Diabetes mellitus has become a common disease not only in developed countries but also in developing countries owingto changes in lifestyle and dietary habits (Horton, 1995). α-Glucosidase inhibitors are usually used to prevent or treat type II diabetes (non insulin dependent diabetes mellitus, NIDDM) (Floris
DNJ, a poly-hydroxylated alkaloid, can be considered as a glucose analogue in which the oxygen ring has been replaced with nitrogen. DNJ has potential antidiabetic applications; it inhibits α-glucosidase, which hydrolyzes α-glucose residues within an oligosaccharide chain.
The anti-diabetes mechanism of powder and extracts of the silkworm
DNJ is naturally synthesized by some higher plants, such as the mulberry and dayflower (Yagi
In this study, we developed a microbial DNJ production method as an alternative to chemical methods. Bacteria isolated from soil were screened for DNJ on medium containing each other SFP, SP, and MLP. We obtained four bacteria that produced DNJ in large quantities based on high performance liquid chromatography (HPLC) and thin layer chromatography (TLC).
Silkworm powder (SP), silkworm feces powder (SFP), Mulberry leaf powder (MLP) as nitrogen sources were collected from the Sericulture and Apiculture Division for Department of Agricultural Biology, RDA, Suwon, Republic of Korea. The samples were kept in plastic bags and stored at 4℃ until use. They were dried at room temperature for a few days, ground to a powder by a mortar and pestle, and passed through 150 μm sieves.
>
Fermentation of silkworm powder, silkworm feces powder, and mulberry leaf powder
Distilled water was added to 5% SP, SFP, and MLP. The suspension (100 mL) containing 5% SP, SFP, and MLP was placed in a 150-mL Erlenmeyer flask and sterilized at 121℃ for 20 min in an autoclave. The sterilized suspension was incubated at 37℃ with shaking at 150 rpm for 5 d. To analyze α-glucosidase inhibitor activity, the culture broth was centrifuged at 6,000 rpm for 15 min at 4℃. The supernatant was filtered and analyzed for α-glucosidase inhibitory activity and TLC was performed.
Potential DNJ producers were isolated from soil samples, Doenjang, Gochujang, and other sources. Pure, single colonies of bacteria growing on LB agar plates were isolated. In particular, highly slimy colonies were picked up and inoculated into mulberry leaf powder (MLP) (g/L): K2HPO4 14, KH2PO4 6, MgSO4·7H2O 0.2, (NH4)2SO4 2, MnSO4 0.0017, Fe2(SO4)3 0.028, ZnCl2 0.007, CaCl2 0.15, and glucose 50. Each sample included 1% to 5% MLP as a nitrogen source (Daniel
>
Determination of α-glucosidase inhibitory activity
Sterilized growth medium was inoculated with 1%(v/v) of the isolated bacteria culture suspension and incubated at 37℃ with shaking at 150 rpm for 5 d. The precipitate of the culture broth was removed by centrifugation at 6,000 rpm for 10 min. The resulting mixture was dialyzed against distilled water at 4℃ overnight and used as sample for TLC analyses.
The inhibitory activity of the fermentation broth against α-glucosidase was determined by a reaction between α-glucosidase and 4-nitrophenyl α-Ɗ-glucopyranoside (4-NPG) according to the protocol of Yamaki and Mori (2006). The fermentation broth was serially diluted with an equal volume of distilled water and dispensed into wells of a plate (20 μL per well), followed by the addition of 5 μL of a suspension of rat intestine acetone powder (Sigma-Aldrich, St. Louis, MO, USA), 12 mM 4-NPG (50 μL) as a substrate, and 75 μL of 0.1M potassium phosphate buffer (pH 6.8). The mixture was incubated at 37℃ for 35 min to allow α-glucosidase to react with 4-NPG and produce 4-nitrophenol. The reaction was terminated via the addition of Na2CO3 (50 μL, 200 mM). The formation of 4-nitrophenol in each well was measured based on absorbance intensity at 405 nm using a microplate reader (Model Synergy HT; BioTek Instruments Korea, Ltd., Seoul, Korea).
< Calculation >
Inhibition (%) = A405(inhibition) – A405(control) / A405(enzyme) - A405(blank) x 100
To test the α-glucosidase inhibitory activity in the culture supernatants of the isolated strains, a TLC analysis was performed by spotting 10 μL of the supernatant of the culture broths onto Silica Gel 60 F254 TLC plates (Merck, Darmstadt Germany) and separating the samples in a TLC chamber containing a solvent mixture of butanol-propanol-acetic acid-water (5: 3: 3: 1.5, v/v/v/v) at room temperature. As the standard control, 10 mM glucose and various disaccharides (10 mM fructose, maltose, sucrose) were spotted under the same conditions. They were visualized by spraying the TLC plate with aniline-diphenylamine reagent and heating the plate to 100℃ for 3 min.
>
Sample preparation procedures for crude DNJ
Sterilized growth medium was inoculated with 1%(v/v) of the isolated bacteria culture suspension and incubated at 37℃ with shaking at 150 rpm for 5 d. The precipitate of the culture broth was removed by centrifugation at 6,000 rpm for 10 min. Three volumes of cold ethanol were added to the supernatant to precipitate DNJ, which was recovered by centrifugation as described above. The precipitate was lyophilized and dissolved in 10 mM Tris/HCl buffer (pH 8.0). The resulting mixture was dialyzed against distilled water at 4℃ overnight and used as a sample for the HPLC analysis.
>
HPLC analysis for DNJ measurement
The DNJ crude extract was mixed with 0.4 M borate buffer (10 μL), 5mM FMOC-CI (9-fluorenylmethyl chloroformate) (20 μL), and then the mixture was reacted on 20℃ for 20 min. Finally, 0.1 M glycine (10 μL) was added to the sample, which was adjusted to 950 μL with 10% acetic acid.
To measure the DNJ content of test materials, HPLC was used SHISEIDO SP3203 for fluorescence detection (EM 254, EX 322) and a C18 (100 x 4.6mm, ID 3 μm) column. The absorbance of the effluent was monitored at 254 nm and the flow rate was 1 mL/min. The mobile phase consisted of solvent A (acetonitrile) and solvent B (0.1% aqueous acetic acid). The initial solvent condition was solvent A: solvent B (20:80, v/v) for 16 min. After each analysis, a gradient was used to solvent A: solvent B (40:60, v/v) for 16 min, A: B (80:20, v/v) for 16.1, A: B (80:20, v/v) for 20 min, and A: B (20:80, v/v) for 20.1 min. The standard material was 1-Deoxynojirimycin hydrochloride (Sigma, 10 mg) and the DNJ content was calculated.
Each experiment was carried out in triplicate. All data are presented as the averages of three independent experiments and were analyzed by SPSS (version 18.0). Results are expressed as means ± standard deviation (SD). Results were considered significant at
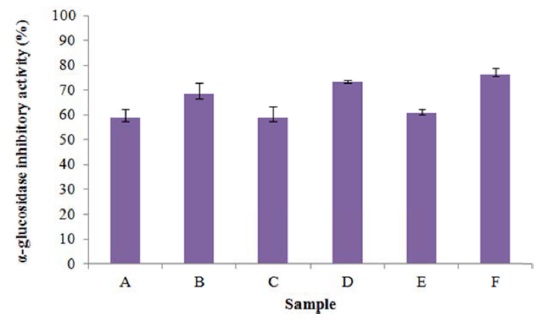
To examine the use of silkworm powder (SP), silkworm feces powder (SFP), mulberry leaf powder (MLP) as nitrogen sources in the culture broth for the inoculated microorganisms, the α-glucosidase inhibitory activity of these powders was monitored during pre-fermentation (Fig. 1). When the sericultural products were fermented for 5 d, α-glucosidase inhibitory activity increased during pre-fermentation. In particular, when MLP was used, α-glucosidase inhibitory activity was greater than 70%. This result indicates that MLP fermentation was optimal for DNJ production. Mulberry leaves are cultivated in many Asian countries including China, Korea, Japan and Thailand. The infusion of its leaves is consumed as antihyperglycemic nutraceutical foods by patients with diabetes mellitus (Kim
The results of this study demonstrate that there is great variation in α-glucosidase inhibitory activity among collected sericultural products. These differences are probably explained by the variation in processing techniques and microorganisms. Previous studies have shown that fermented soybeans products possess anti-diabetic properties (Fujita
>
TLC analysis for α-glucosidase inhibitory capacity from fermented sericultural products
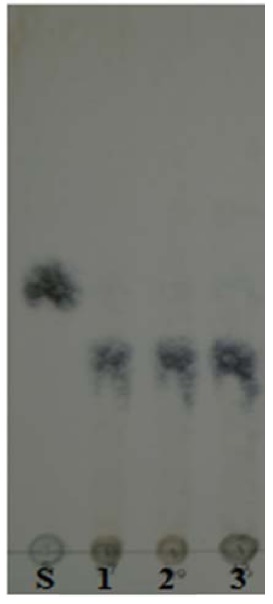
Various chromatographic systems for the analysis of natural products by TLC have been developed. The optimal separation of substances in dietetic products has been achieved on glass-backed precoated silica gel plates and developing reagent as mobile phase. Chromatograms obtained by the analysis of standard solutions of glucose, fructose, sucrose on TLC are shown in Fig. 2 (A). To investigate the α-glucosidase inhibitory capacity of fermented products, TLC was performed. As shown in Fig. 2 (B), we confirmed that disaccarides did not degrade to monosaccarides. α-Glucosidase function in degrading disaccharides to monosaccharide.
Generally, α-glucosidase hydrolyzes terminal non-reducing1-4 linked α-glucose residues to release a single α-glucose molecule. DNJ participates in α-glucosidase activity and inhibits mammalian glucosidase I and glucosidase II. It also inhibits intestinal and lysosomal α-glucosidases, beta-glucosidase from sweet almonds, pancreatic α-amylase, and amyloglucosidase (Romaniouk
>
Screening of DNJ-producing bacteria with α-glucosidase inhibitory activity
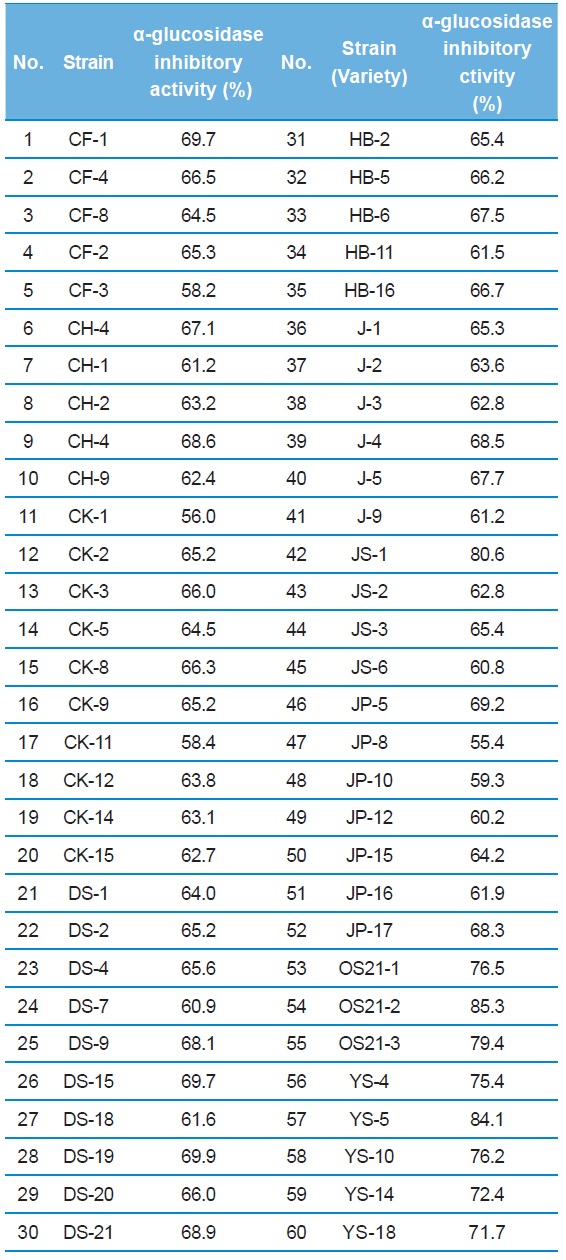
More than 350 colonies of bacteria were isolated from various natural samples. Approxymately 60 colonies were selected as candidates DNJ producers (Table 1). On LB agar plates, 3 strains (JS-1, OS21-2, and YS-5) exhibited a
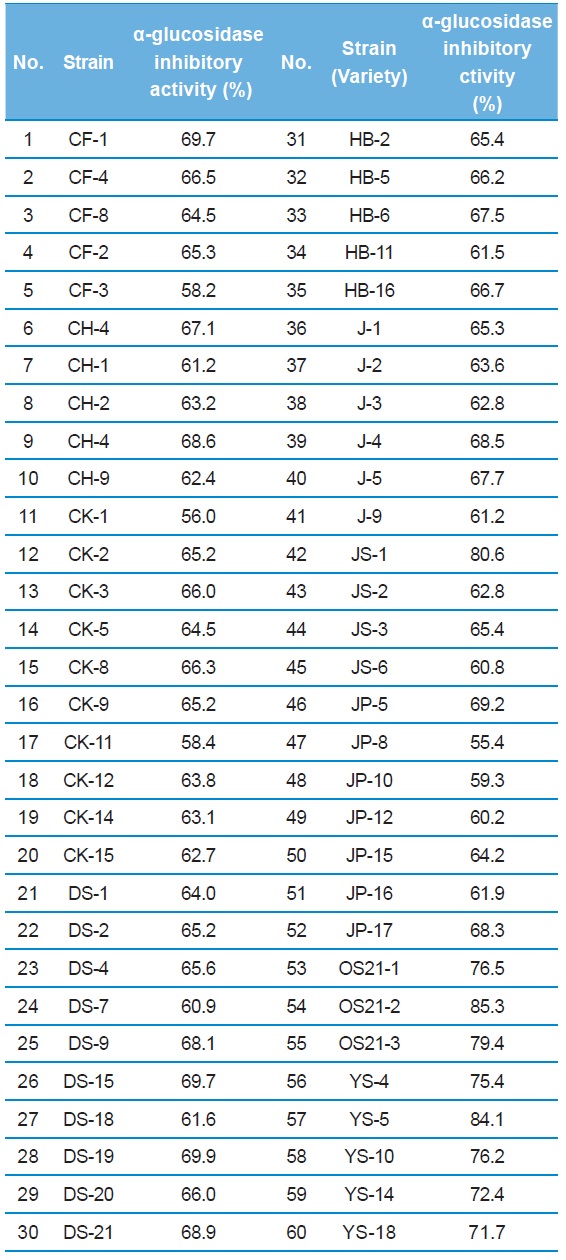
α-glucosidase inhibitory activity of culture broth from the 60 strains in mulberry leaf powder as nitrogen source
>
Effect of mulberry leaf powder (MLP) concentration
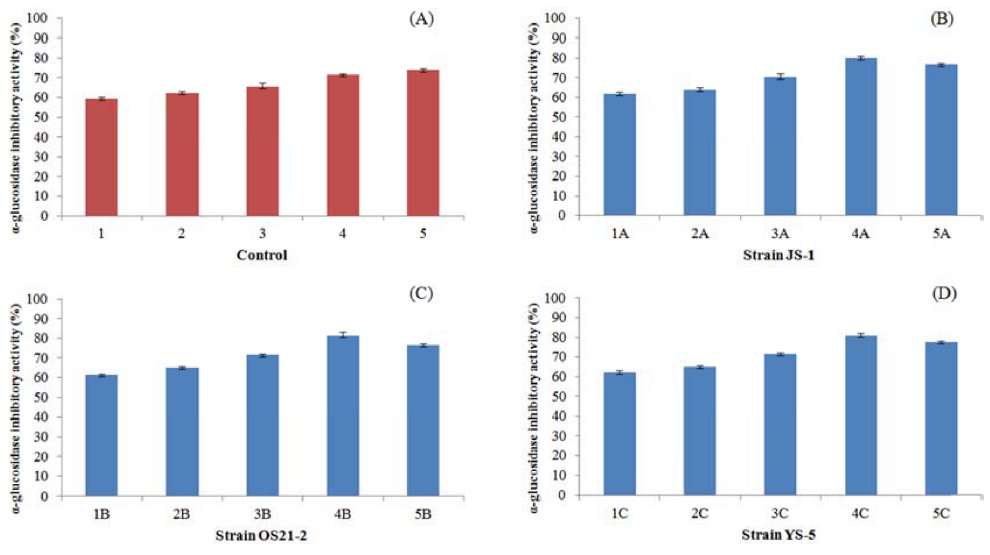
Figure 3 shows the α-Glucosidase inhibitory activity for DNJ produced by mulberry leaf powder (at various concentration) fermented with isolated strains (JS-1, OS21-2, YS-5). When the 4% mulberry leaf powder was used, α-glucosidase inhibitory activity increased (Fig. 3.). It has been suggested that different nitrogen sources may play an important role in the synthesis of α-glucosidase inhibitors because they affect the synthesis of enzymes related to the α-glucosidase inhibitor. This result was consistent with those of a previous report by Zheng
>
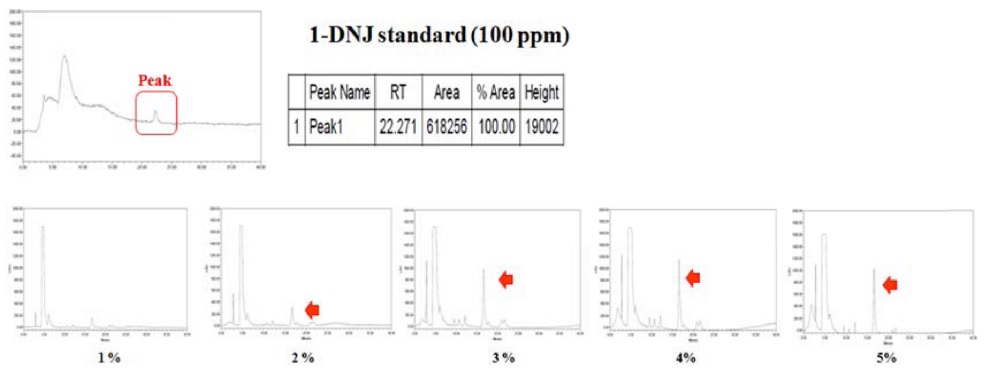
HPLC analysis for DNJ contents
Figure 4 presents a quantitative analysis of DNJ production based on HPLC. When it measured 100 ppm DNJ, it was that result. When the 4% mulberry leaf powder was used as a nitrogen source, the DNJ yield increased by 1.5 times, and α-glucosidase inhibitory activity increased by 10-20% (Fig. 5.). Generally, the DNJ content of mulberry powder, ranges from 0.32% to 0.47% (Konno
In previous reports, the relative DNJ contents of parasitic
These limitations has resulted in the heavy use of chemicals and energy for extraction & purification, causing environmental pollution. Microorganism fermentation processing is a low-cost and high efficiency alternative. The production of DNJ from microorganisms will contribute to the development of commercial scale production and related applications.