The quality of dietary protein is determined by its amino acid (AA) composition and availability (Wilson and Cowey, 1985; Wilson and Poe, 1985). Quantitative estimation of the essential AA requirements of cultured fish species is very important to achieve optimum growth and feed utilization, cost-effective diet formulation, and desirable carcass quality (NRC, 2011). It is well established that a balanced AA profile is required for the effective use of dietary protein in tissue synthesis (D’Mello, 1994; Yamamoto et al., 2000; Berge et al., 2002; Green and Hardy, 2002; Gόmez-Requeni et al., 2003). AA can be supplied as protein-bound AA, free AA, or peptides in formulated diets. Absorption of peptides and free AA (FAA) are the major transport routes for protein use in mammals (Ganapathy et al., 1994; Adibi, 1997). The current understanding of protein use in vertebrates centers on the absorption of peptides in the intestine as a major route of transport. In fishes, small peptides and FAA are absorbed faster than protein (Rust et al., 1993; Rønnestad et al., 2003), and there is evidence that single peptides can be more efficiently absorbed than a mixture of identical AA in both fish (Reshkin and Ahearn, 1991; Boge et al., 2002) and mammals (Matthews, 1991). Previous studies on common carp
Dabrowski et al. (2003) showed that a synthetic dipeptide-based diet could support the early stages of rainbow trout growth, whereas a free AA-based diet could not. Peptides have different transport systems than FAA has. Tetra- and larger peptides do not cover nitrogen requirements in the absence of pancreatic enzymes or brush border peptidase activity (Grimble, 1994; Daniel, 2004), whereas di- or tripeptides have specific peptide transporters (Doring et al., 1998). These transporters are expressed in teleost larvae prior to exogenous feeding (Verri et al., 2003).
Lysine has been identified as an essential AA for all studied fish species (NRC, 2011) and has been found in high concentrations in the carcasses of many fishes (Wilson and Cowey, 1985; Wilson and Poe, 1985; NRC, 1993). Lysine deficiency has been reported to result in decreased growth and low feed efficiency (Ahmed and Khan, 2004; Wang et al., 2005; Zhou et al., 2008). It is the most limiting amino acid in the protein sources, particularly plant proteins, used in aquafeeds (Deng et al., 2010).
Olive flounder
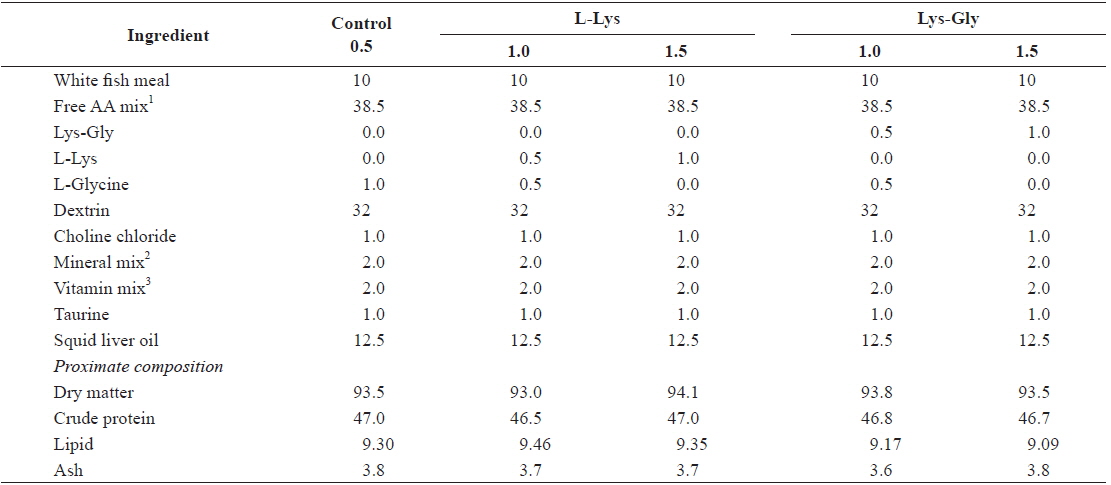
Formulation and proximate composition of the experimental diets are shown in Table 1. Five isonitrogenous (47% crude protein) and isocaloric (4.38 kcal g-1 gross energy) experimental diets were formulated to contain different levels and forms of lysine. A mixture of synthetic free AA without lysine was prepared according to Dabrowski et al. (2003) and used as the main protein source. A control diet was formulated to contain basal levels of lysine (0.5%) from fish meal and supplemented with 0.5% or 1.0% lysine in either the free or the dipeptide form. Lysine-Glycine (LG) (Bachem, Torrance, CA, USA) was used as the dipeptide form, and crystalline L-lysine (L-Lys) (Sigma Chemicals, St. Louis, MO, USA) as the free form (FL). The experimental diets were kept isonitrogenous using glycine at the expense of lysine. All ingredients were well mixed, pelletized, and freeze-dried. The pellets were crushed into desirable particle sizes and stored at –20°C until use.
[Table 1.] Composition and proximate analysis of the experimental diets (% dry matter)
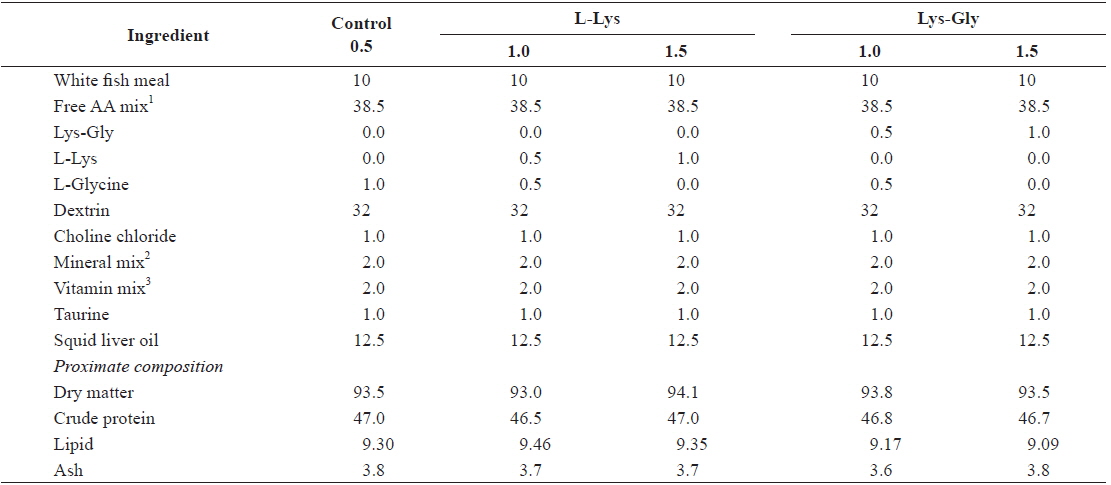
Composition and proximate analysis of the experimental diets (% dry matter)
>
Fish and experimental conditions
Juvenile olive flounder were transported from a private hatchery to the Institute of Marine Sciences at Jeju National University (Jeju, South Korea). All fish were fed the basal diet for 1 week to allow acclimation to both the semi-purified diet and the experimental conditions. At the end of the acclimation period, 20 randomly selected fish (averaging 5.41 ± 0.16 g) were stocked into polyvinyl circular tanks (20-L capacity), supplied with filtered seawater at a flow rate of 1.5 L min-1, and aerated to maintain sufficient dissolved oxygen. Triplicate groups of fish were hand-fed with one of the test diets at a ratio of 3% of their body mass (twice a day, 09:00 and 17:00 h) for 6 weeks. The growth of fish was measured at 2-week intervals. Feeding was stopped 24 h prior to weighing to minimize handling stress. The water temperature during the feeding trial ranged from 14 to 20°C, and the photoperiod was maintained on a 12:12 light:dark schedule.
>
Sample collection and analyses
At the end of the feeding trial, all fish in each tank were bulk-weighed and counted for growth parameters and survival. Five intact fish per tank (15 fish per treatment) were selected and kept at -20°C for whole-body amino acid composition analysis. The samples were freeze-dried and finely ground using a grinder, and their amino acid compositions was analyzed using an automatic amino acid analyzer (Biochrom 30, Pharmacia Biotech, Cambridge, England).
Another set of five fish per tank were sampled for determination of organosomatic indices, including hepatosomatic index (HSI), viscerosomatic index (VSI), and relative intestine length (RIL).
Analyses of the moisture and ash contents of the diets were performed using standard procedures (Association of Official Analytical Chemists, 1995). Crude protein was measured using an automatic Kjeltec Analyzer Unit 2300 (FossTecator, Höganäs, Sweden), and crude lipid content was determined using the Soxhlet method with extraction in diethyl ether (Soxhlet Extraction System C-SH6, Korea).
All experimental diets were assigned by a completely randomized design. Data were analyzed by one-way ANOVA using SPSS version 11.0 (SPSS, Inc., Chicago, IL, USA). When an ANOVA identified differences among groups, the means were compared using Tukey’s HSD multiple range test. Statistical significance was determined by setting the aggregate type 1 error at 5% (
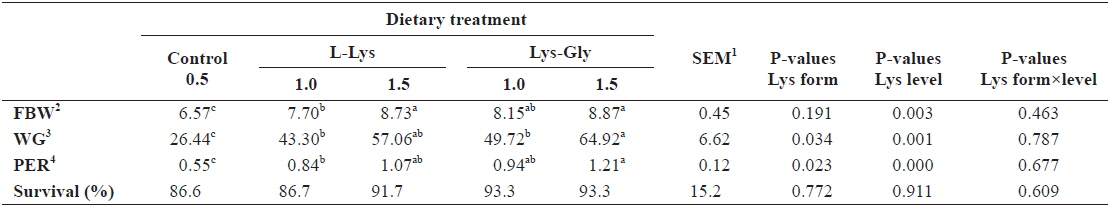
Results showed significant improvement in fish growth with each increment of dietary lysine (
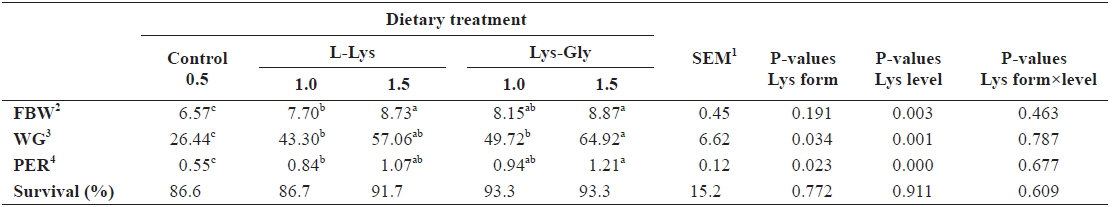
Growth performance of olive flounder Paralichthys olivaceus (initial body weight, 5.41 ± 0.16 g) fed the experimental diets containing different lysine levels and molecular forms for 6 weeks
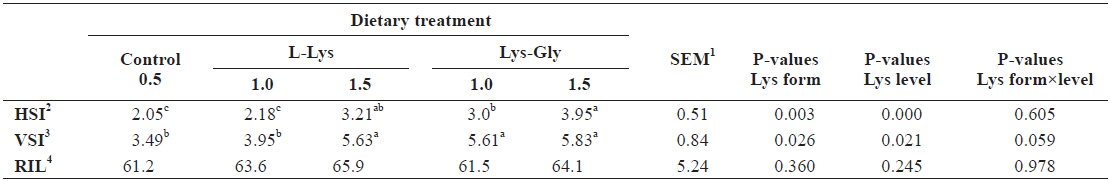
Organosomatic indices of fish fed the experimental diets are presented in Table 3. Significantly higher HSI and VSI were obtained in fish fed diets containing LG, and in those fed the free form diet at 1.5% (
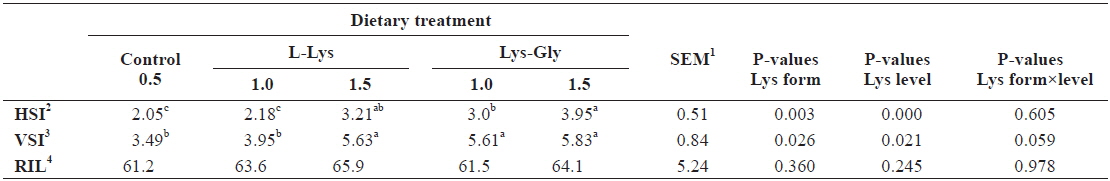
Organosomatic indices of olive flounder Paralichthys olivaceus, (initial body weight, 5.41 ± 0.16 g) fed the experimental diets containing different lysine levels and molecular forms for 6 weeks
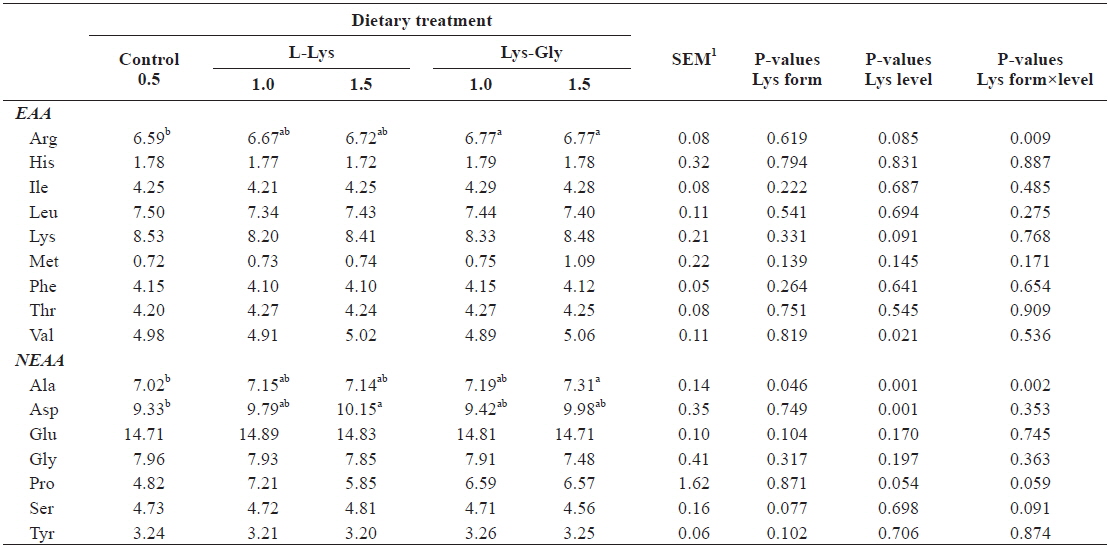
Whole-body amino acid composition was significantly affected by dietary treatment (Table 4). Significantly higher arginine levels were found in fish fed diets containing LG (one-way ANOVA), and we found a significant interaction between lysine level and form (P = 0.009, two-way ANOVA). Two-way ANOVA analysis showed a significant effect of lysine level on whole-body valine and aspartic acid contents. Moreover, alanine content was significantly higher in fish offered 1.5% LG (one-way ANOVA), and it was significantly influenced by both lysine level and form.
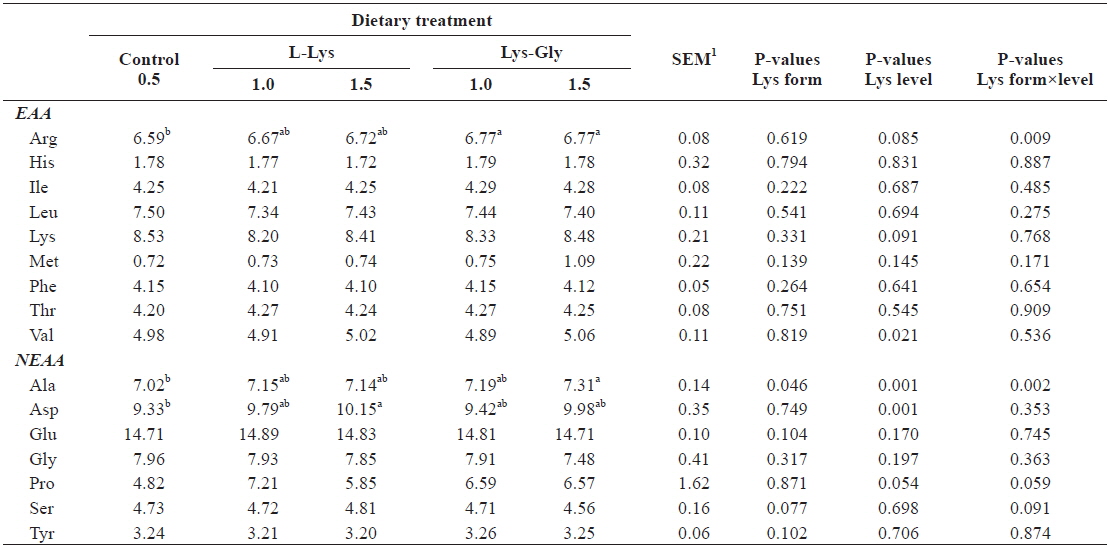
Whole-body amino acid composition (% protein) of olive flounder Paralichthys olivaceus, fed the experimental diets containing different lysine levels and molecular forms for 6 weeks
Fish in our study accepted the semi-purified diets but grew relatively slowly because the trials occurred in low temperature season. Our results show a significant enhancement of fish growth with each increment of dietary lysine, indicating the importance of lysine for maximal growth in juvenile olive flounder. Forster and Ogata (1998) reported that the lysine requirement of olive flounder is 1.5–2.1% of their diet. Accordingly, we used a range of 0.5 to 1.5% lysine to compare the efficiency of its free and dipeptide forms.
Dabrowski et al. (2003) reported that a dipeptide-based diet could support the early-life growth performance of rainbow trout, whereas a FAA-based diet could not. In our study, fish fed LG supplemented diets showed higher growth than those fed FL, and the highest weight gain occurred in those fed 1.5% LG. Similarly, Kim and Lee (2013) found higher weight gain at all supplementation levels of the dipeptide form of leucine than of the free form. In contrast, Kim et al. (2012) reported similar efficiency for the dipeptide and free forms of phenylalanine when they supplemented in diets for red sea bream
Composition of the basal diet is an important factor influencing the results of dipeptide AA supplementation. In this study, the utilization efficiencies of the two lysine forms were examined by supplementing them into a free AA-based basal diet, whereas previous studies have supplemented wholeprotein based diets (Tesser et al., 2005: Kwasek et al., 2012). The differences in AA availability from free AA and wholeprotein-based diets may at least partially explain the observed variations. Moreover, it has been suggested that the use of different dipeptides can profoundly affect absorption characteristics by switching the AA sequence (Daniel, 2004). Cytosolic and brush border aminopeptidases are involved in the utilization of dietary peptides (Cahu and ZamboninoInfante, 1995; Kurokawa and Suzuki, 1998). However, further studies are required to clarify the mechanism by which dipeptides mediate specific peptide transporters in the intestinal brush border epithelium.
We showed that both lysine level and form affected the weight gain of olive flounder. Kim and Lee (2013) reported that the molecular form of leucine, and not its level, was the main factor in olive flounder growth performance. This variation may be due to differences in fish size, feeding period, dietary requirements, and experimental conditions.
The study of HSI and VSI provides important information on the metabolism of fishes, especially as related to digestion and absorption, and on the synthesis and secretion of digestive enzymes (McLaughlin, 1983). HSI is used as an indicator of general nutritional status (Busacker et al., 1990). We obtained significantly higher HSI and VSI values with each increment of dietary lysine; this was more evident when the dipeptide form was used, indicating a higher energy budget in LG fed fish.
The whole-body AA profile has been used as a standard indicator of fish AA requirements (Wilson and Cowey, 1985; Bicudo and Cyrino, 2009). Several studies have shown that whole-body essential AA profiles provide a good estimate of the optimum essential AA profile in fish diets (Akiyama et al., 1997; Green and Hardy, 2002). Aragão et al. (2004) showed that juvenile Senegal sole
In conclusion, juvenile olive flounder can utilize lysine more efficiently for growth if it is provided in dipeptide form, and the dipeptide form can improve AA retention in fish. Overall, we provide further evidence for the notion that previously published data on AA requirements using free AA are likely to be overestimated and need to be reevaluated using dipeptides as the AA source.