The antioxidant and cholinesterase inhibitory activities of the acetone and dichloromethane (CH2Cl2) extracts of the by-products (heads, shells, and tails) of Pandalus borealis, Pandalus hypsinotus, and Pandalopsis japonica belonging to the family Pandalidae were investigated and their bioactivities were compared. The antioxidant and cholinesterase inhibitory activities of the organic solvent extracts of three shrimp by-products were evaluated using 2,2-diphenyl-1-picrylhydrazyl (DPPH) and 2,2'-Azino-bis[3-ethylbenzothiazoline-6-sulfonic acid] (ABTS+) radical scavenging activities, reducing power and xanthine oxidase (XO) inhibitory activity assays and Ellman's colorimetric method. The extracts of P. hypsinotus exhibited the highest antioxidant and cholinesterase inhibitory activities. The acetone extracts showed more potent activities toward antioxidant and cholinesterase inhibition compared with the CH2Cl2 extracts. Furthermore, the total carotenoid contents of the acetone extracts were higher than those of the CH2Cl2 extracts. Thus, the carotenoid contents may affect antioxidant and cholinesterase inhibition. Our results suggest that the shrimp by-products could act as a nutraceutical agent to prevent oxidative stress and Alzheimer’s disease.
Alzheimer’s disease (AD), a progressive neurodegenerative disease, is the most common type of senior dementia, characterized pathologically by the presence of senile plaques, neurofibrillary tangles, and extensive neuronal loss (Giannakopoulos et al., 1996). The cause of AD has not yet been clarified, but one of the most convincing theories is the cholinergic hypothesis of deficiency in the levels of neurotransmitters including acetylcholine (ACh) and butyrylcholine (BCh) (Schneider, 2001). Furthermore, excessive free radicals produced by oxidative stress are associated with the pathological changes observed in AD (Praticò and Delanty, 2000). Accordingly, the activation of cholinergic functions by inhibition of cholinesterase which hydrolyzes the cholinergic neuromediators, and reduction of oxidative stress has been used as a key approach in the treatment of AD (Schneider, 2001).
Shrimp of the family Pandalidae including
Several carotenoids, including β-carotene, α-carotene, lycopene, and lutein, derived from plants or animals, are effective in preventing cancer, cardiovascular disease, osteoporosis, and hypertension (Rao and Balachandran, 2003). However, there are few studies to date on the potential of shrimp by-products of the family Pandalidae and their carotenoid contents (Rao and Rao, 2007) in treating AD.
The aim of this study was to determine the effects on the antioxidant and cholinesterase (ChEs) activities of the acetone and CH2Cl2 extracts of the by-products of three shrimp species, and to investigate the correlation between the carotenoid contents of the extracts and their bioactivities.
The shrimp by-products were lyophilized (1 kg) and stored at −20°C until. The lyophilized samples (20 g) were extracted three times with 50 mL acetone or CH2Cl2, which is used in the analysis of various lipophilic materials including carotenoids (Sachindra et al., 2005; Rayees and Manish 2014). The yields of the shrimp by-product extracts were:
>
DPPH radical scavenging activity
The DPPH radical-scavenging activity was measured using a modification of the method of Blois (1958). DPPH solution (6.5 mM, 160 μL) was added to 40 μL sample. The samples were incubated in the dark for 10 min at room temperature. The absorbance was measured by spectrophotometer (BIO-TEK US/MQX 200, USA) at 540 nm. L-ascorbic acid was used as a positive control and the DPPH radical scavenging activity of each sample was expressed as the 50% inhibition value (IC50).
>
ABTS+ radical scavenging activity
The ABTS+ radical scavenging activity was determined using the method of Re et al. (1999). ABTS+ solution was diluted with water to an absorbance of 0.75 ± 0.03 at 734 nm. Then, 200 μL of ABTS+ solution was added to 10 μL samples of different concentrations. The samples were incubated in the dark for 10 min and measured the absorbance at 734 nm. Trolox was used as a positive control and the ABTS+ radical scavenging activity of each sample was expressed as an IC50 value.
The reducing power of the shrimp-shell extracts was measured using a modification of the method of Jayaprakasha et al. (2001). Various concentrations of samples in 10% DMSO (50 μL) were mixed with 50 μL 0.2 M sodium phosphate buffer (pH 6.6) and 50 μL potassium ferricyanide (10 mg/mL). The mixtures were incubated at 50°C for 20 min. Then, 50 μL of TCA (100 mg/mL) were added and the mixture was centrifuged at 2,000 × g for 10 min. After centrifugation, 100 μL supernatant was mixed with 20 μL iron (Ⅲ) chloride (1 mg/mL). The sample concentration resulting in 0.5 of absorbance (EC50) was calculated from the graph of absorbance at 700 nm, and L-ascorbic acid was used as a positive control.
>
Xanthine oxidase (XO) inhibitory activity
XO inhibition assays were performed using a modification of the method of Nongonierma and FitzGerald (2012). 50 μL of sample were treated in a 96-well plate containing EDTA (12.5 μM), hydroxylamine phosphate (25 μM), and xanthine (0.125 mM). In addition, 50 μL XO (0.1 U/mL) was treated in a 96-well plate and incubated at 37°C for 30 min. The absorbance of the uric acid formed was measured using a spectrophotometer (BIO-TEK US/MQX 200, USA) at 290 nm. BHA was used as a positive control and the XO inhibitory activity of each sample was expressed as an IC50 value.
ChEs inhibition was measured using the spectrophotometric method of Ellman et al. (1961). The reaction mixture contained 140 μL 100 mM sodium phosphate buffer (pH 8.0), 20 μL sample, and 20 μL of either AChE (0.36 U/mL) or BChE (0.36 U/mL). The solution was placed in a 96-well plate and mixed. After incubation at room temperature for 15 min, 10 μL DTNB solution and 10 μL ACh or BCh were added. The absorbance of all reactions was measured using a spectrophotometer (BIO-TEK US/MQX 200, USA) at 412 nm and eserine was used as a positive control. The ChEs inhibitory activity of each sample was expressed as an IC50 value.
A spectrophotometric method was used to evaluate the total carotenoid contents following a modification of the method of Tolasa et al. (2005). Astaxanthin standard (3.0 mg) and BHT (100 mg) were dissolved in 10 mL CH2Cl2. Subsequently, 1 mL of this stock solution was diluted to 10 mL with
Castaxanthin (μg/mL) = A × 10,000/E,
where Castaxanthin is the total carotenoid contents, A is the absorbance at 472 nm, E is the extinction coefficient, and 10,000 is the scale factor.
To provide data to plot the standard curve, 0.1, 0.25, 0.50, 0.75, 1.0, 1.25, and 1.5 mL of stock solution were placed in separate 10 mL flasks and made up to the appropriate volume with n-hexane. The absorbance was measured at 472 nm.
The data were analyzed using analysis of variance following the general linear model procedure (SAS Institute, Cary, NC, USA). Duncan’s multiple-range test was applied to determine the significance of differences between means (
Reactive oxygen species (ROS) including superoxide (O2−), hydrogen peroxide (H2O2), and hydroxyl radical (HO) are generated by respiration and normal metabolism (Yen and Chen, 1995). Their production in the body causes various diseases such as neurodegenerative diseases (e.g., AD), cancer, rheumatoid arthritis, and atherosclerosis through cell or DNA damage (Cerutti, 1985; Squadrito and Pryor, 1998; Cooke et al., 2003).
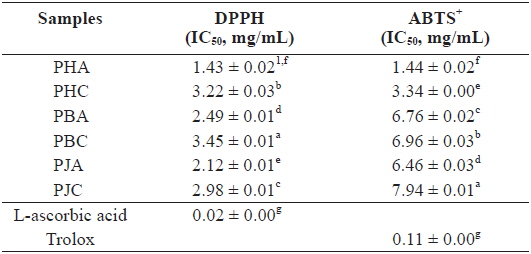
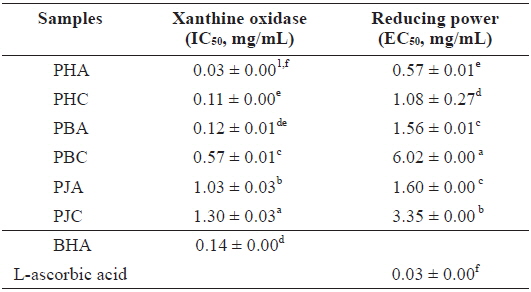
We investigated the antioxidant and ChEs inhibitory activities of the acetone and CH2Cl2 extracts of the by-products of three shrimp species based on the DPPH and ABTS+ radical scavenging, xanthine oxidase inhibitory activities, and reducing power assays (Tables 1 and 2). Of these, PHA showed the most potent DPPH and ABTS+ radical scavenging activities with IC50 values of 1.43 ± 0.02 and 1.44 ± 0.02 mg/mL, respectively. However, all extracts exhibited lower DPPH and ABTS+ radical scavenging activities than those of the positive controls, L-ascorbic acid and trolox. The xanthine oxidase inhibitory activities of the acetone and CH2Cl2 extracts of three shrimp by-products were (Table 2): PHA (IC50 = 0.03 ± 0.00 mg/mL) > PHC (IC50 = 0.11 ± 0.00 mg/mL) > PBA (IC50 = 0.12 ± 0.01 mg/mL) > PBC (IC50 = 0.57 ± 0.01 mg/mL) > PJA (IC50 = 1.03 ± 0.03 mg/mL) > PJC (IC50 = 1.30 ± 0.03 mg/mL). In particular, PHA was found to be 47-fold stronger than the positive control, BHA (IC50 = 0.14 ± 0.00 mg/mL). Moreover, the inhibition of PBA toward the xanthine oxidase activity was similar to that of BHA when used as a positive control. However, PJA exerted no notable activity on xanthine oxidase inhibition.
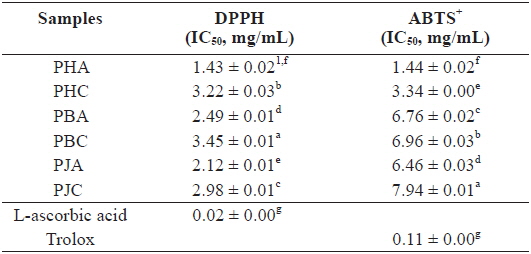
DPPH and ABTS+ radical scavenging activities of the acetone and dichloromethane extracts of three shrimp by-products
The electron donation capacity of the acetone and CH2Cl2 extracts are shown in Table 2. PHA (EC50 = 0.57 ± 0.01 mg/mL) exhibited the greatest reducing power and the other extracts showed moderate electron donation capacity. However, all extracts showed lower reducing power than that of L-ascorbic acid, used as a positive control.
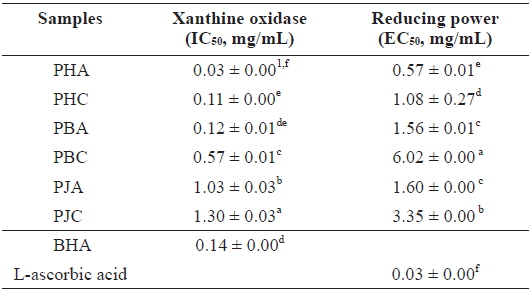
Xanthine oxidase inhibitory activity and reducing power of the acetone and dichloromethane extracts of three shrimp by-products
These results indicate that the acetone extracts of the shrimp by-products possessed potent scavenging activity toward DPPH and ABTS+ radicals, as well as xanthine oxidase inhibitory activity and reducing power, and their activities were higher than those of the CH2Cl2 extracts.
The neurotransmitters including ACh and BCh were decomposed by ChEs (Schneider, 2001). Loss of neurotransmitters at the synaptic site of the AD patient’s brain is associated with memory and cognitive deficits (Johnson et al., 2000). Therefore, prevention of neurotransmitter loss through inhibition of ChEs activation has been accepted as the most effective treatment in the pathology of AD (Giacobini, 2004).
The ChEs inhibitory activity of the acetone and CH2Cl2 extracts of the shrimp by-products was measured using AChE and BChE inhibitory activity assays (Table 3). Among the extracts, PHA showed strong inhibitory activity against both AChE and BChE, with IC50 values of 0.20 ± 0.01 and 0.67 ± 0.03 mg/mL, respectively. PJA (IC50 = 0.30 ± 0.03, 1.16 ± 0.02 mg/mL) and PBA (IC50 = 0.36 ± 0.01, 1.18 ± 0.02 mg/mL) also exhibited ChEs inhibitory activity. The CH2Cl2 extracts, PHC (0.56 ± 0.02 and 0.76 ± 0.03 mg/mL), PBC (0.64 ± 0.02 and 2.05 ± 0.02 mg/mL), and PJC (0.94 ± 0.03 and 1.76 ±0.04 mg/mL), showed moderate ChEs inhibitory activity. However, all extracts showed lower ChEs inhibitory activities than that of Eserine, which was used as a positive control (IC50 = 0.00011 ± 0.00000 and 0.00013 ± 0.00000 mg/mL).
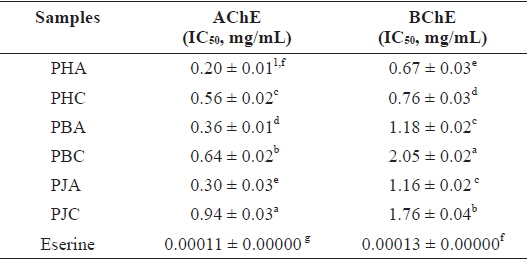
Cholinesterase inhibitory activity of the acetone and dichloromethane extracts of three shrimp by-products
Carotenoids exist as natural pigments in terrestrial plants, seaweeds, microorganisms, and crustaceans, and exert various biological effects, such as an inhibitory effect against oxidative damage, and prevention of cardiovascular disease and cancer (Davies, 1985; Kohlmeier and Hastings, 1995; Stahl et al., 1998; Fraser and Bramley, 2004). Therefore, we evaluated the total carotenoid contents of the acetone and CH2Cl2 extracts and investigated the correlation between the carotenoid contents and their biological activities.
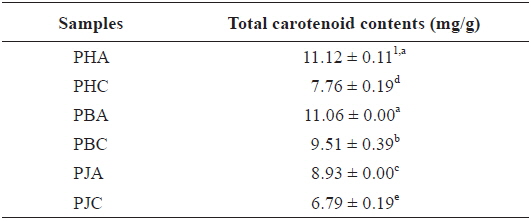
The total carotenoid contents of the acetone and CH2Cl2 extracts (Table 4): PHA (11.12 ± 0.11 mg/g) > PBA (11.06 ± 0.00 mg/g) > PBC (9.51 ± 0.39 mg/g) > PJA (8.93 ± 0.00 mg/g) > PHC (7.76 ± 0.19 mg/g) > PJC (6.79 ± 0.19 mg/g). The acetone extracts including PHA, PBA, and PJA exhibited higher total carotenoid contents compared with the CH2Cl2 extracts. In particular, PHA exhibited the highest total carotenoid contents.
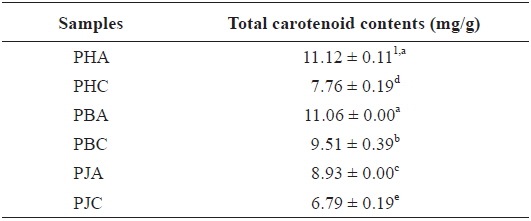
Total carotenoid contents of the acetone and dichloromethane extracts of three shrimp by-products
In the present study, total carotenoid contents was suggested to be related to antioxidant and inhibitory activities. Recent researches have suggested several possible roles of carotenoids in neurodegenerative diseases, including AD and oxidative stress (Rao and Balachandran, 2003). High levels of reactive oxygen species and low antioxidant capacity cause damage to the human brain, which is vulnerable to oxidative damage, and trigger development of neurodegenerative diseases. Moreover, significant reductions in the levels of carotenoids were reported in Alzheimer’s and parkinson’s diseases and vascular dementia patients (Foy et al., 1999).
Thus, the antioxidant and ChE inhibitory activities of shrimp by-product extracts are possibly attributable to carotenoids. Further study should focus on isolating the active components of shrimp by-products and investigating the underlying mechanisms.