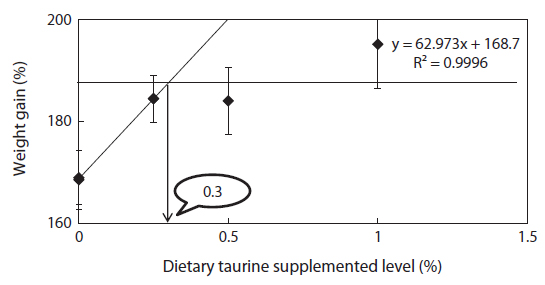
An 8 week feeding trial was conducted to evaluate whether methionine and cysteine would effectively spare taurine supplementation on growth performance in juvenile rock bream Oplegnathus fasciatus. Triplicate groups of 25 fish averaging 2.74 ± 0.04 g (mean ± SD) were fed one of the experimental diets. Five experimental diets including a fish meal based control diet were prepared by adding sulfur amino acid at fixed level of 1.0% and taurine at graded levels of 0%, 0.25%, 0.5% and 1.0% (S + Tau0, S + Tau0.25, S + Tau0.5 and S + Tau1.0, respectively). After the feeding trial, growth performance of fish fed the S + Tau0.25, S + Tau0.5 and S + Tau1.0 diets were significantly higher (P < 0.05) than those of fish fed the Control diet. ANOVA test suggested that when sulfur amino acid were supplemented to the diets, the optimum taurine supplementation level could be 0.25% in the diet, and broken line analysis of weight gain indicated a level of 0.33%, for positive effects on growth and feed utilization. Fish whole-body protein content and taurine concentration steadily increased with the increase of dietary level in the presence of sulfur amino acid in the diets. On the other hand, whole-body lipid content significantly decreased with the incremental levels of dietary taurine. In conclusion, the results of the present study clearly indicated that dietary supplementation of methionine and cysteine at a level of 1% could spare 0.25 to 0.33% of taurine in juvenile O. fasciatus diets.
Taurine (2-aminoethanesulfonic acid) is one of the dietary supplements that have been used to promote growth in aquaculture. Historically, taurine has not been considered as an essential amino acid for fish, however, recently taurine is being considered conditionally essential amino acidto promote growth performance in several species (Park et al., 2002; Kim et al., 2003, 2005, 2008b, 2008c; Takagi et al., 2006, 2011; Gaylord et al., 2007; Lunger et al., 2007; Qi et al., 2012, Lim et al., 2013). The major pathway for taurine synthesis from methionine in mammals is believed to involve the transformation of methionine to cystathionine by cystathionine synthetase, the transformation of cystathionine to cysteine by cystathionase, the oxidation of cysteine to cysteine sulfinate, the decarboxylation of cysteine sulfinate to hypotaurine and further oxidation of hypotaurine to taurine (Worden and Stipanuk, 1985). Rhodes and Davis (2011) reported that taurine in marine finfish is essential in a range of 2.6 to 72.0 g kg-1, besides, taurine requirement is directly related to cysteine sulfinic acid decarboxylase activity (Yokoyama, 2001).
Fish have a total sulfur amino acid requirement rather than a specific methionine requirement (Wilson and Halver, 1985). The presence of dispensable amino acid cysteine in the diet represents a sparing effect in that it reduces the need for methionine (Ahmed et al., 2003). An adequate amount of dietary cysteine spares methionine from cysteine synthesis. Thus, the requirement for total sulfur amino acid can be met by a proper mixture of methionine and cysteine. Consequently, sulfur amino acids are taurine precursors, and methionine and cysteine supplementation may spare dietary taurine requirement, supported by Jirsa et al. (2014) and Wang et al. (2014).
Rock bream
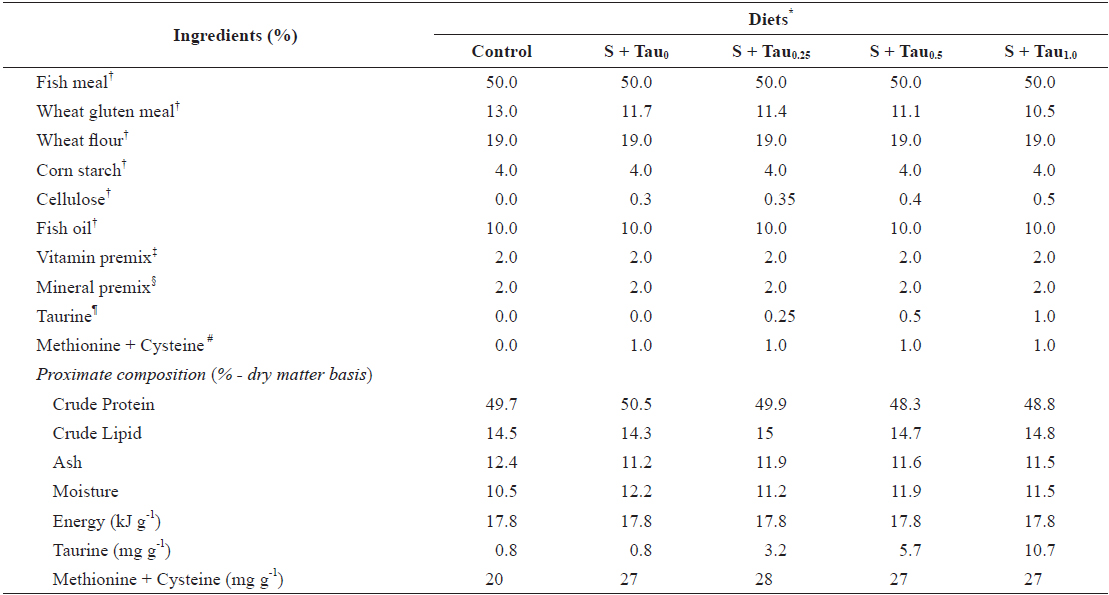
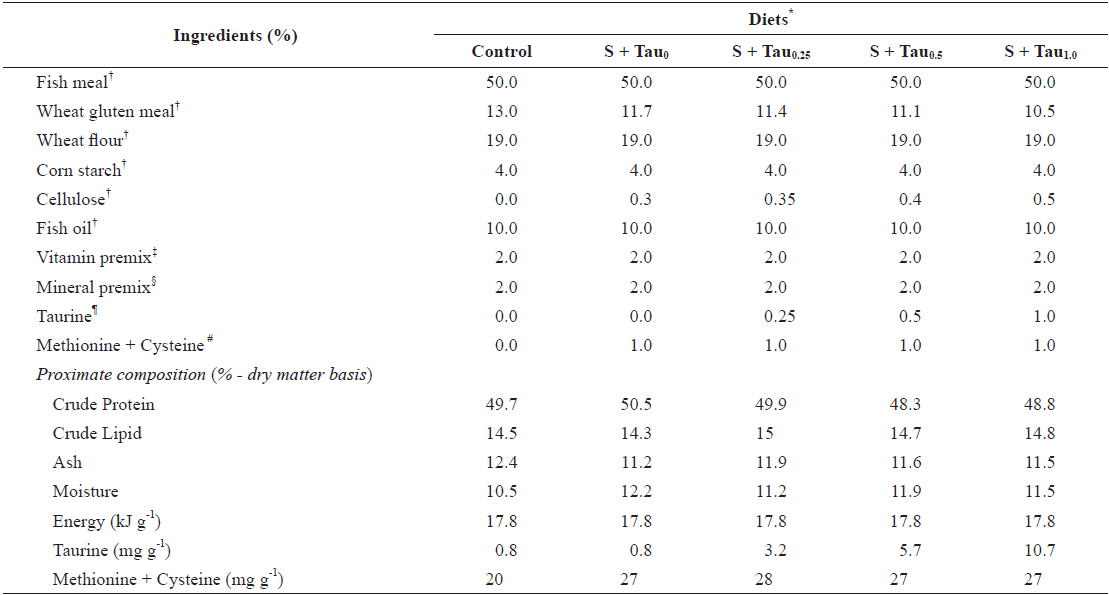
Five experimental diets were formulated using fish meal as the main protein source (Control diet) and also prepared by adding fixed methionine and cysteine at 0.5% each and graded taurine at 0%, 0.25%, 0.5% and 1.0% (S + Tau0, S + Tau0.25, S + Tau0.5 and S + Tau1.0, respectively), as presented in Table 1. The experimental diets were formulated to be isonitrogenous (49% crude protein) and isocaloric (17 kJ gross energy g-1). Energy levels of the diets were calculated based on 23.6, 17.2 and 39.5 kJ g-1 for protein, carbohydrate and lipid, respectively (NRC, 2011). Dietary cysteine and methionine concentrations were similar in levels among the experimental diets. Dietary taurine concentration increased proportionally with taurine supplementation levels, and the concentration (mg taurine g diet-1) was 0.8 in Control and S + Tau0, 3.2 in S + Tau0.25, 5.7 in S + Tau0.5 and 10.7 in S + Tau1.0. Experimental diets were prepared by thoroughly mixing the dry ingredients in an electric mixer, followed by the addition of oil and distilled water. This mixture was formed into dough, and pellets were made by passing the dough through a screw type pelleting machine and air drying the formed pellets for 48 hours. After drying, the pellets were ground, sieved into the proper pellet size, packed and stored at -20°C until use.
[Table 1.] Formulation and proximate composition of experimental diets (% dry matter)
Formulation and proximate composition of experimental diets (% dry matter)
>
Experimental fish and feeding trial
Juvenile rock bream
>
Sample collection and analysis
At the end of the feeding trial, fish were starved for 24 h, counted and weighed to calculate weight gain (WG), feed efficiency (FE), specific growth rate (SGR), protein efficiency ratio (PER) and survival. After the final weighing, three fish per tank were randomly selected and used to determine hepatosomatic index (HSI) and condition factor (CF). Then, 10 fish per tank were randomly selected, minced and stored at -20°C until used for whole-body proximate composition and amino acids (taurine, methionine and cysteine) analysis. Proximate composition analysis of the experimental diets and fish wholebody were analyzed according to standard methods (AOAC, 2006). Samples of diets and fish were dried at 110°C for 24 h using a dry oven to determine their moisture content. Ash content was determined by incineration at 600°C in a muffle furnace. Crude protein content was determined using the Kjeldahl method (N × 6.25) after acid digestion, and crude lipid content was determined by soxhlet extraction using the Soxtec system 1046 (Foss, Hoganas, Sweden) after freeze-drying for 12 h. Samples were prepared for sulfur amino acids and taurine consisted of homogenized whole-body, 1% sulphosalicylic and centrifuged at 3,000 g for 20 min at 4°C. Amino acids concentrations were determined individually with using a Sykam S-433 automatic amino acid analyzer (Sykam, Eresing, Germany).
All data were analyzed by one-way ANOVA. When a significant effect of the treatments was observed, a least significant difference (LSD) test was used to compare means. Treatment effects were considered significant at
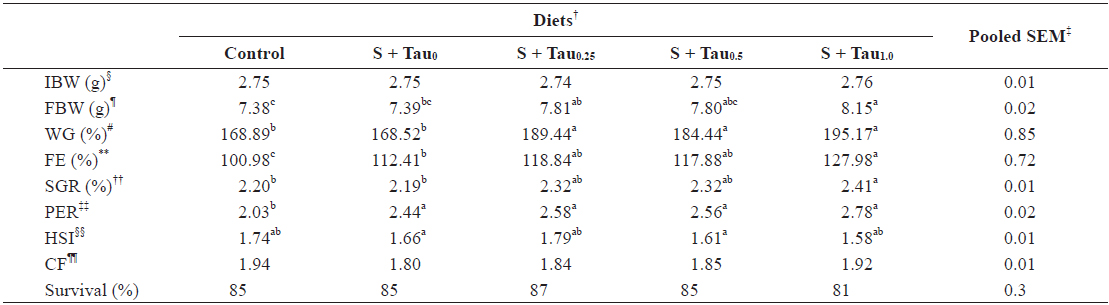
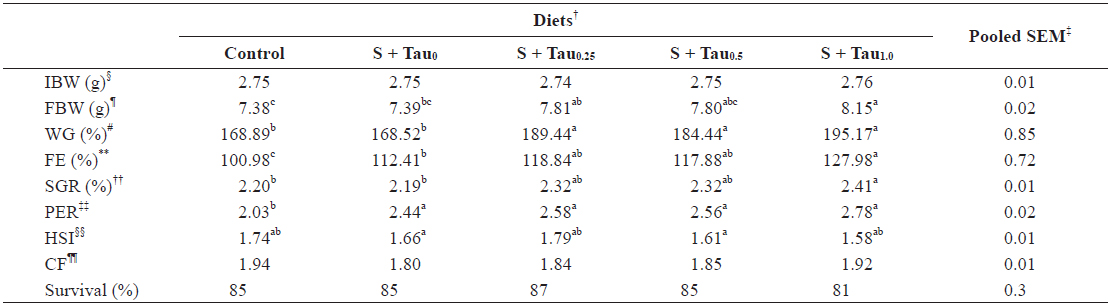
Data on weight gain (WG), feed efficiency (FE), specific growth rate (SGR), protein efficiency ratio (PER), hepatosomatic index (HSI), condition factor (CF), and survival rate of juvenile
Growth performance of juvenile rock bream Oplegnathus fasciatus fed the experimental diets for 8 weeks*
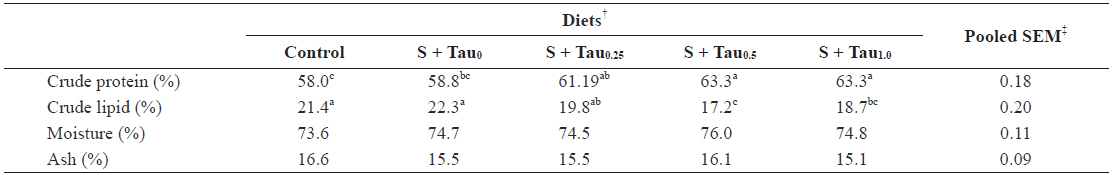
Data on proximate composition of juvenile rock bream fed the experimental diets are summarized in Table 3. Fish whole-body crude protein content steadily increased with the increase of dietary taurine level in the presence of sulfur amino acids in the diets. Whole-body protein content of fish fed S + Tau0.5 and S + Tau1.0 were significantly higher than those of fish fed Control and S + Tau0 diets. No significant differences were noted in fish fed S + Tau0.25, S + Tau0.5 and S + Tau1.0 diets, fish fed S + Tau0 and S + Tau0.25 diets, or among that of fish fed Control and S + Tau0 diets. Whole-body crude lipid content significantly decreased with the incremental levels of dietary taurine up to S + Tau0.5. Fish fed S + Tau0.5 and S + Tau1.0 showed significantly lower lipid content than that of fish fed Control, S + Tau0 and S + Tau0.25; however, there was no significant difference between that of fish fed S + Tau0.25 and S + Tau1.0, or between that of fish fed S + Tau0.5 and S + Tau1.0. No significant differences were evident for moisture and ash content of fish fed the experimental diets.
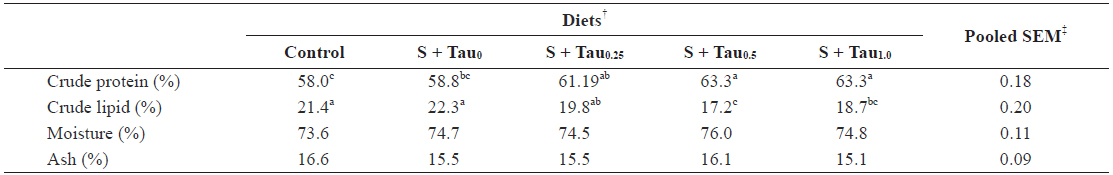
Whole-body proximate composition (% dry matter) of juvenile rock bream Oplegnathus fasciatus fed the experimental diets for 8 weeks*
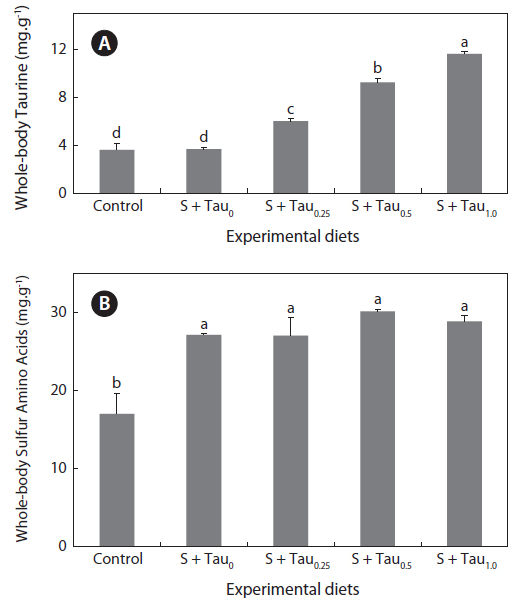
Taurine and sulfur amino acids (methionine and cysteine) concentrations in whole-body of fish fed the experimental diets are presented in Figs. 2a and 2b, respectively. Whole-body taurine content of fish fed the experimental diets showed an increase trend as taurine was supplemented in the diets. Fish fed the S + Tau1.0 diet had a significantly higher whole-body taurine content than those of fish fed the other experimental diets. Also, fish fed the S + Tau0.25 diet showed significantly higher taurine content than fish fed the Control and S + Tau0 diets; however, there were no significant differences between fish fed diets which were not supplemented by taurine (Control and S + Tau0). Whole-body sulfur amino acids content of fish fed the experimental diets supplied with methionine and cysteine (S + Tau0, S + Tau0.25, S + Tau0.5 and S + Tau1.0) was significantly higher than that of fish fed the Control diet.
Methionine, cysteine and taurine supplementations promoted growth performance of rock bream, and indicated an increase trend with dietary taurine supplementation. Based on ANOVA test there seemed to be no benefits of increasing taurine supplementation in diets beyond 0.25%, in the presence of methionine and cysteine supplementation, as fish fed diets with superior supplemented levels of taurine did not significantly improved growth performance. However, according to broken line regression analysis of WG, optimum dietary taurine supplementation was estimated to be 0.33%, which indicates that the optimum inclusion level could be 4.2 mg taurine g diet-1 with sparing. The optimum dietary taurine supplementation level suggested in this experiment is lower than those found previously for rock bream, 7.0 mg (Ferreira et al., 2014) and 8.8 mg (Lim et al., 2013) taurine g-1 diet without sparing. The growth performance results of this experiment indicated that excessive taurine supplementation (S + Tau0.5 and S + Tau1.0) did not improve growth compared to the optimal taurine inclusion (S + Tau0.25), when sulfur amino acid (SAA) were supplemented. Similarly to the present feeding trial results, Qi et al. (2012) also found that with increasing dietary taurine, growth performance increased and then reached a plateau, with no beneficial effects when taurine was supplied above the required levels. However, fish fed methionine and cysteine diet (S + Tau0) did not show higher growth performance compared to Control diet. Conversely, Wang et al. (2014) reported that not only taurine supplementation but also methionine and cysteine significantly promoted growth performance in juvenile turbot
A significantly positive linear relationship between the content of taurine accumulated in whole-body and the level of taurine in the diets was noted in the present study. This result is similar to our previous experiment with juvenile rock bream (Ferreira et al., 2014) as well as for those reported for juvenile flounder (Park et al., 2002; Kim et al., 2003, 2005a, 2005b, 2008b), juvenile red sea bream (Matsunari et al., 2008) and juvenile turbot (Qi et al., 2012). Moreover, Wang et al. (2014) also reported that juvenile turbot fed diets supplemented with methionine and cysteine did not significantly change taurine concentration compared to the Control diet. Whole-body SAA content were found to be higher in fish fed methionine and cysteine supplemented diets compared to that of fish fed diet without supplementation. However, no significant difference was observed in whole-body SAA content among fish fed SAA acids supplemented diets. Once, the major pathway for taurine synthesis in mammals is believed to involve the transformation of methionine to cystathionine, the transformation of cystathionine to cysteine, the oxidation of cysteine to cysteine sulfinate, the decarboxylation of cysteine sulfinate to hypotaurine and further oxidation of hypotaurine to taurine (Worden and Stipanuk, 1985), our result is more likely resulted from the accumulation of taurine and sulfur amino acids through the feed intake. The results of the present study associated to the above taurine biosynthesis pathway and the previous results with rock bream (Lim et al., 2013; Ferreira et al., 2014) may suggest that there was a possible taurine insufficiency in the Control and low taurine supplemented (S + Tau0) diets, as well as that the sulfur amino acids accumulated in the whole-body of rock bream may indicate that juvenile rock bream has a limited taurine biosynthesis ability through sulfur amino acids pathway. Park et al. (2002) reported similar results with olive flounder, and Kendler (1989) observed a decrease of whole body cystathionine in rainbow trout fed taurine supplemented diets, suggesting that species with taurine biosynthesis ability tend to show a reduced content of cystathionine in the whole-body with the increase of dietary taurine.
In the present study, while whole-body crude protein was found to be increased with increasing dietary taurine. In the presence of sulfur amino acids, crude lipid content significantly decreased with increasing of dietary taurine. These results are similar to our previous publication (Ferreira et al., 2014); however, the supplementation of methionine and cysteine in the diets enhanced the protein content in whole-body. In lipid metabolism, the roles of taurine include conjugation with bile acids in the liver which increase the use of the bile acids, the degradation of cholesterol metabolites and the participation in micelle formation contributing to fat absorption in the small intestine (Yokogoshi et al., 1999).
Although rock bream seems to have limited ability to synthesize taurine through sulfur amino acids pathway, its taurine requirement ranged between 3.3 to 4.2 mg taurine g-1 diet through supplementation of sulfur amino acids (methionine + cysteine) at a level of 27 mg g-1 diet. In our previous study, we reported that 0.62% taurine supplementation are required to improve growth in juvenile rock bream through broken line analysis of WG (Ferreira et al., 2014). Therefore, the findings of the present study indicated that growth could be improved in juvenile rock bream by supplementing taurine at a level approximately 50% lower than our previous experiment result (Ferreira et al., 2014). In other words, dietary supplementation of methionine and cysteine at a level of 1% could spare 0.25 to 0.33% of taurine in juvenile
This conclusion is based on broken line regression analysis of WG and ANOVA. Further studies are recommended to investigate the adequate supplementation levels of methionine and cysteine and the dietary sulfur amino acids modulations of taurine biosynthesis in juvenile rock bream.