Most of LNT catalysts use noble metals such as Pt for low temperature NOx oxidation but there is an economic weakness. For the purpose of overcoming this, this study is to develop DeNOx catalyst for LNT excluding PGM (platinum group metal) such as Pt, Pd, Rh, etc. To do so, Al/Co/Ni catalyst selected as a preliminary test is used to study fundamental property and NOx’s conversion according to calcined temperature. Ultimately, that is, Al/Co/Ni mixed metal oxide which does not use PGM is selected and physicochemical characterization is performed by way of XRD, EDS, SEM, BET and ramp test and NOx conversion is also analyzed. This study shows that all samples consist of mixed oxides of spinel structure of Co2AlO4 and NiAl2O4 and have enough pore volume and size for redox. But as a result of NH3-TPD test, it is desired that calcined temperature needs to be maintained at 700 ℃ or lower. Also only samples which are processed under 500 ℃ satisfied NO and NOx conversion simultaneously through ramp test. Based on this study’s results, optimum calcined temperature for Al/Co/Ni=1.0/2.5/0.3 mixed metal oxide catalyst is 500 ℃.
대부분의 LNT 촉매는 낮은 온도 영역에서의 NOx 산화를 위하여 Pt와 같은 귀금속류를 사용하는 것은 경제적인 부담을 가지고 있다. 따라서, 본 연구는 이러한 문제를 해결하기 위하여 시도되었다. 즉, Pt, Pd, Rh 등과 같은 귀금속류(platinum group metal, PGM)를 사용하지 않는 LNT (lean NOx trap)용 DeNOx 촉매를 개발하기 위해 시도하였다. 이를 위해서 예비실험을 통해 Pt등 귀금속류등의 PGM (platinum group metal)을 사용하지 않는 Al/Co/Ni 혼합 금속 산화물을 선정하였다. 궁극적으로는, 선정된 촉매의 소성온도에 따른 물리화학적 특성 변화가 NOx 전환율에 미치는 영향을 살펴보고자 하였다. 이들의 물리화학적인 성질을 평가하기 위해 XRD, EDS, SEM, BET 분석을 실시하였다. 이러한 평가를 실시한 결과, 모든 소성온도에서 혼합금속 산화물은 Co2AlO4 및 NiAl2O4의 스피넬 구조가 혼재되어 있는 것으로 나타났고, NOx 기체들의 산화-환원 반응이 이루어지기에는 충분한 기공부피와 기공크기를 갖고 있음을 알 수 있었다. 그러나 NH3-TPD 분석 결과에서는 소성온도가 700 ℃ 이하를 유지해야 하는 것으로 판단되었다. 더욱이 ramp test를 통해서는 NO 및 NOx 전환율을 동시에 만족할 수 있는 시료는 소성온도는 500 ℃에서 처리된 경우임을 알 수 있었다. 이러한 결과 등을 바탕으로, Al/Co/Ni=1.0/2.5/0.3 혼합 금속 산화물의 최적 소성온도는 500 ℃임을 알 수 있었다.
Under the environmental standards tightening fuel efficiency regulation following greenhouse gas emissions around the world, demand for a diesel engine is increasing more and more because it has many advantages such as a power, an economy, and the emission of carbon dioxide into the atmosphere compared with a gasoline engine. But to handle the emissions of particulate matters and nitrogen oxides, the responsive techniques are necessary to control combustion characteristics of a diesel engine[1].
To decrease nitrogen oxides of exhaust gas from diesel or lean burn gasoline engines, selective catalytic reduction (SCR)[2,3] and a lean NOx trap (LNT)[4-6], termed as nitrogen storage reduction (NSR) is recommended.
The SCR method causes a reaction between NO in exhaust gas and ammonia with a metal-substituted zeolite catalyst[7] or the vanadia-tungsta-titania catalyst[8], but the LNT catalyst does not use any reductant species. LNT catalyst systems are use a fuel-lean long cycle with NOx gases in the exhaust stored onto the catalyst and a short-cycle fuel-rich area with NOx captured by H2, CO and hydrocarbons in fuel gas reduced to N2 alternately[4-6]. A combination of the SCR and the LNT method has been considered as well due to its conversion efficiency[2,9-15], but it is difficult to operate due to the complicated equipment required.
In general, LNT catalyst involve alkali/alkaline earth metals with supporters having a high specific surface area, such as γ-Al2O3, and a catalyst using CeO2 with noble metals such as Pt, Rh and Pd[16,17]. General DeNOx catalysts use the V2O5/TiO2 catalyst, which is active at temperature exceeding 200 ℃ and zeolite-type catalysts but an expensive noble metal, Pt, is necessary for conversion in low temperature region. In order to cope with the regulations on air pollution, improving engine systems and the use of catalysts with noble metals for conversion under low temperature as active species are very important. However, this requirement is considered to be economically disadvantageous[18]. For this reason, PGM-free metal oxides for oxide diesel gas exhaust excluding the use of platinum group metals (PGM) such as Pt and Pd have been introduced[11-14]. In particular, according to recent research, there is an increasing possibility that perovskite oxide catalysts can replace PGM catalysts to remove or decrease NOx[19,20].
This study was conducted to develop a DeNOx catalyst for LNT without PGMs. For an automotive catalyst, the performance can differ based on the carrier structure, oxide component, manufacturing method and the method used to contain active species such as noble metals. Therefore, this study analyzes the physicochemical and NOx conversion characteristics according to the calcination temperature of Al/Co/Ni mixed metal oxide selected through a preliminary test[21]. We also provide basic data regarding the development of catalyst with enhanced performance.
The Al/Co/Ni (henceforth ACN) mixed metal oxide is produced by the following procedure. A mixed solution is produced to dissolve aluminum nitrate nonahydrate at 1 mole and cobalt (II) nitrate hexahydrate and nickel nitrate hexahydrate, respectively at 1.8 mole and 0.2 mole. We slowly added this mixed solution into a buffer solution with NaOH and Na2CO3 at room temperature and identified the creation of the precipitate. At this point, after ascertaining that the mixed solution keeps the pH 11, we obtained a catalyst precursor via a cleansing process until the solution reached a pH of 7 while filtering the precipitate for 16 hours at 65 ℃. After drying and grinding the mixed solution in a dryer at 110 ℃ for more than 12 hours and placing, it into a furnace and increasing the temperature, we produced a mixed metal oxide ACN catalyst with calcination at 500 ℃, 600 ℃, 700 ℃, 800 ℃ and 900 ℃.
2.2. Analysis of the Properties of the Catalyst
2.2.1. X-ray Diffraction Pattern
The X-ray Diffraction Pattern (XRD) to identify the crystal structure was determined at 40 kV and 40 mA using a Rigaku Ultima IV Diffractometer with a Cu tube and Graphite with an attached monochromater. Data reduction was done with the PDXL program.
2.2.2. Analysis of the Components and Morphology
In order to analyze the calcined mixed metal oxides, we measured the energy resolution under the following condition : “peak shifts of <127 eV, 5-300 kcps and <5 eV, with a Si drift detector (SDD), (Detection : Be (z=4) ~Am (z=95))”-using a Bruker, Quantax 200 energy dispersive X-ray spectrometer. In addition, with a Tescan Mira 3 LMU FEG, scanning electron microscope (SEM), we carried out an analysis of the surface structure and size at an acceleration voltage of 20 kV, with a Quorum Q150TES Coater operated at 10 mA, and 120 s for the Pt coating step.
2.2.3. Pore Characteristics
The pore characteristics and specific surface area of the samples were analyzed by means of the adsorption isotherm of nitrogen gas at the temperature of liquid nitrogen (77 K) using automatic adsorption equipment (Micrometrics, ASAP 2010).
2.2.4. NH3-TPD Test
Using an Autochem II2920 TPD (Temperature Programming Desorption) with an attached thermal conductivity detector, the ammonia desorption patterns depending on the temperature of the samples were characterized. After outgassing the calcined samples up to 800 ℃ at a steady speed of He at 25 mL/min and cooling 70 ℃, the samples were adsorbed with flowing ammonia gas for 3 hours, followed by purging with He gas until the base line was stabilized. Ammonia desorption was analyzed with desorbed gas via TCD by increasing the temperature to 800 ℃ at 5 ℃/min while injecting He at 25 mL/min.
2.3. Evaluation of the NOx conversion
This study, we attempted to forecast the actual performance of the mixed metal oxide catalyst for DeNOx. To do this, we determined the NOx efficiency while accelerating the catalytic reduction reaction of NOx in engine exhaust gas under an excess oxygen condition, using only CO as a reductant in the exhaust gas without the addition of any reductant species. In more detail, in order to ensure similarity of the characteristics of the exhaust gas and to keep the concentration of oxygen at 10% of oxygen, 300 ppm of NO, with 900 ppm of CO and 700 ppm of HC, with the flow of each gas done using a MFC (Mass Flow Controller), we conducted a ramp test while increasing the temperature of the reactor to 470 ℃ at 20 ℃/min. The concentration of the gases coming from the catalytic reactor was assessed using by MK9000 analysis equipment. Figure 1 shows a schematic diagram of ramp test.
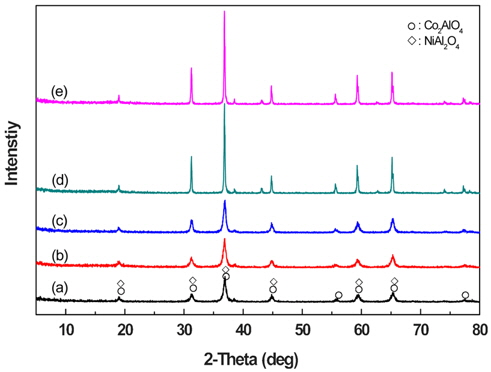
Figure 2 shows the X-ray diffraction patterns of a catalyst sample according to calcination temperature. As shown in Figure 2, the crystallinity increased abruptly when the calcination temperature exceeded 800 ℃. In this case, the increase in the temperature does not create a new type of crystal as a result of reactions such as fusion, when each crystal is increased separately. Moreover, the crystal structures of the samples calcined from 500 ℃ to 900 ℃ show a mixed oxide of Co2AlO4 and NiAl2O4, which has a spinal structure. This result differs from those in earlier studies which demonstrated the creation of PGM-free pervoskite oxides as a DeNOx catalyst for LNT[19,20]. According to these studies, the perovskite oxide catalyst can be a PGM-free catalyst and can be used to remove or decrease NOx. Therefore, the spinel structure of Al/Co/Ni mixed oxide as well as perovskite oxide can serve as a PGM-free DeNOx catalyst. In order to clarify this, we tested this with another analysis method.
As shown in the scanning microscope images in Figure 3, at calcination temperatures of 500 ℃, 600 ℃ and 700 ℃, the crystal structure remains at tens-of-nanometers in size without a change, but the crystal structures at 200 nm or more start to increase when the calcination temperature exceeds 800 ℃. However, in Figure 2, instead of a creating new crystal structure with each calcination temperature, it is known that the Co2AlO4 and NiAl2O4 structures are created separately.
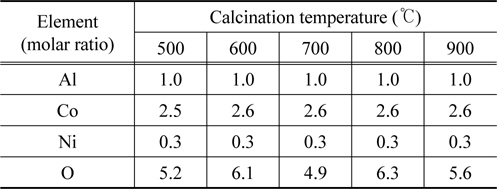
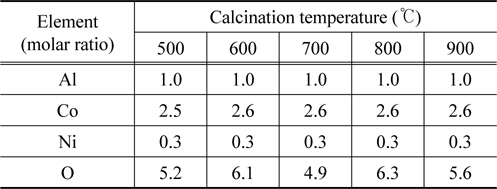
Table 1 explains this reason. This table shows the energy dispersive x-ray spectrometry results for some locations randomly selected on each sample, with each structure maintaining a steady mole fraction of Al/Co/Ni=1/2.5/0.3 at each temperature instead of showing a different structure.
[Table 1.] The EDS analysis results of the mixed oxide catalyst samples
The EDS analysis results of the mixed oxide catalyst samples
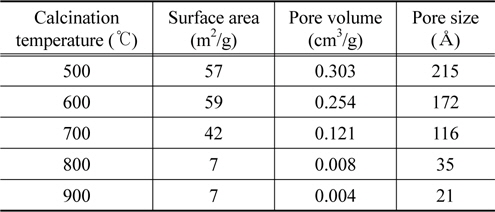
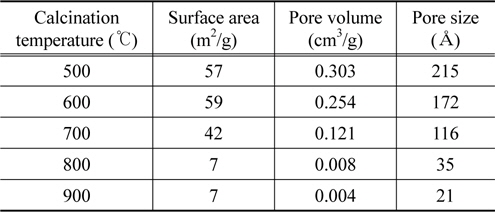
Table 2 summarizes the pore characteristics of the sample's according to the different calcination temperatures. The pore characteristics were determined with the BET (Brunauer-Emmett-Teller) equation. As shown in Table 2, the more the calcination temperature increases, the more the specific surface area, pore volume, and pore size decrease. Specifically, as identified in the SEM image at the top of Figure 3, this decreased abruptly at 800 ℃ or more due to the increase in the crystal size when the temperature exceeded 800 ℃. However, because the bonding length between nitrogen atoms and oxygen atoms is 1.188 Å and the bond angle is 126°, even the sample calcined at 900 ℃ with the smallest pore size can have a sufficient pore volume and size to trigger an oxidation-reduction reaction due to the reactive gases in the pores. However, it can be expected that catalyst samples with a greater pore volume and pore size among samples with similar surface areas have an advantage with regard to catalysis, needing less time. It is expected that the removal efficiency of nitrogen oxide is the most favorable in the samples calcined at 500 ℃.
[Table 2.] The BET properties of the mixed oxide catalyst samples
The BET properties of the mixed oxide catalyst samples
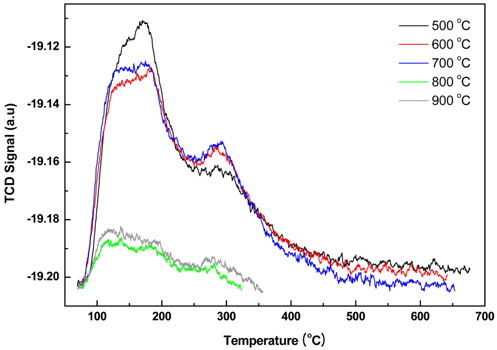
Figure 4 shows the results of the NH3-TPD test conducted to predict the possibility of using the DeNOx catalyst for nitrogen oxides. Every five samples showed a trend in which ammonia desorption occurred smoothly at a temperature of 300 ℃ or lower. This is an encouraging result, considering the LNT condition in which oxidation and reduction occur at the same time in the low-temperature region, as shown by the weak acid point and the strong acid point peaks of approximately 185 ℃ and 290 ℃, respectively. Samples calcined at temperatures of 500 ℃ to 700 ℃ are similar in terms of their ammonia desorption amounts, but samples calcined at 800 ℃ to 900 ℃ are substantially lacking in the amount of ammonia desorption. Based on this result, a suitable calcination temperature to use with the DeNOx catalyst having a variety of acid sites and a number of ammonia desorption amount is less than 700 ℃.
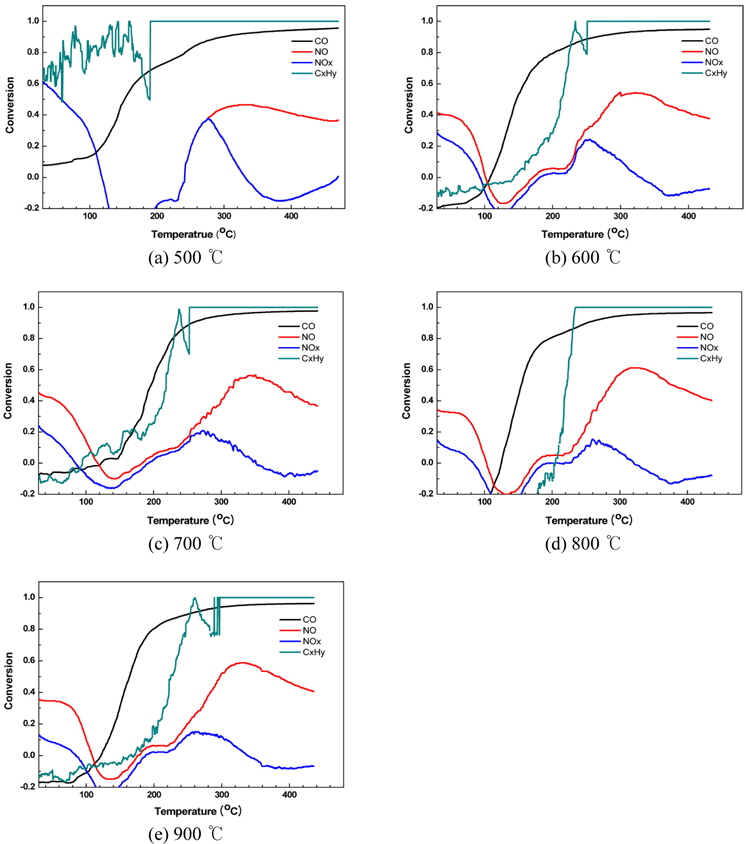
Figure 5 shows the results of the ramp test, which determined the removal or conversion efficiencies of lean gases with excess oxygen to forecast the performance levels of catalytic samples with different calcination temperatures as a DeNOx catalyst for LNT. This test was conducted because, among the assessed characteristics of mixed metal oxide catalysts for DeNOx, the NOx removal efficiency is a critical factor when using only CO as a reductant without any additional reductant under the condition of an engine exhaust gas operated in an excess oxygen condition. Therefore, in this case, the ramp test was conducted with gases concentration of oxygen 10%, 300 ppm of NO, 900 ppm of CO, and 700 ppm of HC. This condition is similar to that of an actual engine.
As shown in Figure 5, the more the samples are treated at a high calcination temperature, the more the NO conversion increases. The reaction for LNT is required simultaneously is that NO, which exists in exhaust gas, is oxidized to NO2 and the newly created NO2 and NO2 which originally existed is reduced to N2. That is, the system requires a continuous oxidation-reduction reaction to progress. However, as shown in Figure 5, samples which are calcined at a high temperature tend to show a low conversion of NOx. As seen in Figure 4, the catalyst is treated up to 800 ℃ did not have a various acid site and a sufficient amount of ammonia adsorption. Lower conversion is considered to be caused from this phenomenon.
As explained earlier, we conclude that the catalyst does not need to be calcined at 800 ℃ or more because it is not suitable for a mixed metal oxide for DeNOx if the conversion of NOx is not high even when the change rate of NO is high. According to the results of this study, considering the NO and NOx rates of change, the calcination temperature that satisfies all of the requirements is 500 ℃. As shown in the SEM images and BET analysis results, considering that samples with the largest specific surface areas, pore volumes and pore sizes are those which were calcined 500 ℃, it can be concluded that these characteristics have an effect on the DeNOx reaction.
In this study we attempted to develop a DeNOx catalyst for LNT without PGMs (platinum group metals) such as Pt in order to overcome this problem. To do this, we reviewed the basic properties and NOx conversion of the Al/Co/Ni catalysts according to the calcination temperature. That is, the Al/Co/Ni mixed metal oxides were carried out XRD, EDS, SEM, BET, NH3-TPD analyses and a ramp test, and assessed the removal or conversion of NOx while also analyzing the physiochemical characteristics.
As a result of these assessment, irrespective of the calcination temperature, all samples show a mixed oxide structure of spineltype Co2AlO4 and NiAl2O4 with a pore volume and pore size sufficient to trigger an oxidation-reduction reaction. Moreover, it was identified that the mole fraction of Al/Co/Ni is 1.0/2.5/0.3. However, it was also found that the calcination temperature should remain at 700 ℃ or less because the amount of ammonia desorption is substantially decreased when samples are calcined at temperature of 800 ℃ or more.
This study also found that samples calcined at 700 ℃ or less have weak acid and strong acid characteristics, allowing oxidation and reduction reactions to occur at the same time. As a result of the ramp test, samples calcined at 500 ℃ showed acceptable NO and NOx conversion concurrently. According to all of the analyse, it was identified that the optimum calcination temperature of a mixed metal oxide consisting of Al/Co/Ni=1.0/2.5/0/3 as a DeNOx catalyst for LNT is 500 ℃.
According to the results of this study, it can be expected that a DeNOx catalyst for LNT developed in the future if research concentrates on the conversion change trends based on changes of the components and additions of active species using a catalyst selected based on this study as a basic component.
![Schematic diagram of the ramp test[21].](http://oak.go.kr/repository/journal/16886/CJGSB2_2015_v21n3_184_f001.jpg)