The genetic information in living organisms is regulated, replicated, and repaired by DNA-binding proteins that can recognize and bind specific DNA sequences.1 In eukaryotes, DNA-binding proteins such as histones stabilize DNA by forming nucleosomes, which are essential in preventing unnecessary DNA damage and controlling gene expression. Thus, various DNA-binding proteins have been studied in an attempt to understand the biochemical control of gene expression.2 More specifically, DNA-binding motif consisting of several peptides in a protein facilitates binding to double-stranded DNA, owing to interactions with DNA bases, without unwinding the double helix structure of the DNA. Therefore, the characterization of peptides from DNA binding motifs such as zinc fingers,2 leucine zippers,3 etc. is an active area of research. Moreover, DNA-binding peptides have enormous potential in a variety of biotechnological and biomedical applications for biosensors and peptide-based drugs, and others. Accordingly, the discovery of novel DNA-binding peptide motifs is of paramount importance for a variety of biochemical applications.
In the discovery of novel DNA-binding motifs, mass spectrometry (MS) has emerged as a powerful tool to investigate non-covalently bound complexes and presents advantages4 over traditional techniques such as DNA footprinting5 and electrophoretic mobility shift assays.6 Importantly, MS measurements are fairly simple to conduct and MS provides the molecular weight of intact noncovalent complexes, stoichiometry, and information on subunit interactions. Moreover, MS measurements require only a small amount of sample (femtomolar level). Two soft ionization methods, namely electrospray ionization (ESI) and matrix-assisted laser desorption ionization (MALDI) have been utilized to provide MS peaks of intact, non-covalently bound complexes. ESI-MS is often preferred for non-covalent DNA-peptide complexes, which are formed within a buffer solution.7 As an example, we previously reported the identification of non-covalent interactions between DNA and zinc finger motifs utilizing ESI-MS.8 Although MALDI uses co-crystallized analytes within a matrix,9 its use in the analysis of DNA-peptide complexes is attractive, because some of the difficulties encountered with ESI-MS can be avoided. First, MALDI spectra are much easier to interpret owing to the predominant singly charged species. Second, sample preparation for MALDI is much simpler than that for electrospray. Importantly, MALDI-TOF has been implemented to analyze numerous non-covalent complexes since Juhasz and Biemann utilized MALDI-TOF to analyze non-covalent complexes between DNA and positively charged peptides for the first time.10 Subsequently, a number of studies regarding DNA-peptide interactions were conducted using MALDI-TOF11.
As a novel DNA binding peptide, we recently discovered that a neutravidin-coated surface could bind large DNA molecules without biotin12. Although it is well known that neutravidin is used to link biotin-labeled molecules, it was an unexpected result that neutravidin could bind DNA without biotin. From the crystal structure of avidin protein, we hypothesized that binding was facilitated by the protruding tryptophan (
Chemicals. All oligo-(deoxyribo)-nucleotides were purchased from Bioneer (Daejeon, Korea) and all peptides were purchased from GL Biochem (Shanghai, China). 2, 5-Dihydroxybenzoic acid was purchased from Tokyo Chemical Industry Co., Ltd. (Tokyo, Japan). Other chemicals were obtained from Sigma-Aldrich Korea (Yong-In, Korea).
Mass spectrometric analysis. For MALDI-TOF analysis, oligonucleotides were used as DNA substrates for peptide binding reaction. For these reactions, peptides were dissolved in water to a concentration of 100 µM. Peptide sequences used for the experiments are shown in Table 1. DNA sequences used for the experiments are shown in Table 2. Oligo-(deoxyribo)-nucleotides were dissolved in a TE buffer solution (10 mM Tris–HCl; 1 mM EDTA; pH 8.0) at room temperature to a concentration of 100 µM. DNA-peptide binding reactions were performed in 20 µL total volume: 10 µL of 100 µM oligo-(deoxyribo)-nucleotides and 10 µL of 100 µM peptides were mixed. The reaction was incubated at room temperature for 30 minutes. Samples were mixed with matrix solution onto a Bruker MTP 384 polished steel MALDI sample support target plate. The matrix solution was 130 mM 2, 5-Dihydroxybenzoic acid in methyl alcohol. MALDI-TOF analysis was performed using a Bruker AutoflexTM Speed (Bruker Daltonics Inc., Billerica, MA, USA) in the linear positive ion mode. Mass spectra were obtained as sums of 500 laser shots at 1000 Hz (at least 10,000 shots from different spots).
[Table 1] Peptides used in mass spectrometry

Peptides used in mass spectrometry
[Table 2] Oligonucleotides used in mass spectrometry

Oligonucleotides used in mass spectrometry
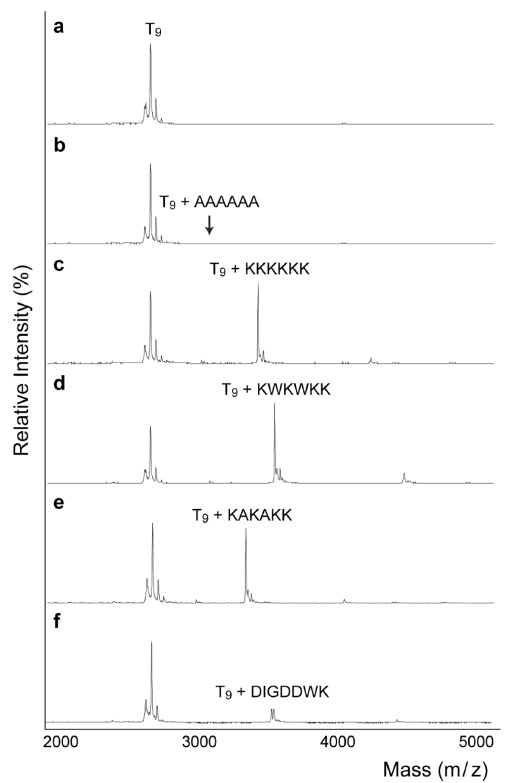
The mass spectra in Fig. 1 demonstrate the relative abundances of non-covalent complexes formed between DNA and positively charged peptides. Even though there are structural differences between single stranded DNA and double stranded DNA in the non-covalent binding to peptides, it is problematic to obtain mass spectra of double stranded DNA in MALDI-TOF, because of strand displacement; therefore, we conducted entire experiments using single stranded DNA instead of duplexes. Furthermore, in the series of studies, amino acid sequences are deeply related with their binding capability with single stranded oligonucleotides,19-21 likewise with double stranded DNA. As a control, the mass spectrum of an alanine oligomer showed no complex formation (Fig. 1b). In contrast, Fig. 1c clearly shows the formation of a DNApeptide complex. The positive charges on lysine facilitate the interaction between the DNA and peptide. In 1969, Shapiro
The ratios of the peak areas for the DNA-peptide complex over the unbound oligomer were 1.1 for KKKKKK (Fig. 1c) and 1.4 for KWKWKK (Fig. 1d). Previously, Mascoti and Lohman used a series of peptides (KWK
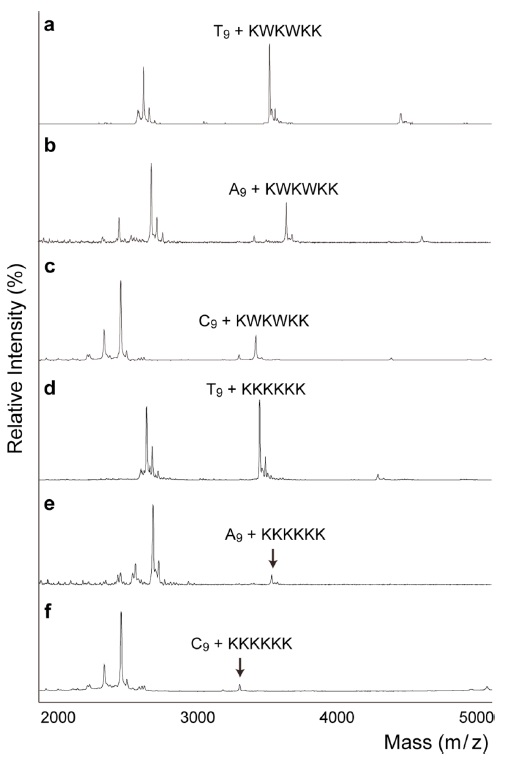
The next question was whether the peptide has selective preference for DNA sequences. Figure 2a~2c shows the order of binding affinities of T9 >A9 >C9, which was consistent with previous studies. Previously, using circular dichroism and fluorescence, Maurizot
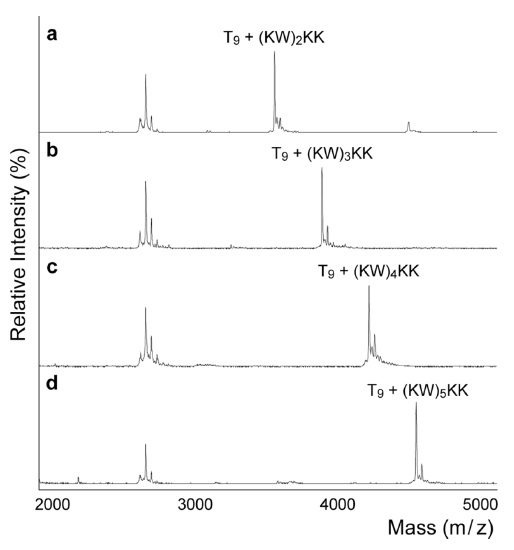
The purpose of this study was to discover novel strong DNA-binding peptides using MALDI-TOF. As mentioned earlier, increasing the number of positive charges enhanced the peptide-DNA binding affinity. Furthermore, the addition of tryptophan also increased this affinity by the base intercalation with indole rings. Therefore, we prepared a number of lysine and tryptophan (KW) repeats. Figure 3 demonstrates that the addition of KW repeats increased DNA binding capability. This trend is consistent with a previous study reported by Mascotti and Lohman.27,28 In particular, (KW)5 KK formed a complex with DNA; the ratio of the peak intensity was 2.1 for the complex over free DNA, which was almost double that of (KW)2 KK (
In this paper, we demonstrated an approach to measure DNA binding affinity against the peptide consisting of lysine and tryptophan residues using MALDI-TOF. Although there are traditional ways to measure DNApeptide binding such as footprinting, electrophoretic mobility shift assays, and tryptophan fluorescence quenching, MALDI-TOF is simple and fast compared with other methods. More importantly, since there are numerous combinations to make peptide oligomers, a simple and fast analytical approach, such as MALDI-TOF, is promising for quick identification of non-covalent DNA and peptides binding.