To examine the interrelationship between transgenic insertion patterns and transgene expression profiles in established transgenic fish lines, four stable transgenic marine medaka Oryzias dancena germlines harboring β-actin regulator-driven RFP reporter constructs were selected. The established transgenic strains were characterized with regard to their transgenic genotypes (insertion pattern, concatemer formation, and transgene copy number based on genomic Southern blot hybridization and qPCR assay) and expression characteristics at the mRNA (qRT-PCR), protein (western blot), and phenotypic (fluorescent appearance) levels. From comparative examinations, it was found that transgenic expression at both the transcription and translation levels could be significantly downregulated in transgenic strains, potentially through methylation-mediated transgene silencing that was particularly associated with the formation of a long tail-to-head tandem concatemer in the chromosomal integration site(s). When this occurred, an inverse relationship between the transgene copy number and fluorescence intensity was observed in the resultant transgenic fish. However, with the other transgenic genotype, transgenic individuals with an identical Southern blot hybridization pattern, containing a tandem concatemer(s), had very different expression levels (highly robust vs. low expression strengths), which was possibly related to the differential epigenetic modifications and/or degrees of methylation. The concatemer-dependent downregulation of transgene activity could be induced in transgenic fish, but the overall pattern was strain-specific. Our data suggest that neither a low (or single) transgene copy number nor tandem transgene concatemerization is indicative of strong or silenced transgene expression in transgenic fish carrying a ubiquitous transgene. Hence, a sufficient number of transgenic lineages, with different genotypes, should be considered to ensure the establishment of the best-performance transgenic line(s) for practical applications.
Experimental transgenesis in fish is useful in genomics research to utilize the recombinant animal model for acquiring the unique genotype and phenotype (Hackett and Alvarez, 2000; Dunham and Winn, 2014). In addition, the transgenic manipulation of aquaculture fish species using gene(s) related to quantitative traits has received attention due to its potential as a powerful tool in the molecular genetic breeding of farmed fishes for productivity enhancement (Nam et al., 2001, 2007). The microinjection of DNA into fertilized eggs has long been utilized to produce transgenic fish strains, although novel methodologies to induce homologous and/or targeted transgenesis have been challenged recently in some fish models (Rembold et al., 2006; Grabher and Wittbrodt, 2007; Casco-Robles et al., 2010). In microinjection-mediated gene delivery, it is widely accepted that the exogenously introduced DNA integrates into vertebrate genomes in an approximately random fashion. Multiple copies of the injected construct usually form a concatemer(s) in tandem arrays, in which the copy sequences are often incomplete at the ends, and DNA nibbling is sometimes observed between repeat units (Bishop, 1996; Baup et al., 2010). The repeat number of transgene copies is reported to be unregulated and varies widely between different microinjection experiments within a given host species injected with identical constructs (Würtele et al., 2003).
The context of transgene integration in the host genome provides the basis for the expression and functionality of transgenic individuals. Transgene expression has been found to be correlated with the integrant copy number in some cases, but studies in mammals have suggested it is related to heterochromatin-associated gene silencing (Koetsier et al., 1996; Kong et al., 2009). Mammalian transgenics have shown that high-copy number transgenes arranged in a large tandem concatemer(s) are prone to structural modification, with a repressive influence on transgene expression, and the resulting transgenic individuals have often displayed inconsistent expression (Dorer and Henikoff, 1997; Geurts et al., 2006). However, this inverse relationship between repetitive copy numbers and transgene expression has also been disputed (Pena et al., 2004).
In contrast to the relatively rich information available in mammals, the interrelationship between transgene insertion patterns and expression profiles has been less well studied in established transgenic fish germlines, with only a few previous studies reported (Wu et al., 2005; Uh et al., 2006). Unlike mammalian transgenics, high-copy number-related depressive regulation has not always been observed in transgenic fish strains. High-copy number transgenic fishes, carrying a long concatemer, have been reported to drive robust transgene expression, sometimes with markedly higher levels than those of single-copy or low-copy number transgenic fishes, particularly when measured absolutely (Nam et al., 2001; Cho et al., 2013a). For this reason, the copy number- and concatemer-dependent expression patterns that are common in mammalian transgenics might not apply to fish transgenics, and hence empirical data on the consequences of non-homologous, illegitimate recombination of transgenes are required in transgenic fish. The paucity of such information may be preventing the development of an effective method to establish stable and functional transgenic germlines.
Herein, we report the transgenic strain-specific patterns of either faithful or repressive expression, with the involvement of transgene concatemerization, as exemplified by marine medaka
Various transgenic marine medaka founders were generated by the microinjection of a fluorescent reporter construct consisting of the RFP gene (DsRed2; Clontech Laboratories Inc., Mountain View, CA, USA) under the control of endogenous β-actin regulator (podβ-actRFP; Cho et al., 2011). Microinjection was performed on one-cell embryos using the
>
Characterization of the transgenic genotypes of selected strains
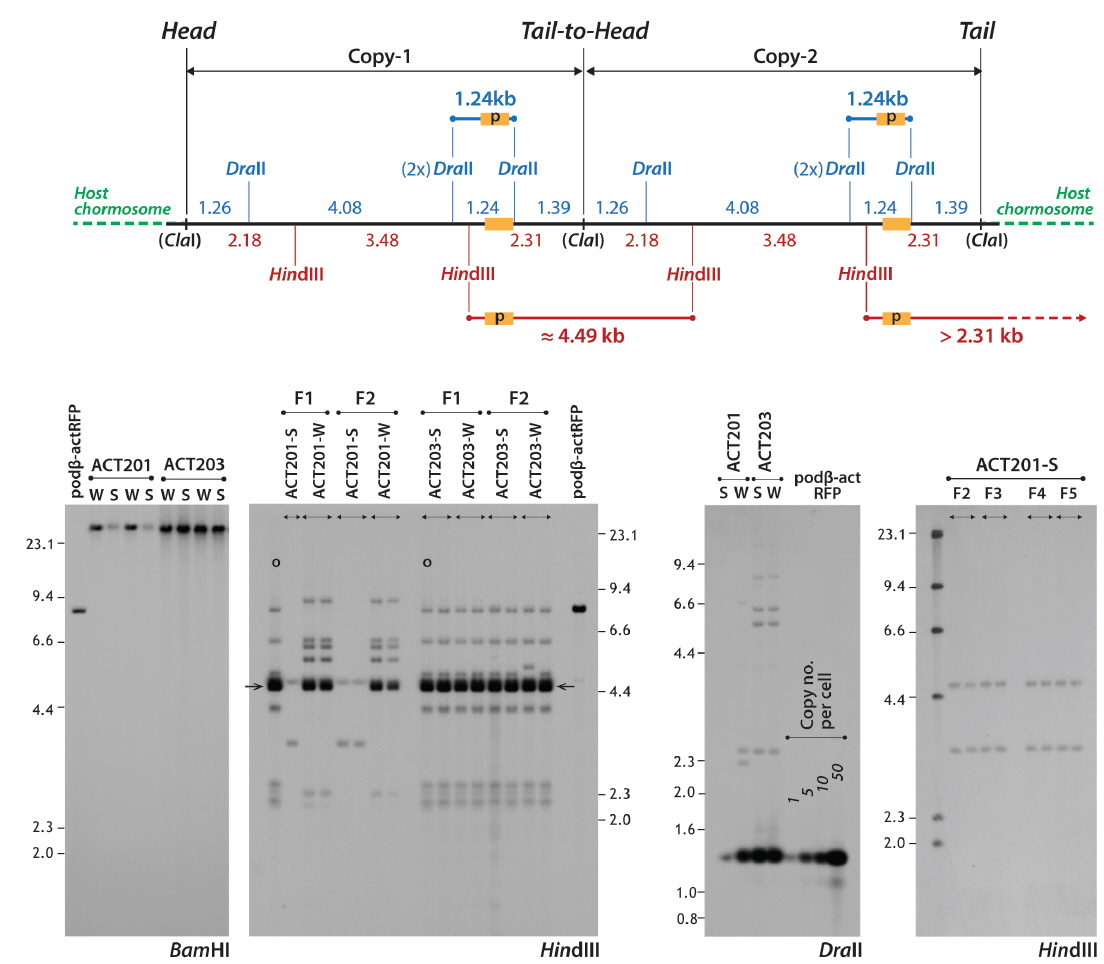
To examine the genomic integration and inheritance patterns of the transgene in the selected strains, a series of Southern blot hybridization analyses was performed. First, the absence of an extrachromosomally persistent transgene copy in the selected transgenic lines was confirmed by Southern blot hybridization using
To assess the transgene copy numbers, qPCR was performed using 10 ng genomic DNA as a template. The thermal cycling reaction included 2 × iQ™ SYBR® Green Supermix (Bio-Rad, Hercules, CA, USA) and a PCR primer pair (qDsRed-1F: CACAACACCGTGAAGCTGAA and qDsRed-1R: GAAGGACAGCTTCTTGTAG). The amplicon was a 141-bp internal
>
Analysis of transgenic expression
Transgene expression in the selected lines was characterized at the mRNA (qRT-PCR assay of
For Western blot analysis, the amount of soluble protein in the homogenate supernatant prepared with whole body fry was measured using a BCA protein assay kit (Pierce Biotechnology, Woburn, MA, USA). Resolved proteins from sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDSPAGE) were electrotransferred onto a nitrocellulose membrane (Amersham Pharmacia Biotech, Piscataway, NJ, USA) using a Trans-Blot® SD semi-dry transfer cell (Bio-Rad) and probed with Living Colors® DsRed monoclonal antibody (Clontech) diluted 1:2,000, and then with alkaline-phosphatase conjugated anti-mouse IgG secondary antibody (Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA) diluted 1:4,000. Positive signals were visualized using a colorimetric method with nitroblue tetrazolium salt (Sigma-Aldrich, St Louis, MO, USA) and 5-bromo-4-chloro-3-idolyl phosphate (Sigma-Aldrich) and then analyzed using Quantity-One image analysis software (Bio-Rad).
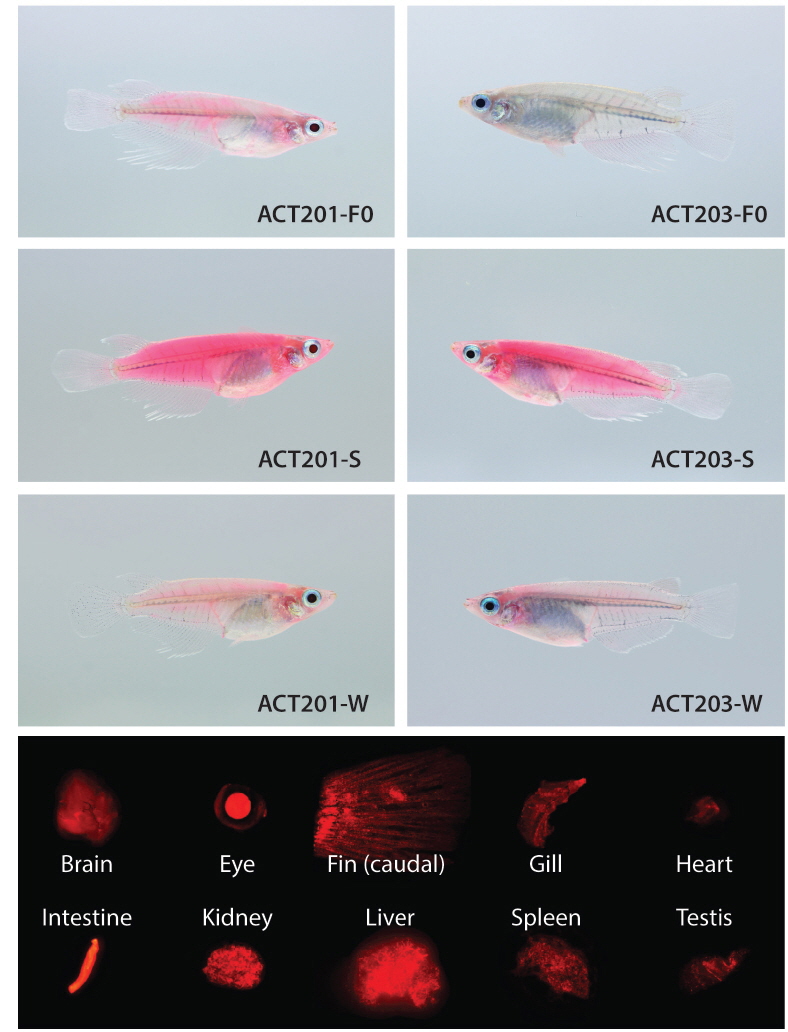
At the fluorescent phenotypic level, the RFP signals in transgenic models were analyzed using an AZ100 fluorescence microscope equipped with NIS-Elements BR image analysis software (ver. 3.1; Nikon Corporation Instruments Company, Tokyo, Japan) as described previously (Cho et al., 2011). The external appearance of RFP phenotypes in transgenic individuals were photographed using an EOD 5D Mark II digital camera (Canon, Tokyo, Japan) under either normal daylight or ultraviolet (UV) light (black light lamp; 352 nm; 15W; Sankyo Denki Co., Ltd., Tokyo, Japan). RFP signals in the internal organs (brain, eye lens, fin, gill, heart, intestine, kidney, liver, spleen, and gonads) were examined by fluorescence microscopy as described above.
Two germline positive founders (ACT201 and ACT203) passed on their fluorescent transgene to F1 progeny, with respective frequencies of 25 and 37% for RFP (+) F1 embryos, suggesting that the mosaic status in their germ cells was similar to those of most transgenic founders developed by microinjection (Iyengar et al., 1996; Kim et al., 2004). However, interestingly, the expression pattern achieved in F1 individuals from each of the two founders was not uniform. Strikingly different RFP levels were apparent among F1 individuals from a given founder, in which some F1 offspring displayed very strong RFP signals whereas others exhibited only faint RFP expression by external appearance. From the ACT201 founder, the proportion of F1 progeny with strong RFP expression (labeled ACT201-S) of the total RFP (+) F1 fish was approximately 20%, while the remaining 80% RFP (+) F1 displayed only weak expression (labeled ACT201-W). Conversely, the ACT203 founder resulted in similar incidences (1:1 ratio) of strong (ACT203-S) and weak (ACT203-W) expression. The transgenic F1 fish selected from ACT201-S, ACT201-W, ACT203-S, or ACT203-W lines were able to transmit their phenotypic characteristics to F2 fish, exhibiting Mendelian frequency. Differences in the external fluorescent appearance between strong and weak RFP types within a given lineage were clearly apparent (Fig. 1). The red fluorescent signals visualized over the whole body in transgenic individuals belonging to either the ACT201-S or ACT203-S lines were stunningly bright, and red fluorescence could be observed easily with the unaided eye under normal daylight conditions. Conversely, the ACT201-W and ACT203-W lines had only moderate or weak red fluorescence in their external appearance, although they could easily be distinguished from non-transgenic, wild-type individuals. Notably, several individuals belonging to the ACT203-W line lost the clear RFP signal in their eyes (data not shown). From microscopic examinations of the internal organs, transgenic individuals usually retained the ubiquitous distribution pattern over a wide array of tissues examined (Fig. 1), indicating that the transgenically introduced β-actin promoter could induce expression resembling the pattern of the endogenous β-actin gene (Cho et al., 2011; Lee et al., 2012). However, in several transgenic individuals belonging to the ACT203-W line, the presence of RFP (-) organs was determined under fluorescence microscopy conditions, although the organs lacking RFP signals (eye, heart, spleen, and/or testis depending on individuals; photograph not shown) were not uniform among these transgenic individuals.
Transgenic fish belonging to one of the four phenotypic groups (
Unlike the ACT201 lineages, only a single hybridization pattern was found in the
Another Southern blot assay using the
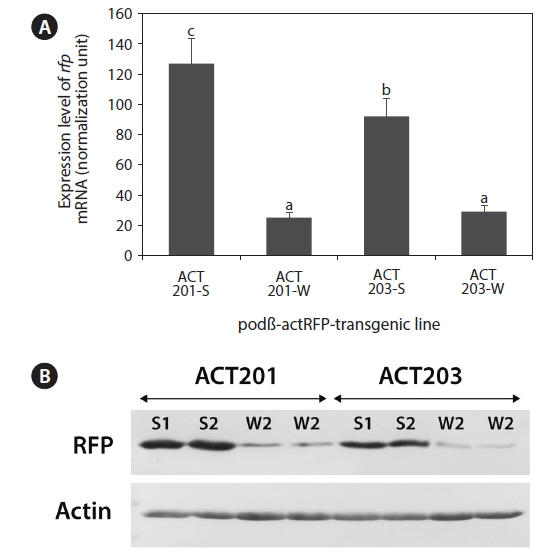
The transgenic lines ACT201-S/W and ATC203-S/W were subjected to expression analyses at the mRNA (qRT-PCR), protein (Western blot), and phenotype (fluorescent appearance) levels. qRT-PCR of
Taken together, the data presented here suggest strain-dependent patterns for the interrelationship between insertion structure and expression characteristics of the transgene expressed in marine medaka. As evidenced in the ACT201-S/W families, transgenic expression could be downregulated in transgenic fish strains by the formation of a long concatemer. Low-copy number transgenic fish without a long concatemer (ACT201-S) had a significantly higher level of transgene expression than that of their siblings with high transgene copy numbers in the