The particle sizes of 0.95(K0.5Na0.5)NbO3-0.05BaTiO3 powder were controlled by secondary milling time after calcination. The average particle sizes, Dmean, of 0.95(K0.5Na0.5)NbO3-0.05BaTiO3 powders were critically changed from 14.31 μm to 0.91 μm by secondary milling time. The dielectric and piezoelectric properties of 0.95(K0.5Na0.5)NbO3-0.05BaTiO3 ceramics depended on the particle sizes of powders after calcination and the secondary milling process. As secondary milling times after calcination were increased to more than 48 hr, the dielectric and piezoelectric properties of 0.95(K0.5Na0.5)NbO3-0.05BaTiO3 ceramics were deteriorated.
Due to its excellent piezoelectric properties, perovskite Pb(ZrxTi1−x)O3 (PZT) based ceramics are widely used in sensors and actuators [1]. However, it is well known that PZT ceramics have disadvantages, which generate toxic PbO in the sintering process and compositional fluctuation through PbO evaporation. To overcome these problems, extensive research has been conducted on Pb-free piezoelectric ceramics, such as the Bi layer-type [2], Tungsten-bronze type [3], BaTiO3 based piezoelectric ceramics [4] and alkaline niobate-based perovskite ceramics [5-6]. Among them, potassium-sodium niobate((K0.5Na0.5)NbO3, KNN) ceramics have attracted much attention because they are eco-friendly and have good electrical properties [7]. However, it is difficult to obtain KNN ceramics, having dense and good electrical properties, when sintered under an atmospheric condition. It was found that K and Na ions can be evaporated easily in the high temperature sintering process and cause deviation of compositional stoichiometry. Therefore, a lot of research has been conducted to improve electrical properties of KNN ceramics by additions of aids and changes of the sintering process [8,9]. The BaTiO3 (BT) ceramic has been used widely as capacitors, sensors, and actuators, due to its excellent dielectric and piezoelectric properties, with the exception of its low Curie temperature and high sintering temperature. The combination of KNN ceramics with BT composition showed new possibilities to extend applications of lead-free piezoelectric ceramics. However, there are few studies about the effects of the KNN-BT powder condition, such as particle size, on dielectric and piezoelectric properties.
In this study, we fabricated 0.95(K0.5Na0.5)NbO3-0.05BaTiO3 ceramics that were different particle sizes by changing the secondary milling time after calcination. We then, investigated the effects of particle size on the dielectric and piezoelectric properties.
K2CO3 ( Junsei Chemical, Japan, 99.0%), Na2CO3 (Kanto Chemical, Japan, 99.5%), Nb2O5 (Junsei Chemical, Japan, 99.9%), BaCO3 (Kanto Chemical, Japan, 99.0%), and TiO2 (Hanawa Chemical, Japan, 99.0%) were used as raw materials to prepare 0.95(K0.5Na0.5)NbO3-0.05BaTiO3 (KNN-BT) ceramics using the conventional mixed oxide method. The powders were mixed by ball-milling in ethanol for 24 hr and dried in an oven at 100℃ for 24 hr. The dried powders were calcined at 900℃ for 24 hr and the calcined powders were underwent secondary milling for 0 hr, 24 hr, 48 hr and 72 hr to investigate the effects of particle size on the dielectric and piezoelectric properties of KNN-BT ceramics. The calcined and secondary milled powders were mixed with a 3 wt% polyvinyl alcohol (PVA) solution, and then compacted at a 1.5 ton pressure to form the pellets with a size of 15.8 mm in diameter and 2mm in thickness. The disks were sintered at 1,200℃ for 2 hr with a rising rate of 3℃/min at atmospheric pressure. In addition, silver pastes were fired on both surfaces of the sintered specimens at 600℃ for 30 min for the electrodes. Subsequently, the specimens were poled in a silicon oil bath at 100℃ by applying a DC field of 3 kV/mm for 30 min.
The particle size of the powders after calcination and the secondary milling (0 hr, 24 hr, 48 hr, and 72 hr) process was analyzed by using a particle size analyzer (Cilas 1190, France). The bulk density of the sintered KNN-BT ceramics was measured and calculated using an Archimedes method. The crystal structures of all KNN-BT ceramics were examined by X-ray diffraction (XRD, Rigaku, Japan) with Cu Kα radiation (step:0.01°) to analyze the phases of the KNN-BT ceramics. A JSM-7001F scanning electron microscope (SEM, JEOL, Japan) was used for the microstructural analysis of the KNN-BT ceramics. The temperature dependence of the dielectric constant of the KNN-BT ceramics was examined using an LCR meter (Ando, AG-4303) in an electric furnace by measuring the capacitance in the range from room temperature to 500℃ and 1 kHz. The electromechanical coupling factor (
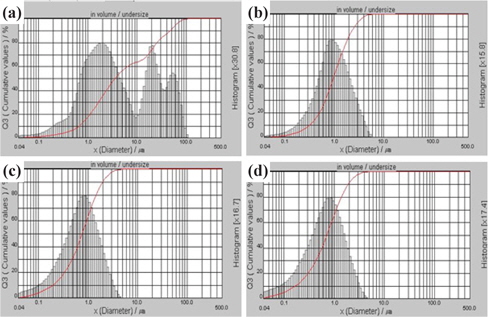
Figure 1 shows particle size distributions of the KNN-BT powders after calcination and secondary milling times of: a) 0 hr, b) 24 hr, c) 48 hr and d) 72 hr. Average particle sizes, Dmean, of the NKN-BT powders after calcination and secondary milling were 14.31 μm, 1.27 μm, 0.96 μm and 0.91 μm, respectively. The particle size distribution of the powder without the secondary milling process (0 hr) showed double peak distribution and, consequentially, had a high value of Dmean. In addition, we can see that the amount of submicrometer particle powders increased at conditions when the secondary milling time was more than 48 hr. From the above results, we can estimate that the excess secondary milling process after calcination increased the amount of submicrometer particle powders. Subsequently, the submicrometer particle powders may easily be lost or agglomerated with each other during the process.
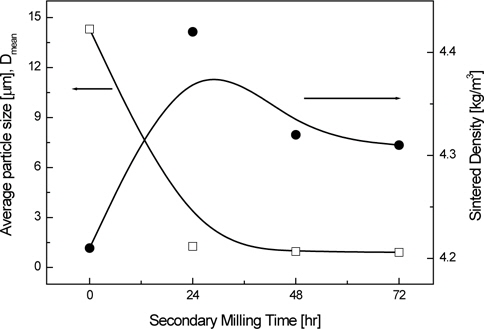
Figure 2 shows the relationship between the variations in the average particle size and sintered density of the NKN-BT ceramics, and secondary milling time. As secondary milling time increases and particle sizes decrease, the sintered density increased and then dropped. We can interpret this as follows: the powder that is a larger size has a lower packing density of particles in lattices and, consequently, low sintered density. On the other hand, the lowered sintered density by submicrometer particle powders that are smaller than 1 μm could be due to the formation of the liquid phases.
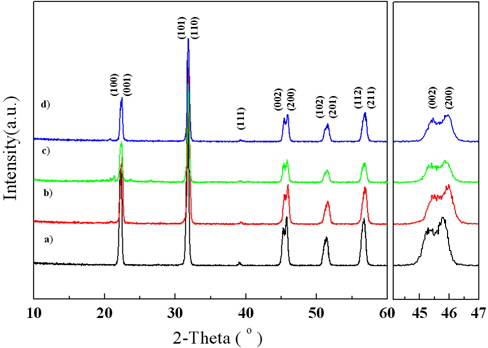
Figure 3 shows the XRD patterns of the sintered KNN-BT ceramics. All the sintered KN-BT ceramics had a single phase with tetragonal structures and a homogeneous phase without any second phases. Furthermore, we can see that the (200) peak weakened and the space distance between the (002) and (200) peaks increased gradually. As the secondary milling timeincreased, the Na and K ions may have undergone much loss, but the amount of K ions might be smaller. Therefore,this might cause the higher content of K ions in all KNN-BT ceramics. Therefore, the increased space distance between the (002) and (200) peaks of KNN-BT ceramics can be explained asoccupying more of the K ion (1.33 Å) than that of the Na ion (0.97 Å) in the A-site as secondary milling time increases [10].
Figure 4 shows the SEM images of the KNN-BT ceramics that were fabricated by powders with different particle sizes after calcination and then the secondary milling process. The grain size slightly decreased and then increased as the secondary milling time increased after calcination. When the specimen with an average powder particle size of 14.31 μm, was sintered under the same sintering conditions, non-uniform grains were developed as we expected above. On the contrary, for the specimens sintered with an average particle size of 1.27 μm, uniform grain sizes were developed. It is interesting to notice that the grain sizes were slightly increased for the specimens when the secondary milling time exceeded 48hr after calcination. This can be interpreted as follows: the submicrometer particle powders easily made a large amount of the liquid phase and a nuclei during the sintering process for grain growth at the early stage of sintering, as shown in Fig. 4(c) and (d) [11].
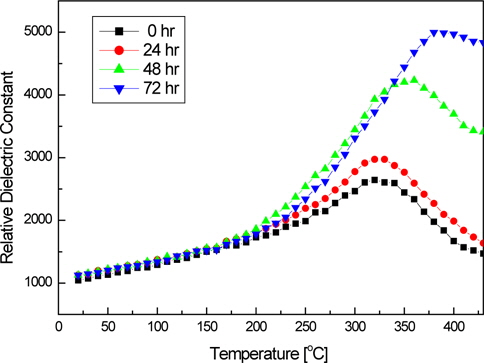
Figure 5 shows the temperature dependence of the dielectric constant of the KNN-BT ceramics that were fabricated by powders with different particle sizes after calcination and the secondary milling process, which were measured at 1 kHz. The maximum dielectric constant increases as the particle size decreased. The specimen sintered with a submicrometer particle size showed a larger dielectric constant than those that had a micrometer particle size. This might be due to the increase of grain size despite the low sintered densities.
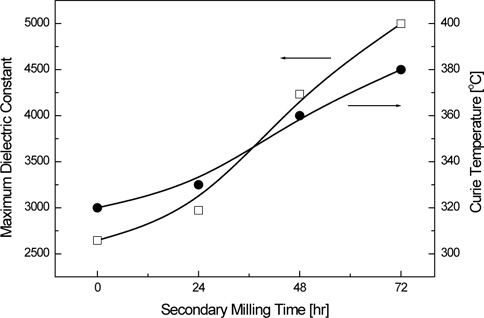
Figure 6 shows the variations of the maximum dielectric constant and Curie temperature of the KNN-BT ceramics that were fabricated by powders of different particle sizes after calcination and the secondary milling process. As seen in Fig. 6, Curie temperatures (
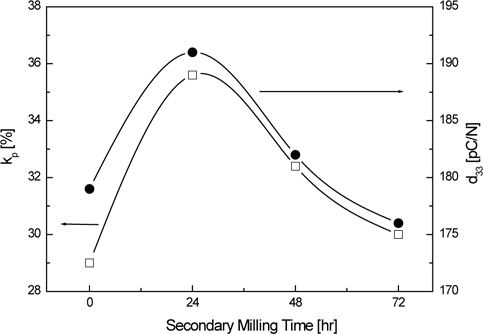
Figure 7 shows the variations of electromechanical coupling factor (
We investigated the effects of the different particle sizes of the powder on the dielectric and piezoelectric properties of KNN-BT lead-free ceramics.
The particle sizes of the powder with increasing secondary milling time after calcination for the KNN-BT ceramics were decreased from 14.31 μm to 0.91 μm. As the particle size of the powder decreased, the sintered density of KNN-BT ceramics increased. It, subsequently, decreased due to the formation of the liquid phase. The Curie temperature (
The electromechanical coupling factor (