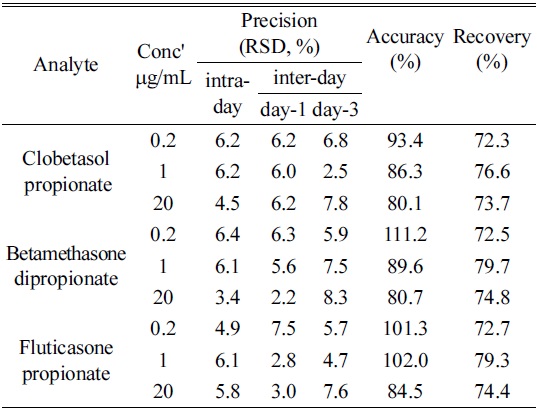
The values of precision, recovery and accuracy are listed in Table 1 (n=6).
[Table 1.] Intra- and inter-day precision, accuracy and recovery of analytical method
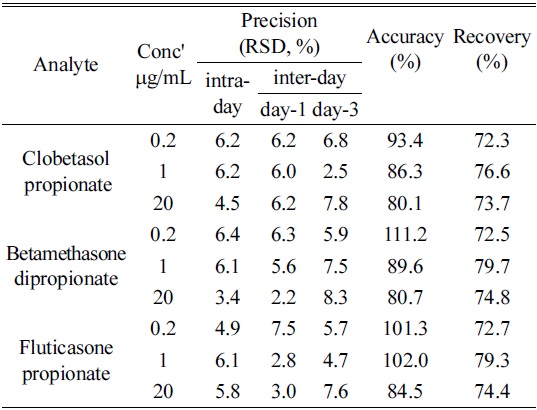
Intra- and inter-day precision, accuracy and recovery of analytical method
The limits of detections (LODs) for analytes, clobetasol propionate, betamethasone dipropionate and fluticasone propionate were 0.031, 0.027, 0.028 μg/mL respectively, and the limits of quantitation (LOQs) were 0.094, 0.081, 0.085 μg/mL respectively. Thus we were able to confirm the possibility of determining even trace amount of corticosteroids in moisturizers.
In order to confirm applicability to real moisturizer samples (lotion, cream and ointments), we applied the developed method to prescribed moisturizer that contain corticosteroids. The relative errors between measured concentration and marked value are in the range of ± 5% for clobetasol propionate, betamethasone dipropionate or fluticasone propionate in moisturizers by this method.
The method for simultaneous determination of three corticosteroids was developed using LC-MS/MS. Liquid-liquid extraction with acidic condition was conducted to extract corticosteroid in moisturizers. The developed method was validated. As a result, this method is suitable for testing moisturizers to check the presence of three corticosteroids in moisturizers.