Abalones which belong to mollusc are important in aquaculture, and their cell lines can be used as
To date, considerable effort has been put into culturing abalone cells, including hemocytes (Lebel et al., 1996; Auzoux-Bordenave et al., 2007; Farcy et al., 2007; van der Merwe et al., 2010; Gaume et al., 2012; Latire et al., 2012) and tissue-dissociated cells derived from the gills (Suwattana et al., 2010; Gaume et al., 2012; Pichon et al., 2013), heart (Suwattana et al., 2010), larvae (Naganuma et al., 1994; van der Merwe et al., 2010), mantle (Poncet et al., 2000; Sud et al., 2001; Poncet et al., 2002; Suja and Dharmaraj, 2005; Auzoux-Bordenave et al., 2007; Suja et al., 2007), and radula (Kim et al., 2014a). However, despite these efforts, culture outcomes thus far have been generally unsatisfactory. The factors involved in cell survival and growth
For these reasons, we conducted primary cultures of cell populations derived from Pacific abalone
Pacific abalones were obtained from the Genetics & Breeding Research Center of the National Fisheries Research and Development Institute (Geoje, Korea). To obtain mature ovaries, abalones with swollen, dark green ovaries were selected and transported directly to the laboratory. The ovaries contained many late vitellogenic oocytes with distinct nuclei and nucleoli, and were classified as stage III of maturity (Najmudeen, 2007). Thirty abalones were sacrificed for this study, with an average shell length and total weight of 9.85 ± 0.36 cm and 99.07 ± 9.07 g, respectively.
Prior to dissecting the abalones for tissue collection, we collected hemolymph from the body cavities around the heart using 3-mL syringes as a medium supplement. Hemocytes were separated by centrifugation at 3,500
>
Tissue dissociation and cell collection
Abalones were disinfected in 70% ethanol (SK Chemicals, Sungnam, Korea) for 2 min, and the swollen, dark green ovaries surrounding the hepatopancreas were dissected with sterile scissors and tweezers. Two abalones were sacrificed for each replicate. Ovaries were placed in 35-mm petri dishes (SPL life Sciences, Pocheon, Korea) and rinsed five times with washing solution (WS) consisting of 0.2 μm filtered seawater supplemented with a 1% (v/v) mixed solution of penicillin and streptomycin (P/S; Gibco, Grand Island, NY, USA) and 2.5 μg/mL amphotericin B (Mediatech, Manassas, VA, USA). For cell dissociation, ovaries were finely minced with a surgical blade in an enzymatic solution consisting of Leibovitz’s L-15 medium (L15; Gibco), adjusted to 35 psu by dissolving Red Sea salt (Red Sea, Houston, TX, USA), supplemented with 0.05% trypsin–EDTA (Gibco) and 500 U/mL collagenase type I (Worthington Biochemical Corporation, Lakewood Township, NJ, USA), and incubated for 30 min at 23℃. After enzyme inactivation via the addition of L15 adjusted to 35 psu containing 10% (v/v) fetal bovine serum (FBS; Gibco) and 1% (v/v) P/S, all tissue derivatives were filtered on a 40-μm cell strainer (SPL Life Sciences) and centrifuged at 400 ×
>
Culture media and supplements
Basal culture medium was L15 supplemented with 1% (v/v) P/S, 1% (v/v) nonessential amino acids (Gibco), 100 μM β-mercaptoethanol (Gibco), and 2 nM sodium selenite (Sigma-Aldrich, St. Louis, MO, USA). To enhance cell proliferation, seven supplements consisting of 15% (v/v) FBS (Gibco), 15% (v/v) hemolymph, 10 ng/mL basic fibroblast growth factor (bFGF; Gibco), 25 ng/mL epidermal growth factor (EGF; Sigma-Aldrich), 1000 U/mL leukemia inhibitory factor (LIF; Millipore, Billerica, MA, USA), 50 μg/mL abalone embryo extract (AEE), and 50 μg/mL medaka embryo extract (MEE) were added to the basal culture medium. The final salinity of all culture media was adjusted to 35 psu. MEE was prepared according to the method reported by Lee et al. (2013). Briefly, collected blastula embryos of
Dissociated cells were seeded in 0.1% gelatin (Sigma- Aldrich)-coated 48-well culture plates (Becton Dickinson, Franklin Lakes, NJ, USA) filled with 800 μL of culture medium at 2 × 105 cells/well density; 200 μL of mineral oil (Sigma-Aldrich) was used to cover the medium to prevent its evaporation. Cells were cultured in an 18℃ incubator in an air atmosphere. After 3 days of culture, cells were washed twice with WS and filled with fresh culture medium. Thereafter, half the media were replaced every 4 or 5 days. When cells reached 100% confluency and cell sheets were formed, we conducted subculturing. For the subculture, cells were washed twice with 1% (v/v) P/S-containing calcium magnesium-free artificial seawater (CMFAS) consisting of 26.2 g/L NaCl, 0.67 g/L KCl, 4.62 g/L Na2SO4, 0.21 g/L NaHCO3, and 0.37 g/L Na2∙EDTA and trypsinized with CMFAS containing 0.05% trypsin–EDTA. After detachment of cells, L15 adjusted to 35 psu containing 10% (v/v) FBS and 1% (v/v) P/S were added for trypsin inactivation, and the cell suspension was centrifuged at 400
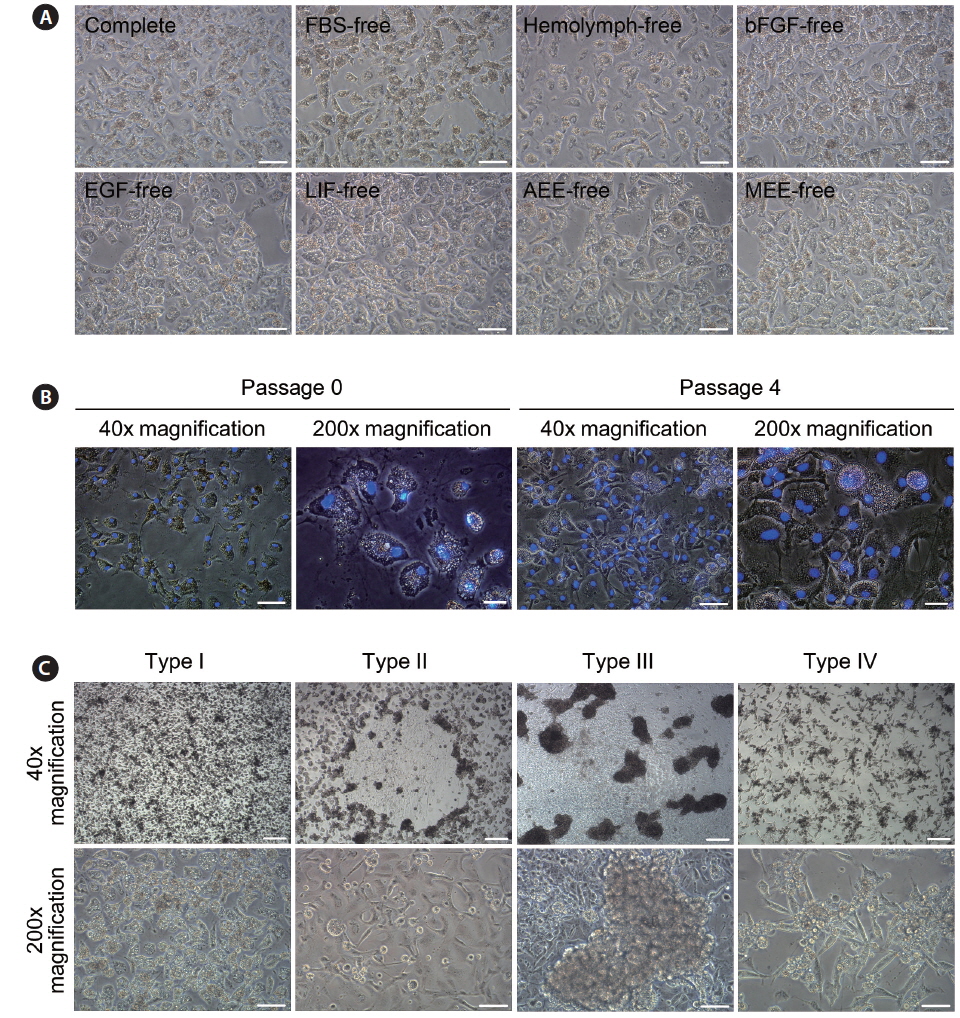
To investigate the state of the nuclei in cultured cells, we conducted nucleus staining. Live cells after initial attachment (passage 0) and the fourth subculture (passage 4) were washed twice with WS, and fresh media containing Hoechst 33342 (Molecular Probes, Eugene, OR, USA) at a final concentration of 5 μg/mL were added. After incubation for 30 min, cells were washed twice with WS and culture wells filled with fresh culture medium. Stained nuclei were visualized under an inverted microscope equipped with a fluorescent lamp (Nikon).
>
Experimental design and statistical analysis
To investigate the effects of seven medium supplements consisting of FBS, hemolymph, bFGF, EGF, LIF, AEE, and MEE, the composition of the culture medium was varied by removing one supplement from culture media containing all seven supplements. That is, we produced a total of eight culture media including one complete medium (containing all seven supplements) and one each without FBS, hemolymph, bFGF, EGF, LIF, AEE, or MEE, and cultured
>
Initial cell attachment of ovary-dissociated cell populations
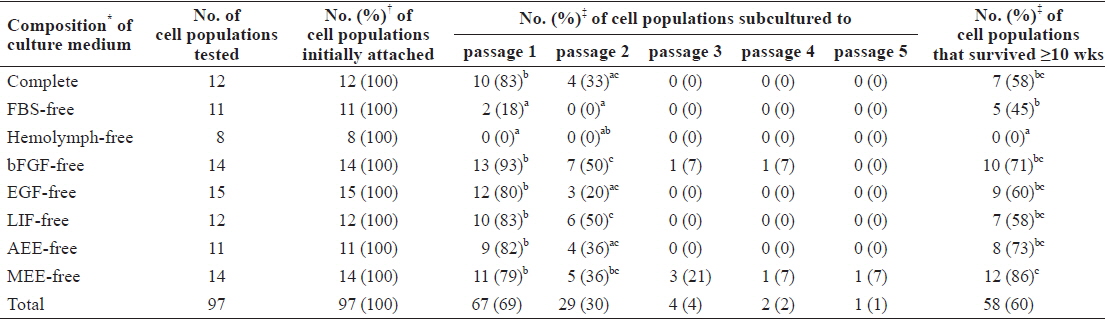
On day 3 of culture after initial cell seeding, we identified initial cell attachment. All ovary-dissociated cell populations cultured in these experiments showed successful initial cell attachment regardless of medium composition (total attachment in all 97 trials = 100%; Table 1). Moreover, the rate of initial cell attachment was an average of 70% in all cell populations and reached a maximum of 100% when scored visually. The morphologies of initially attached cells were very similar in all cell populations regardless of treatment, and all displayed thick, irregular forms harboring one nucleus with two or three nucleoli and many granules within their cytoplasm (Fig. 1A and 1B).
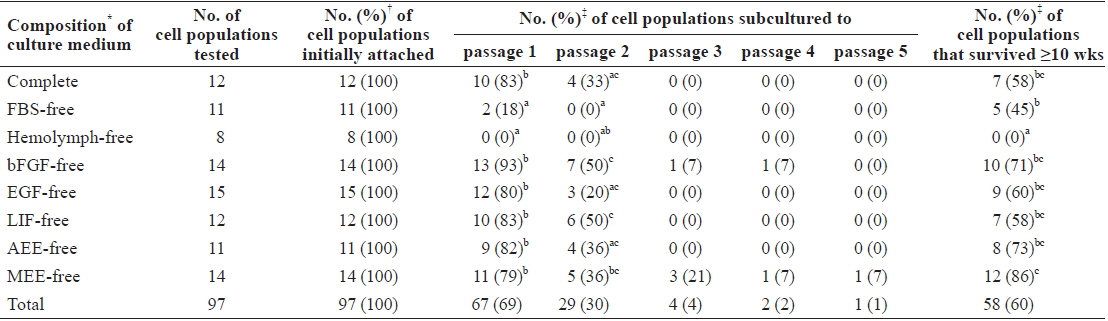
Effects of medium supplements on primary culture of Haliotis discus hannai ovary-dissociated cells
>
Effects of medium supplements on primary culture and in vitro maintenance
In contrast to the results for initial cell attachment, we detected significant differences in the growth of initially attached cells among treatments. As shown in Table 1, cell populations cultured in FBS-free or hemolymph-free media showed significantly lower rates in the first subculture. Only 18% (2 of 11) and 0% (0 of 8) of the initially attached cell populations cultured in FBS-free and hemolymph-free media, respectively, survived beyond the first subculture, while 79%–93% of cell populations cultured in the other six groups did so (0%–18% vs. 79%–93%,
>
Effects of individual differences on primary culture outcome
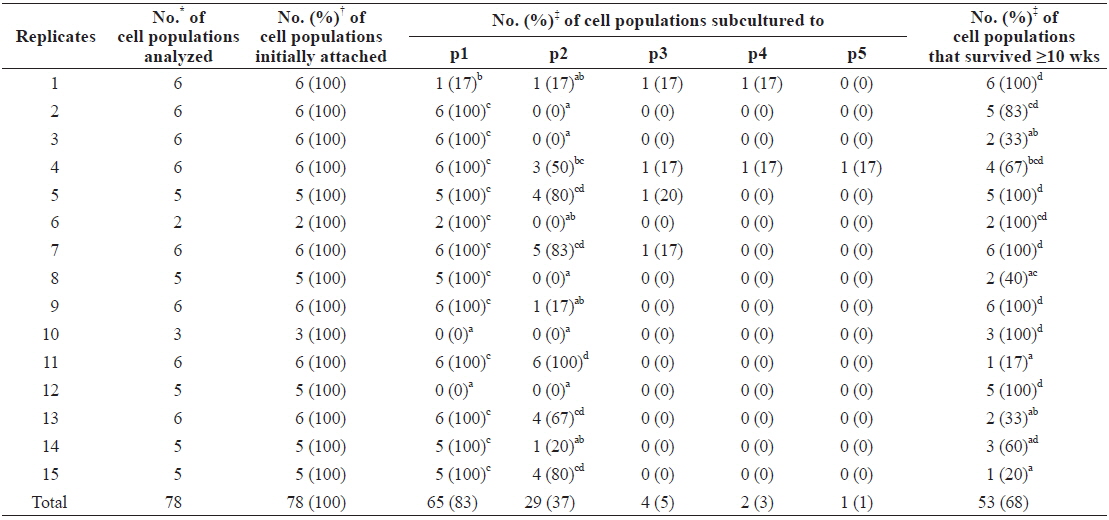
To examine the effects of individual differences among abalones on culture outcomes, we rearranged the data from Table 1 according to the replicates (Table 2). In this reallocation, data from cell populations cultured in FBS-free and hemolymph-free media were excluded as they differed significantly from the other six groups. Results showed that 65 of 78 (83%) cell populations and all cell populations in 12 of 15 replicates grew and survived beyond the first subculture, while significantly different results were detected in cell populations from the other three replicates. These did not grow enough to progress to subculturing, with the exception of one cell population (0%–17% vs. 100%,
[Table 2.] Effects of individual difference of abalones used on primary culture outcome
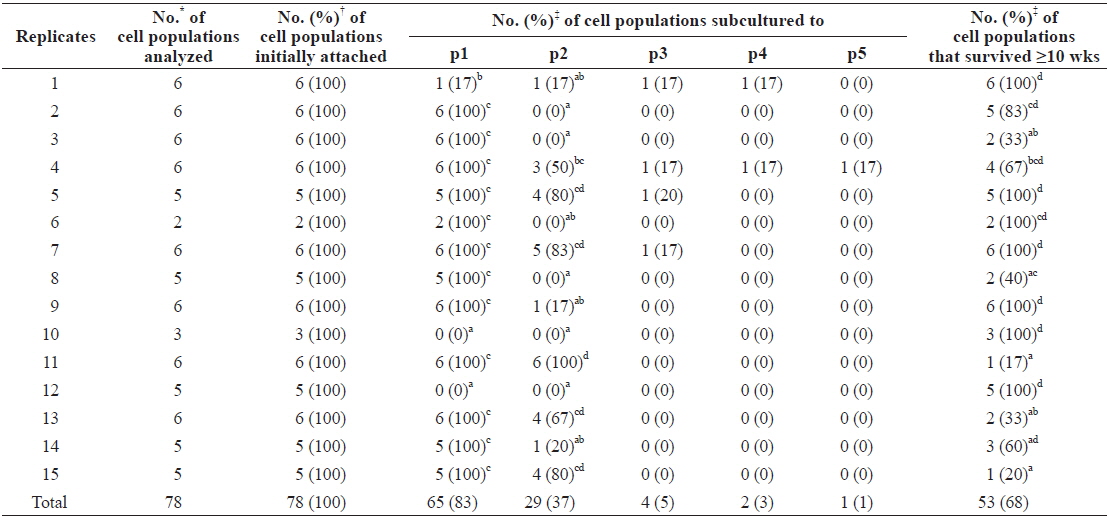
Effects of individual difference of abalones used on primary culture outcome
>
Cell morphology during culture
Throughout the culture of
>
Subculture period and cell death in early-phase cultures
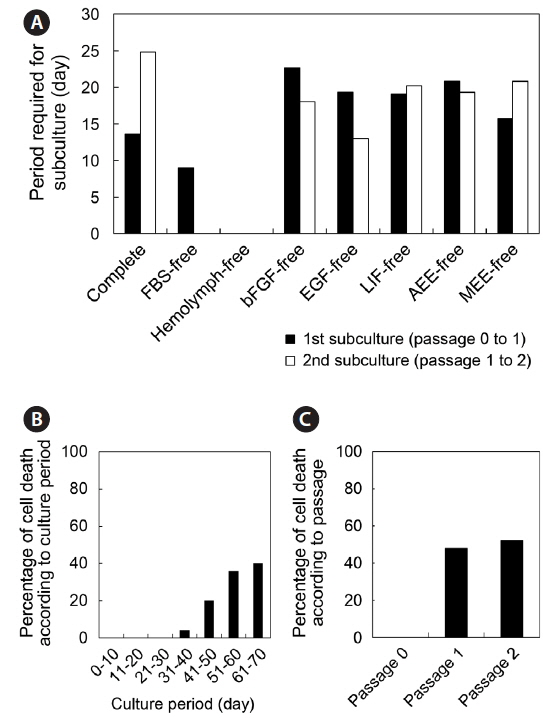
The period after initial cell seeding until first subculture varied from 7 to 65 days according to the cell populations analyzed, and the average period was 18.4 days. Likewise, the period between the first and second subculture varied from 4 to 43 days, with an average of 19.5 days (Fig. 2A). For analysis of early-phase cell deaths, the cell populations that died before 10 weeks of culture were subjected to these analyses but those cultured in FBS-free and hemolymph-free media were excluded. No cell death took place during the first 30 days of culture; the first deaths occurred on day 31, and 32% (25 of 78) of cell populations died prior to day 70. Cells deaths were concentrated between days 41 and 70, and increased over time until day 70 (Fig. 2B). In terms of passage number, cells deaths did not occur in passage 0, and their frequencies in passages 1 and 2 were 48% and 52%, respectively (Fig. 2C).
In this study, we investigated the effects of medium supplements on cultures of
We demonstrated that initial attachment of freshly isolated cell populations is not affected by either a lack of specific medium supplements or by individual differences, showing that all cell populations cultured attached at an average rate of 70% regardless of treatment. In a previous study, we revealed that supplements added to media containing FBS and hemolymph improved the initial attachment of abalone cells (Kim et al., 2014a). Since the same factors used in this previous report were added in this study, we assumed that similar effects on cell attachment would be induced, even if one factor was omitted in each experimental group. Another possibility, based on previous reports that gonad extract enhances the growth performance of oyster heart cells (Chen and Wen, 1999) and abalone mantle cells (Suja et al., 2007), is that ovary tissue extracts, which might be extracted during the tissue dissociation procedure, might positively affect cell attachment. Otherwise,
The growth of cell populations cultured with FBS-free or hemolymph-free media was significantly lower than that of other groups, indicating that FBS and hemolymph contain critical components for the stable growth and maintenance of
The growth and maintenance of cell populations cultured in several replicates were significantly lower, suggesting that individual differences play a major role in the outcome of culturing—although, as we used abalone pairs as single replicates, the term “individual difference” may be inappropriate. Regardless, the presence of individual differences is unsurprising and is a well-known phenomenon in biological experiments (d’Agostino, 1998). This should be carefully considered in invertebrate cell culturing, a field that is still under development, to reduce experimental error in future studies.
During maintenance of cell populations, we investigated general culturing aspects such as which cell morphologies appeared, the time required for subculturing, and the cell death rate during the early culture phase. We identified four distinct morphologies among the cultured cells, which may reflect different cell types. Alternately, cell populations initially consisting of one cell type may change their cell morphology during culture according to the culturing environment. Whatever the case, our data provide fundamental information on the morphologies that may appear during the culture of
Because abalone ovarian tissues consist of germline cells and various types of somatic cells (Wetakan et al., 2001; Awaji and Hamano, 2004), the cell populations cultured and maintained in this study need to be characterized to identify their cellular origin. The cell populations derived from germline cells can be used directly in biotechnological applications. On the other hand, somatic cells can be used as feeder cells for culturing ovary-derived germline stem cells. Molecular characterization of the cells cultured in this study should be conducted in further studies.
In conclusion, primary cultures of