Silkworm powder has been investigated and used as a functional food source after Ryu’s group identified the antidiabetic effect of silkworm powfder (Chung
Alkaline protease of various types of enzymes produced by microorganisms of the genus Bacillus is changed while the high molecular weight protein with a low molecular weight protein and increasing the amount of soluble extract (Parekh
Freeze-dried silkworm powder was purchased from a Korean sericulture farm in Boeun, Korea. FoodPro Alkaline Protease was obtained from Bision Corporation (SeongNam, Korea) and stored at 4°C until use. Phosphate buffered saline (PBS, pH 7.4) was purchased from Life Technologies (California, USA) for use as an enzyme reaction buffer. Azocasein was purchased from Sigma-Aldrich (St. Louis, USA).
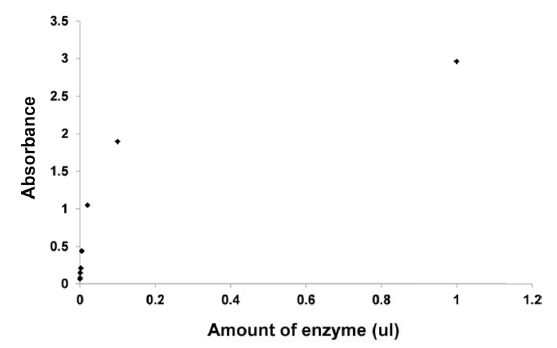
Azocasein was used as a substrate in a proteolytic activity assay (Plantner, 1991) for alkaline protease. For this 450 μL of a 1% solution of azocasein in PBS was used, and 1 μL of the following dilutions of alkaline protease was added to the azocasein substrate: ×1, ×10, ×50, ×200, ×500, ×1,000 and ×5,000. The reaction mixture was incubated at 60°C for 15 min. Subsequently 250 μL of 25% cold trichloroacetic acid (TCA) was added to each tube and mixed well. The reaction solution was clarified by centrifugation at 10,000 rpm for 5 min. From this 600 μL of supernatant for each dilution was used for further analysis, 600 μL of 1 N NaOH was added to each tube containing supernatant. The absorbance of the alkalinized solution was read at 440 nm (A440) in an automated microplate reader.
>
Enzymatic treatment of silkworm powder
For the enzymatic treatment, 0.1 g of silkworm powder was used as a substrate: 100 mL of PBS and 100 μL of alkaline protease were added to the flask containing the powder. Silkworm powder in 100 mL PBS solution was used as the control. The reaction mixture was incubated at 60°C for 3 h. After this period, the mixture was incubated at 90°C for 15 min to inactivate the enzyme. The reaction solution was centrifuged at 13,000 rpm for 10 min. The supernatant collected was analyzed using a UV spectrometer. All experimental groups were repeated three times.
>
SDS-PAGE analysis of silkworm powder protein
Samples for electrophoresis were prepared as follows; silkworm powder(0.1 g) was incubated with 100 μL of enzyme. The enzymatic supernatant and pellet were separated by centrifugation. Then 20 μL of supernatant was mixed with 4× SDS-sample buffer, and the mixture was boiled for 5 min at 100°C. Next, the mixture was loaded onto 4-12% gradient gels for analysis of electrophoretic patterns. The protein gradient gels were stained with silver staining. All experimental groups were repeated three times.
To determine the enzymatic activity of commercial enzyme, the azocasein assay was performed. Fig. 1 shows the UV spectra of the azocasein solution. As shown in the figure, azocasein releases the azo group, and a strong absorption band is observed at 440 nm (Kilcawley
>
Effect of alkaline protease on silkworm powder
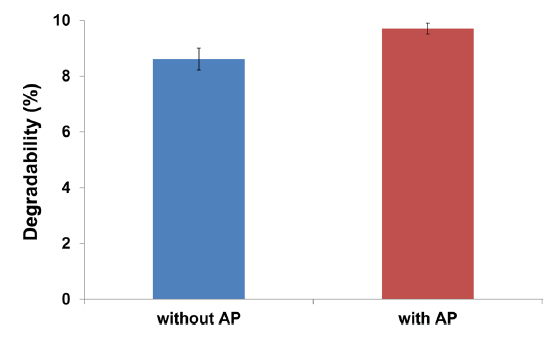
To confirm the effect of the enzyme on silkworm powder, silkworm powder in PBS solution was treated with the enzyme. The soluble protein content (%) of silkworm powder after enzyme treatment was 9.7%, which was significantly higher than that for the control (Fig. 3); this finding indicates that the protein in silkworm powder was hydrolyzed by treatment with the enzyme.
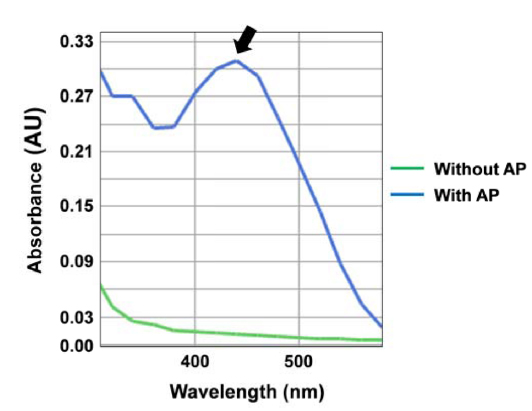
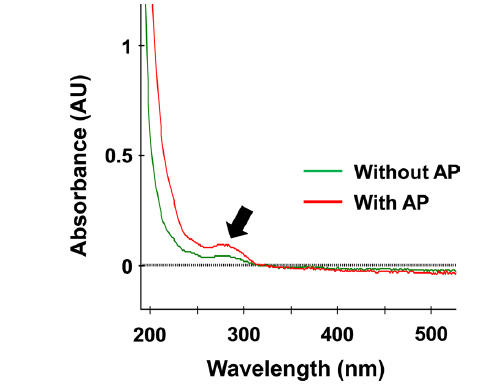
In general, proteins show a strong absorption band at 280 nm. The UV-VIS spectra of silkworm powder supernatant are shown in Fig. 4. All samples showed a characteristic protein absorption band. However, on treatment with enzyme, the absorption band at 280 nm became stronger indicating that the soluble and degradable protein content of silkworm powder treated with the enzyme was higher than that of the control.
>
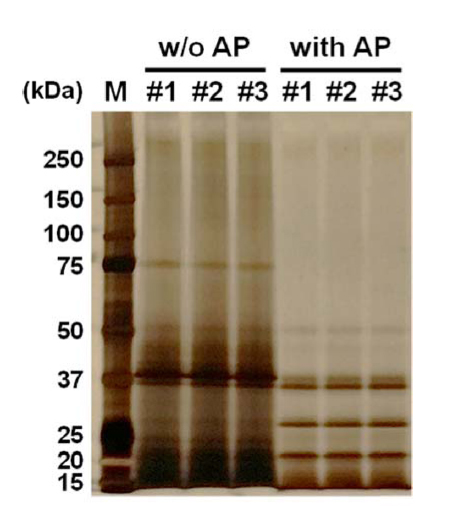
SDS-PAGE analysis of silkworm powder protein
SDS-PAGE electrophoresis is a commonly used method for determining the molecular weight distribution of natural proteins. Fig. 5 shows the SDS-PAGE electrophoretic pattern of silkworm powder supernatant after enzyme treatment. The molecular weight pattern of silkworm powder extracts changed after this treatment. The silkworm powder supernatant (control) showed strong bands around 75, 50, 40, 37, and 20 kDa. After enzyme treatment, some protein bands around 75 and 40 kDa weakened and disappeared and new bands were observed around 25 kDa. Therefore, we plan to further study the diary efficiency and functionality of silkworm powder treated with enzyme.