This study was carried out in order to find both the single-dose intramuscular injection toxicity and the approximate lethal dose of samjeong pharmacopuncture (SP) in Sprague-Dawley (SD) rats.
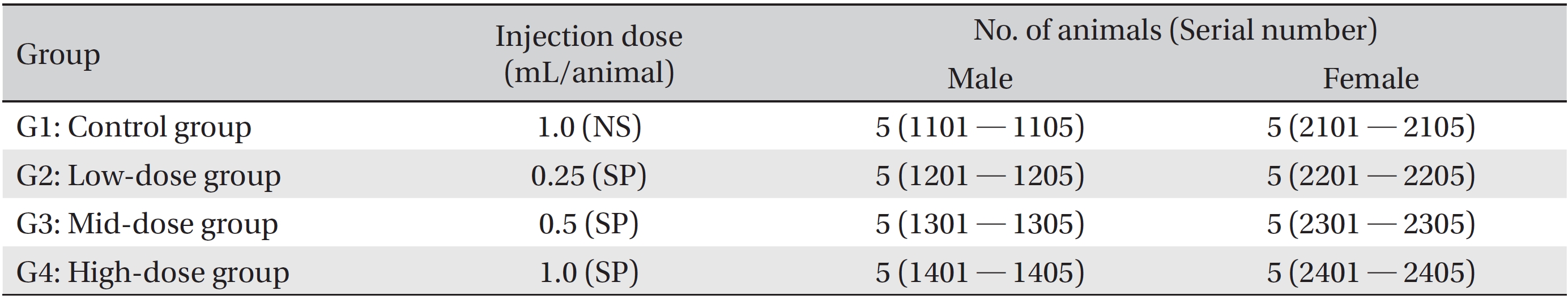
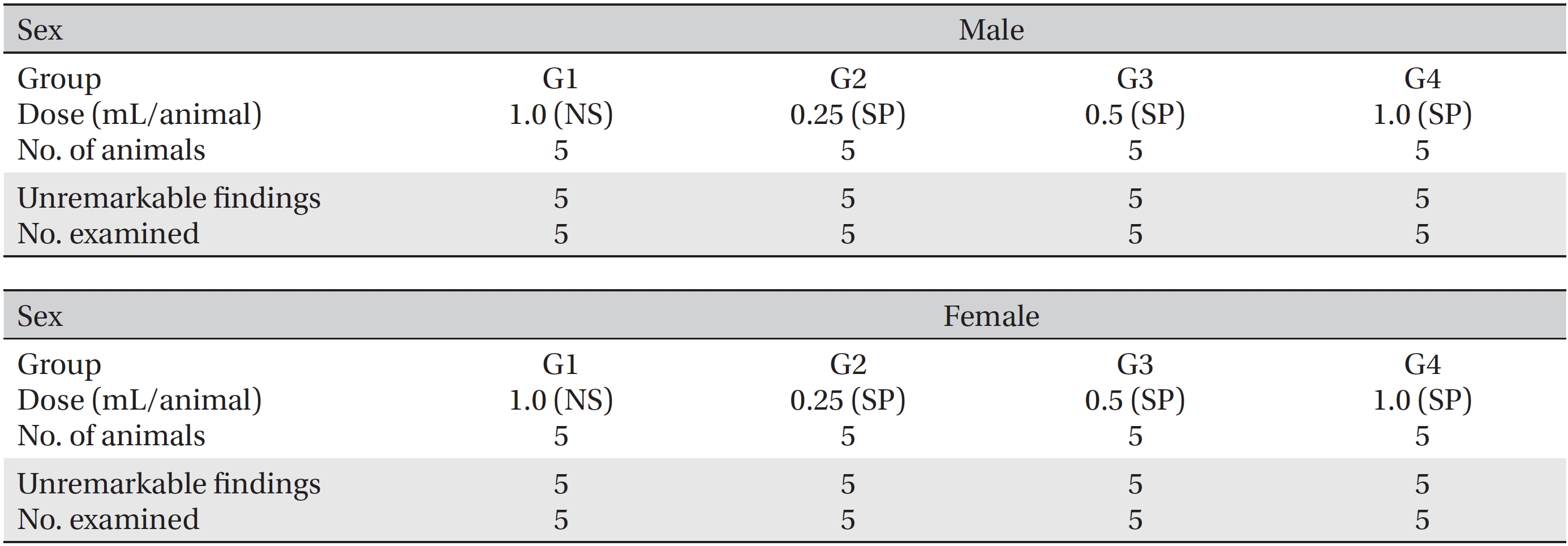
The SD rats in this study were divided into four groups, one control group (1.0 mL/animal, normal saline) and three experimental groups (0.25, 0.5, and 1.0 mL/animal, SP). All groups consisted of five male and five female rats. SP was injected as a single-dose intramuscularly at the thigh. After the injection, general symptoms and weight were observed for 14 days. After the observations had ended, hematologic and serum biochemical examinations, necropsy and a local tolerance test at the injection site were performed. The experiments were carried out at the Good Laboratory Practice firm, Biotoxtech Co. (Cheongwon, Chungbuk). Animal experiments were approved by the Ethics Committee (Approval Number: 130379).
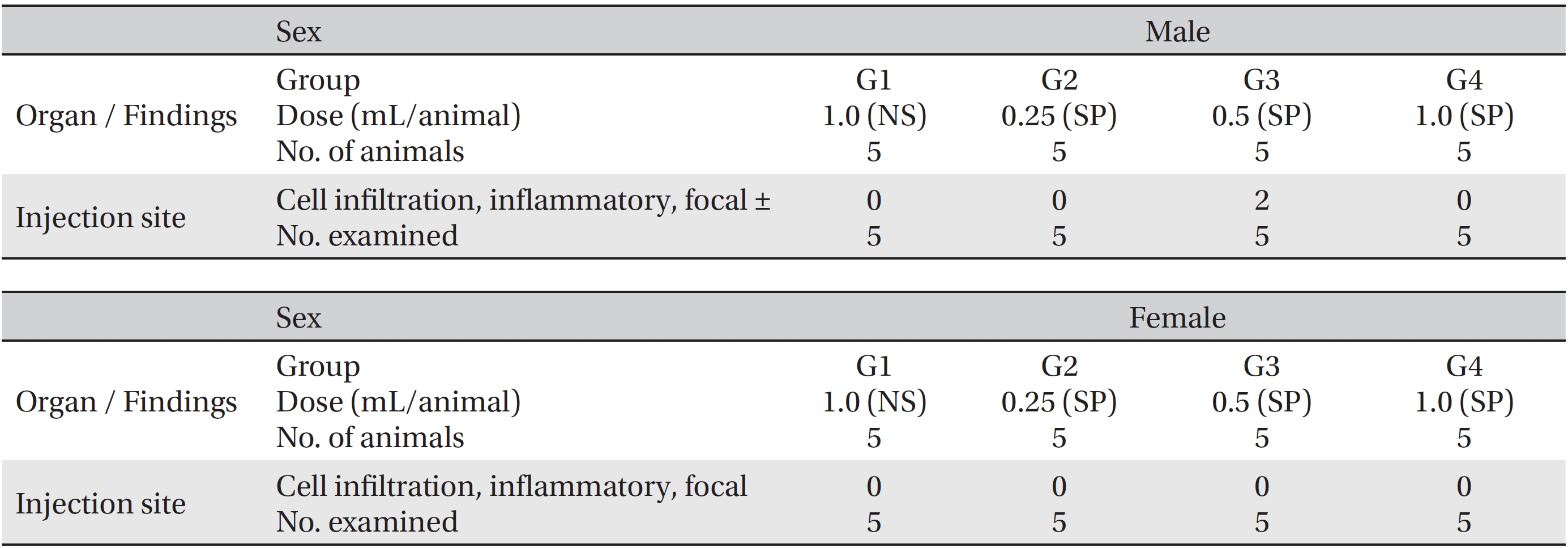
No deaths occurred in any of the three experimental groups. The injection of SP had no effects on the general symptoms, body weights, results of the hematologic, and serum biochemical examinations, and necropsy findings. In local tolerance tests at the injection sites, mild inflammation was observed in the experimental group, but it did not appear to be a treatment related effect.
Under the conditions of this test, the results from the injection of SP suggest that the approximate lethal dose of SP is above 1.0 mL/animal for both male and female SD rats. Therefore, the clinical use of SP is thought to be safe.
Pharmacopuncture is a treatment that combines herbal medicine and acupuncture, and is characterized by injections at acupoints [1]. Most pharmacopuncture contains certain amounts of herbal extracts and is injected intramuscularly. On the basis of the drug toxicity testing standards of the Korea Food and Drug Administration (KFDA), toxicity test should be carried out in case of injections. Single-dose toxicity tests [2-9] and repeated dose toxicity tests [10-15] have been performed on many pharmacopuncture.
In 2012, samjeong solution (SS), which has an anti- inflammation effect, and consists of
The SS used in this study was manufactured by using a low temperature extracting process and was made at the Good Manufacturing Practice (GMP) facilities of the Korea Pharmacopuncture Institute (KPI). The SS was used for the experimental groups, and normal saline (NS) was used for control group.
Because Sprague-Dawley (SD) rats (Orientbio Inc., Korea) are widely used for safety tests, they were used in this study. Visual inspections were done when the 5 week old SD rats (males: 24, females: 24) were received, and their weights were 119.0 ─ 142.9 g (males) and 104.0 ─ 119.9 g (females). During the 6 days stabilization period, the general symptoms were observed daily. The body weights, general symptoms and weight changes were measured on the last day of stabilization. The animals showed no abnormal changes. Six week old SD rats (males: 20, females: 20) were used in the experiment, and their weights were 180.3 ─ 197.5 g (males) and 139.1 ─ 156.1 g (females). Five rats of each sex were assigned to each of four groups: one control group and three experimental groups. During the observation period, SD rats were bred in cages (260 ㎜ (w) × 350 ㎜ (d) × 210 ㎜ (h)). The breeding environment was maintained at a temperature of 21.1 ─ 22.9℃, a relative humidity of 43.2% ─ 59.6%, and a ventilation rate 10 ─ 15 times/hour with artificial lighting from 07:00 to 19:00 (150 ─ 300 Lux). Animals were allowed free access to pellet feed (Teklad Certified Irradiated Global 18% Protein Rodent Diet 2918C) and irradiation sterilized water.
The clinical dose of SP is an intramuscular injection of 1.0 mL/time. The results of a pilot test (Biotoxtech Study No.: B13479P) showed no deaths for a single-dose intramuscular injection of 1.0 mL/animal in both male and female SD rats. Based on that result, in this study, we divided 40 SD rats into four groups of 5 male and 5 female rats per groups: 1 control group and 3 experimental groups (Table 1). SP was injected intramuscularly. The injected doses in the experimental groups were 1.0 (high dose), 0.5 (mid dose) and 0.25 (low dose) mL/animal; the injected were done by using disposable syringes (1 mL, 26G). In the high dose group (1.0 mL/animal of SP) and the control groups (1.0 mL/animal of NS, 0.5 mL was injected intramuscularly into both thighs. In the mid and the low dose groups, the SP was injected into the left thigh muscle.
General symptoms, toxic signs, onset and recovery times, etc., were noted at 30 minutes, 1, 2, 4 and 6 hours after the first injection. General symptoms were observed daily for 14 day after injection.
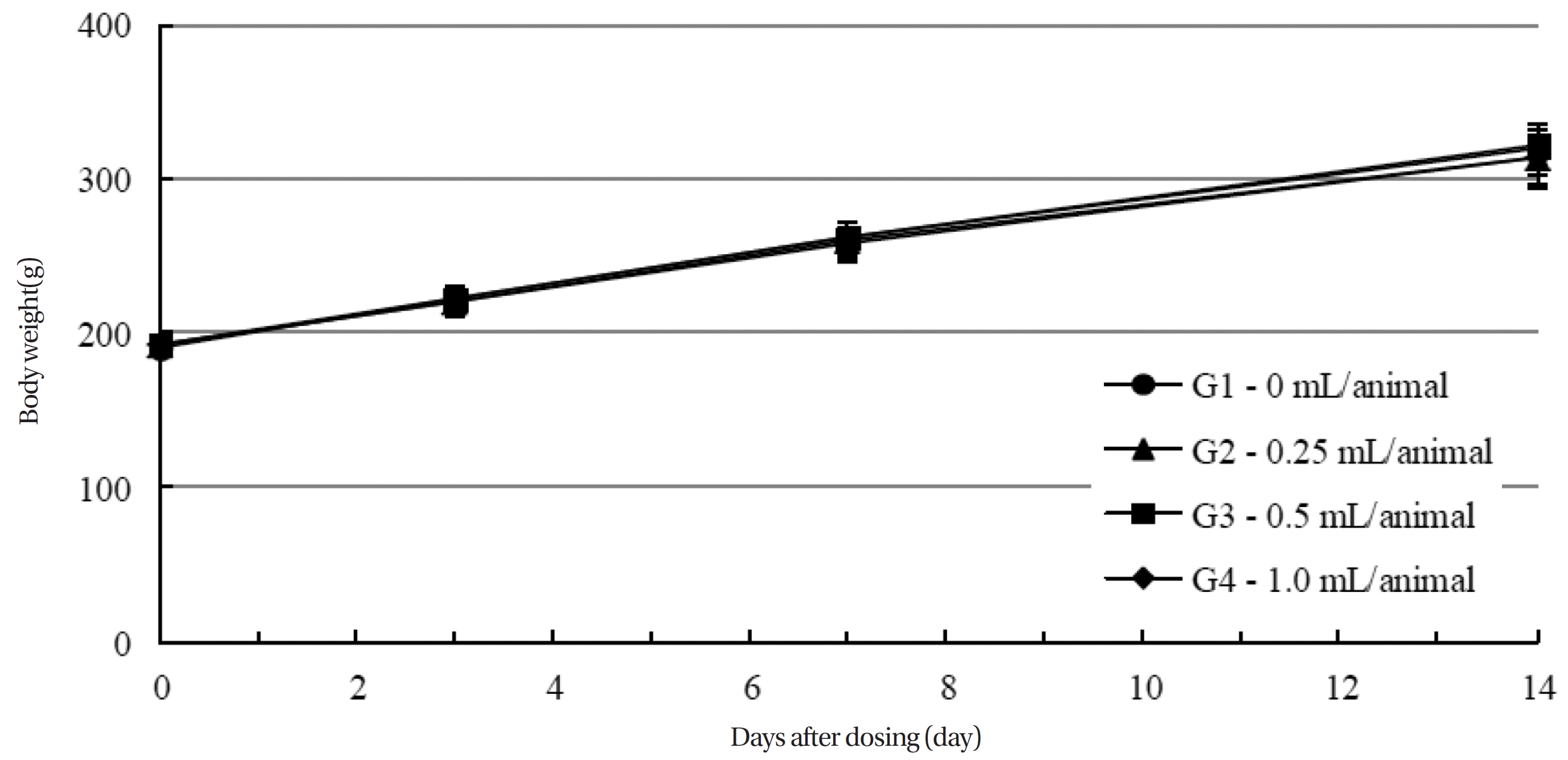
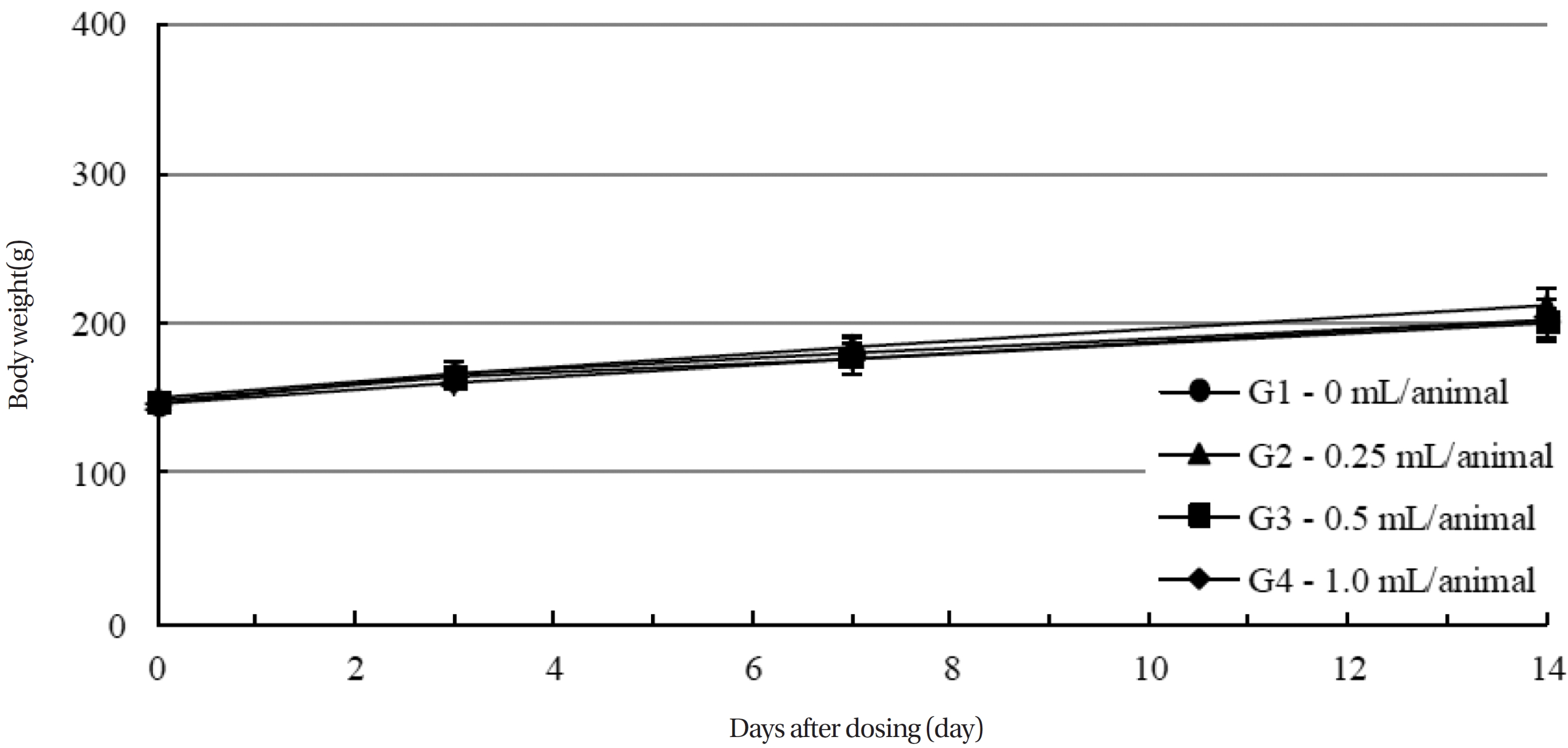
Body weights were measured on the day of the injection (before injection), and on the third, seventh and fourteenth days after injection.
After all animals had fasted for about 18 hours before necropsy, the animals were anesthetized by using isoflurane anesthesia, and blood was collected from the abdominal aorta on the fifteenth day (necropsy day). The collected blood was encased in a bottle containing ethylenediaminetetraacetic acid (EDTA); hematologic examinations were performed by using a blood counting analyzer (ADVIA 2120i, Siemens, Germany). Items measured were the erythrocyte count (RBC), hemoglobin (HGB), hematocrits (HCT), mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), platelets (PLT), leucocyte count (WBC), WBC differential count (neutrophils, lymphocytes, monocytes, eosinophils, basophils) and reticulocytes (Reti). About 2 mL of the taken blood was placed into a tube containing 3.2% sodium citrate and after centrifugation (3,000 rpm) for 10 minutes, coagulation tests were performed by taking the blood plasma. The prothrombin time (PT) and the activated partial thromboplastin time (APTT) were measured by using an automated coagulation analyzer (Coapresta 2000, Sekisui, Japan).
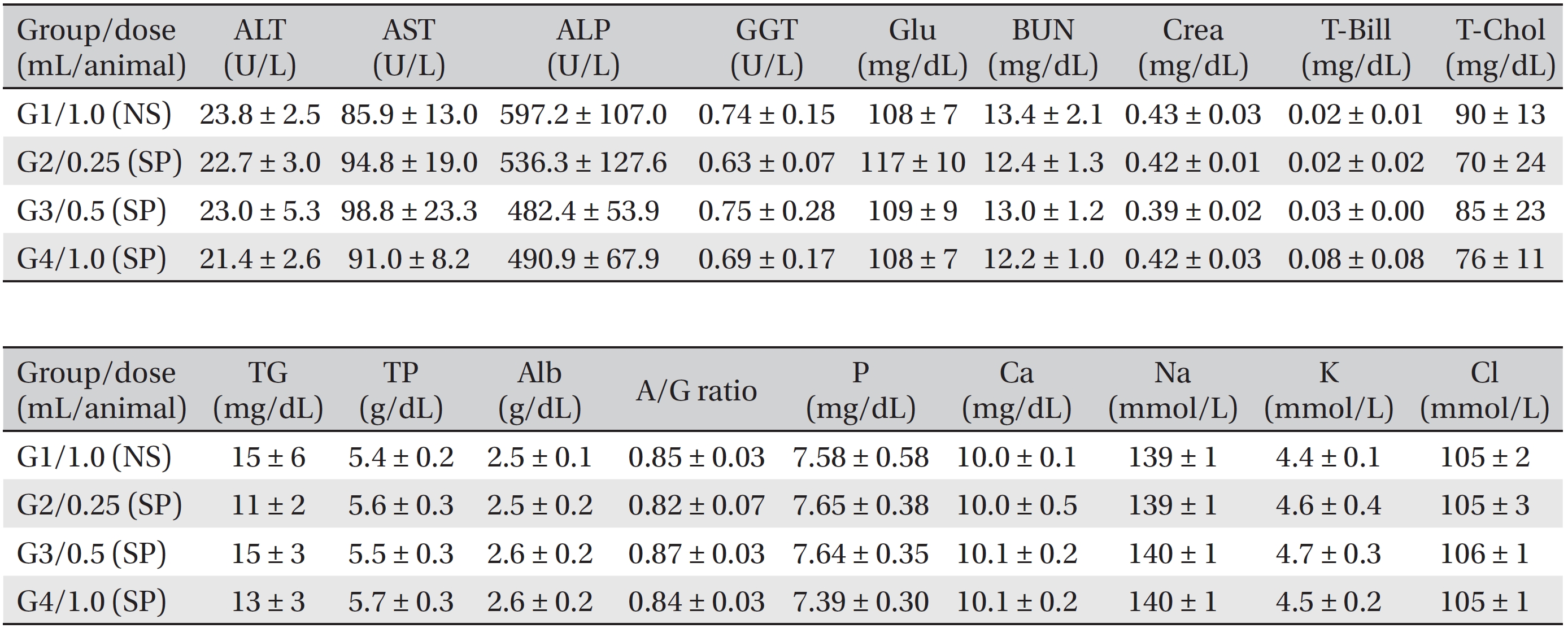
Blood biochemical examinations were performed after the blood samples taken from the abdominal aorta had been centrifuged at 3,000 rpm for 10 minutes. Items, including alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), gamma glutamyl transpeptidase (GGT), blood urea nitrogen (BUN), creatinine (Crea), total bilirubin (T-Bili), total protein (TP), albumin (Alb), albumin/globulin (A/G) ratio, total cholesterol (T-Chol), triglycerides (TG), phosphorus (P), glucose (Glu), calcium (Ca), chloride (Cl), sodium (Na) and potassium (K) were measured by using a blood biochemical analyzer (7180, Hitachi, Japan) and an electrolyte analyzer (AVL9181, Roche, Germany).
After the observation period, necropsies were conducted, and detailed visual inspections of the organs and the tissues of all animals were done. The harvested organs and tissues were fixed with 10% neutral buffered formalin. The sections of the fixed organs and tissues were made by using a routine tissue processing procedure. The sections were stained with hematoxylin and eosin (H&E). In addition, histopathological examinations of the injection sites were done, and all organs and tissues were visually inspected.
Weights and hematology and blood biochemistry data were analyzed by using statistical analysis system (SAS, version 9.3, SAS Institute Inc., U.S.A.). Data are presented as means ± standard deviations (SDs). The test for equal variance was the Bartlett test (
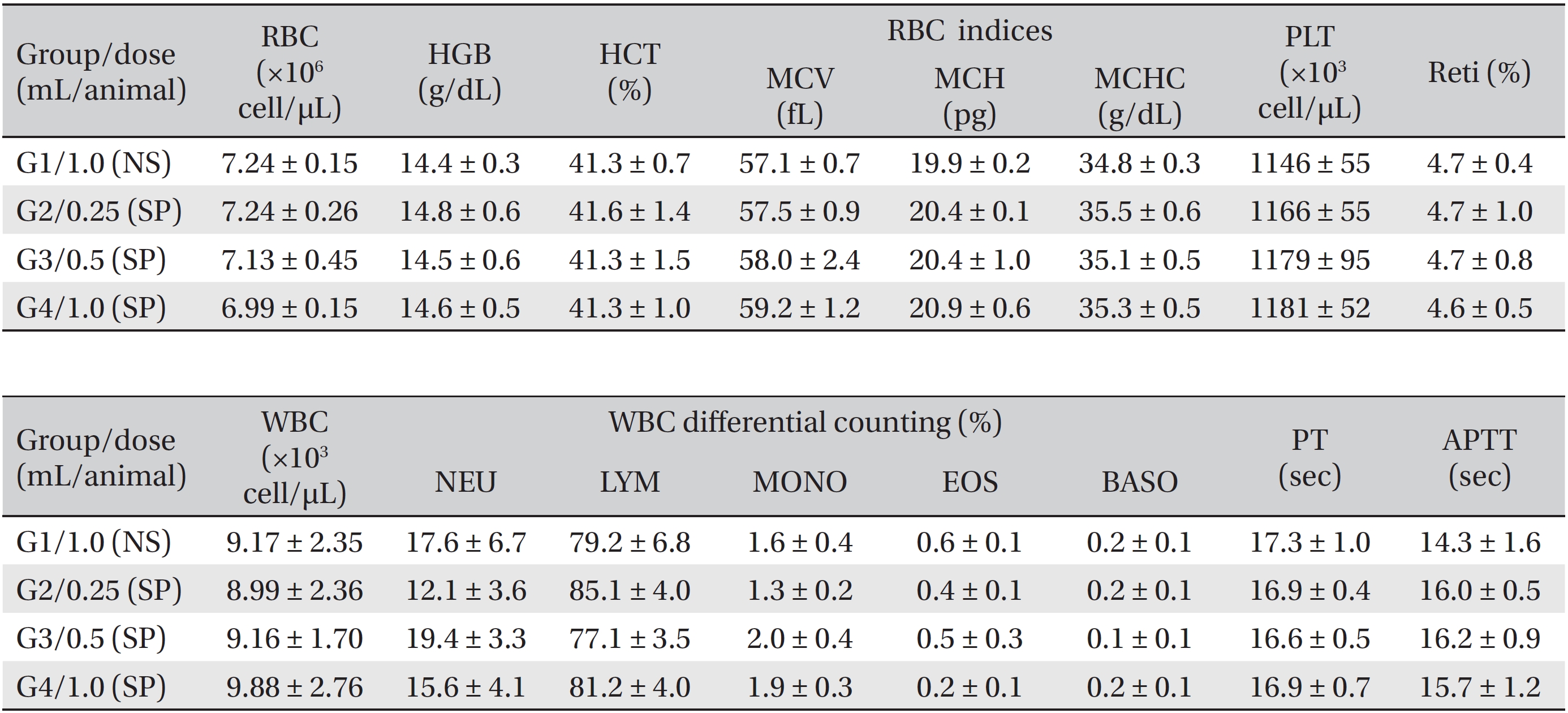
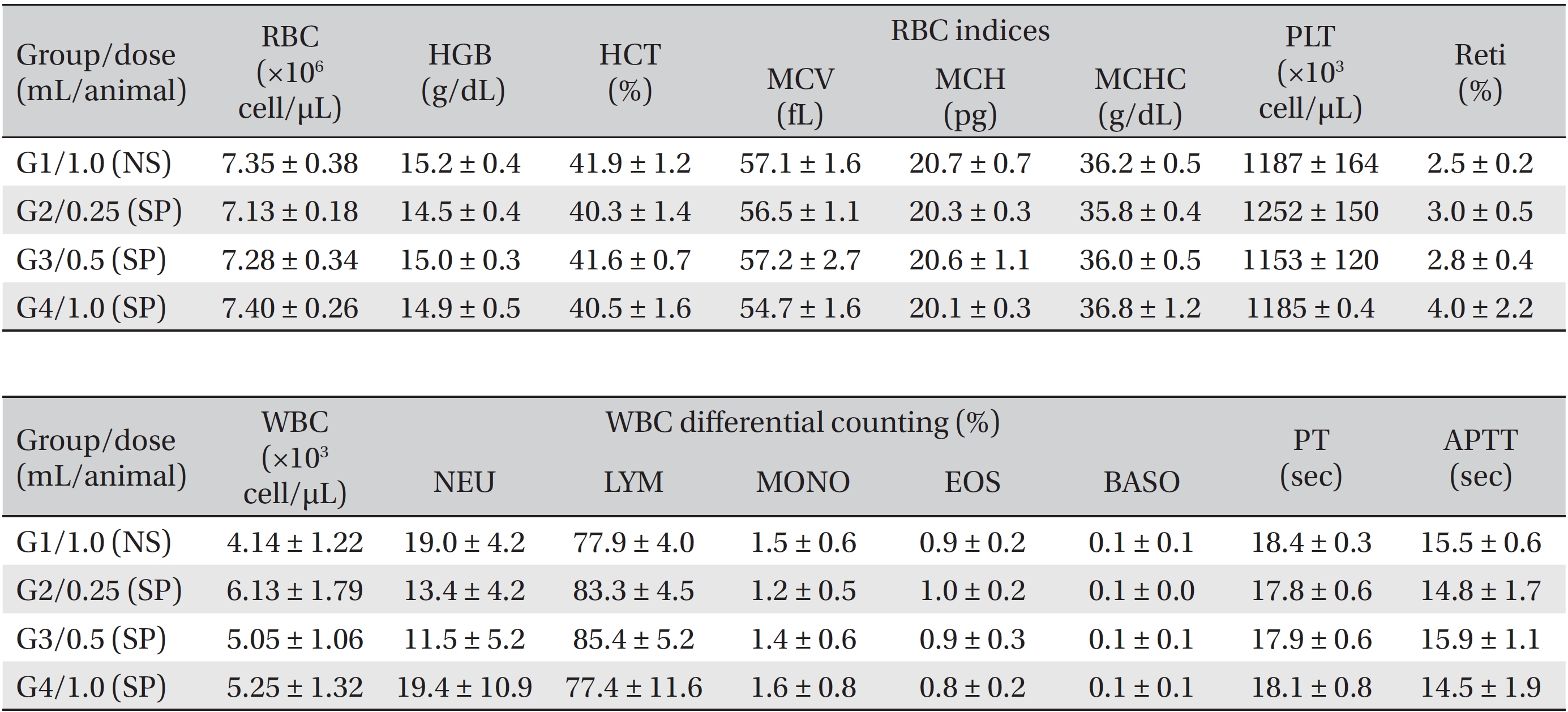
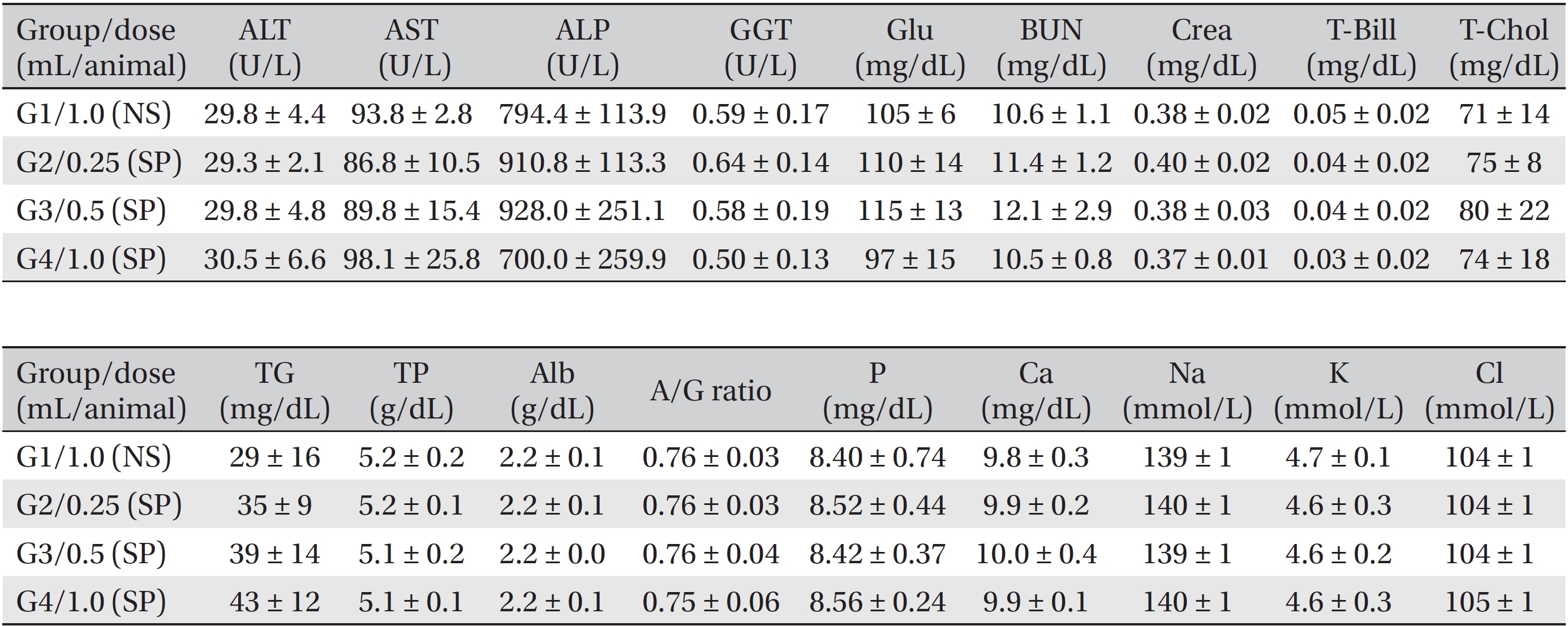
During the observation period, no mortalities or abnormalities of clinical signs were found neither male or female rats in the control or experimental groups. No statistically significant changes in the weights of either male or female rats were observed between the control and the experimental groups (Figs. 1,2). Hematology and blood biochemistry data on both male and female rats in the control and the experimental groups showed no significant changes (Tables 2-5). After necropsy, detailed visual inspections of the organs and tissues of both male and female SD rats showed no abnormal changes (Table 6). Histopathological findings for both male and female SD rats in the control and the experimental groups showed no abnormal changes (Table 7). In local tolerance tests at the injection sites, mild inflammation was observed in two male SD rats in experimental group 3, but the inflammation was found not to be a treatment related effect.
Pharmacopuncture is a treatment by injecting herbal extracts at acupoints [1]. Pharmacopuncture, taking the form of an injection, requires at least two factors for safe use. One is safety in the manufacturing process and the other is the safety of the manufactured products. GMP facilities are required to manufacture injections. At GMP facilities, the biological clean rooms in which the injections are manufactured must be class 1, 10 or 100 clean rooms. All facilities of KPI satisfy Korea Good Manufacturing Practice (KGMP). General equipment of KPI maintains a class 100 zone, and subdivision and filtration equipment maintain a class 10 zone.
Injections manufactured in KGMP facilities follow the toxicity testing standards of the KFDA for a safe use. Pharmacopuncture preparations at KPI are done in KGMP facilities, and toxicity testing for safe use has been continuously performed.
SS for use as eye drops was found to be a non toxic, non irritanting medicine [16]. SS, which consists of
According to the results of a pilot test (Biotoxtech Study No.: B13479P), the clinical dose of SP was an intramuscular injection of 1.0 mL/time. Based on that result, in this study, we divided 40 SD rats into 4 groups of 5 male and 5 female rats per group: 1 control group (NS, 1.0 mL/animal) and 3 experimental groups (SP, 0.25, 0.5, and 1.0 mL/animal) (Table 1). During the experimental period, observed items were mortality, clinical signs, weight changes, hematological and blood biochemistry finding, necropsy findings, histopathological findings.
After SP had been injected, no abnormal changes in the clinical signs, weights, hematological and blood biochemistry results, and necropsy findings were observed. In the histopathological findings for the organs and tissues, except for slight changes, no particular abnormalities were observed. Slight changes were observed sporadically, but they were not dose dependent. Therefore, those changes were found to be accidental changes with no toxicological significance.
As a result, SP in a single-dose intramuscular injection is safe. Now, a repeated dose toxicity test of SP is required to confirm its safety for clinical use.
Under the conditions of this test, the results from the injection of SP suggest that the approximate lethal dose is above 1.0 mL/animal for both male and female SD rats.
[Table. 1] Grouping of animals
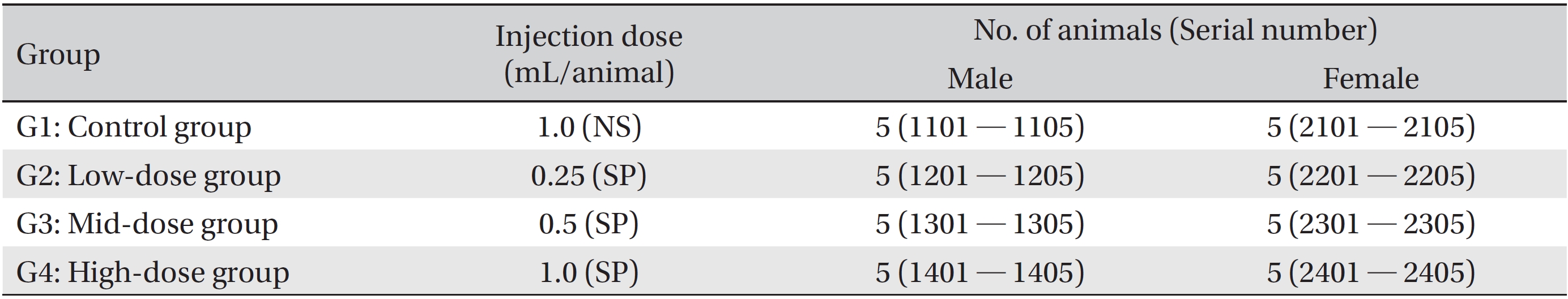
Grouping of animals
[Table. 2] Summary of hematological data (male)
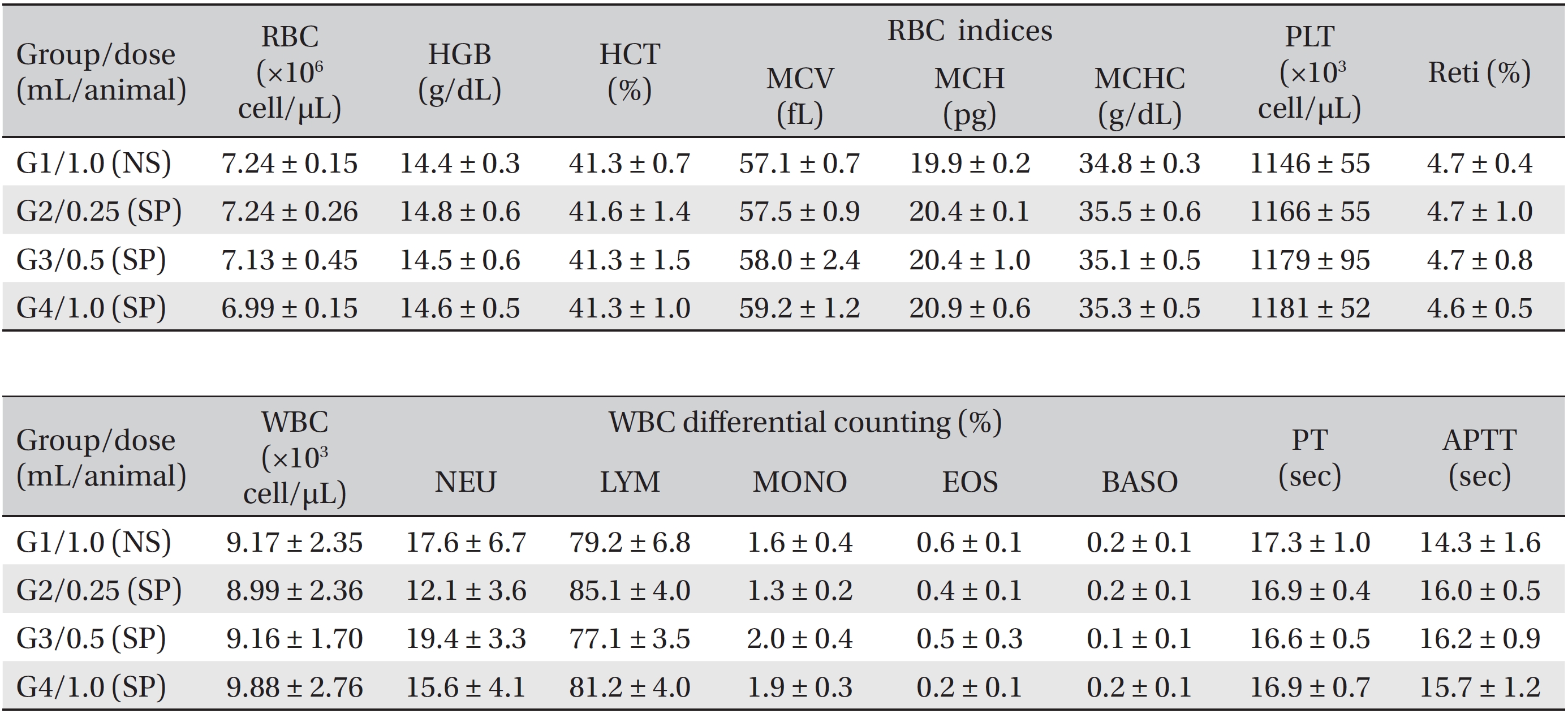
Summary of hematological data (male)
[Table. 3] Summary of hematological data (female)
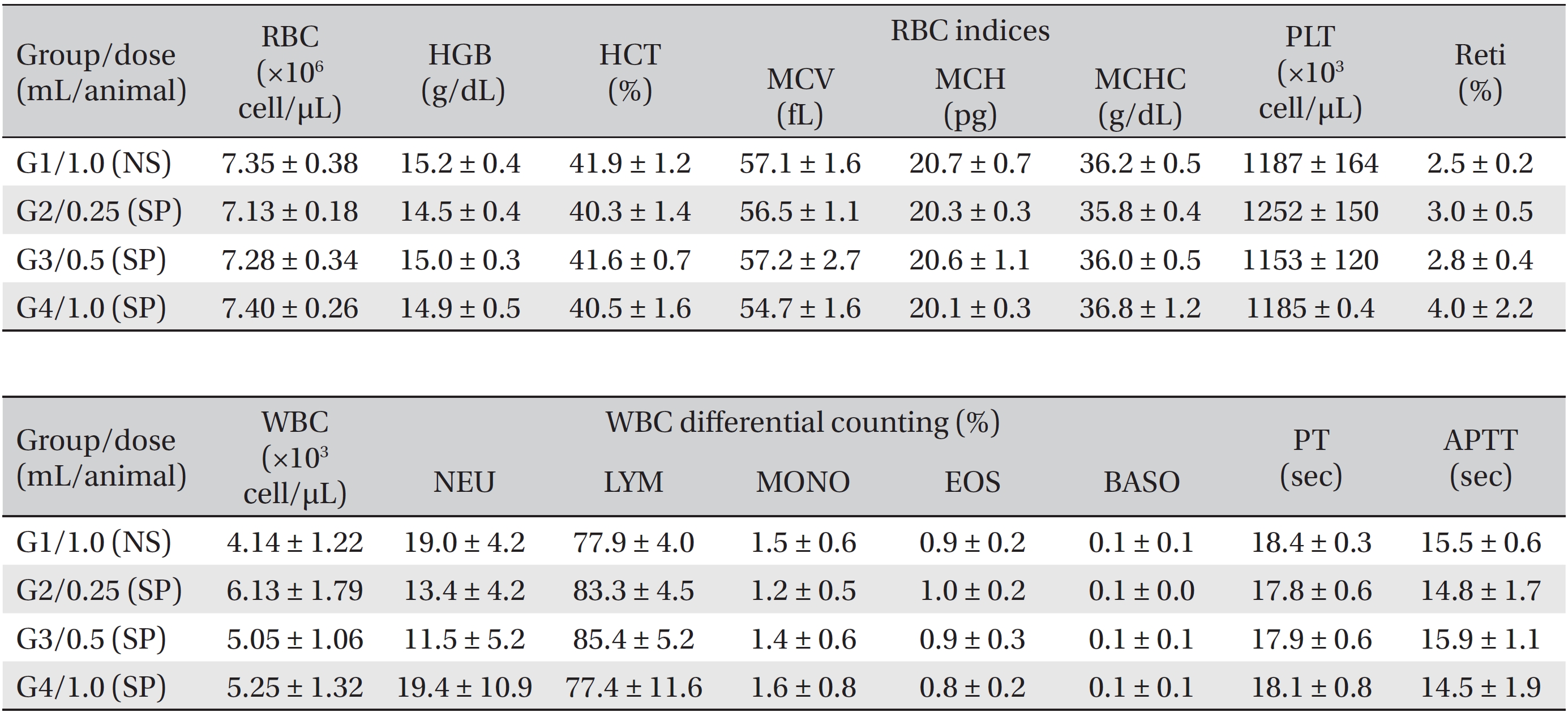
Summary of hematological data (female)
[Table. 4] Summary of blood biochemistry data (male)
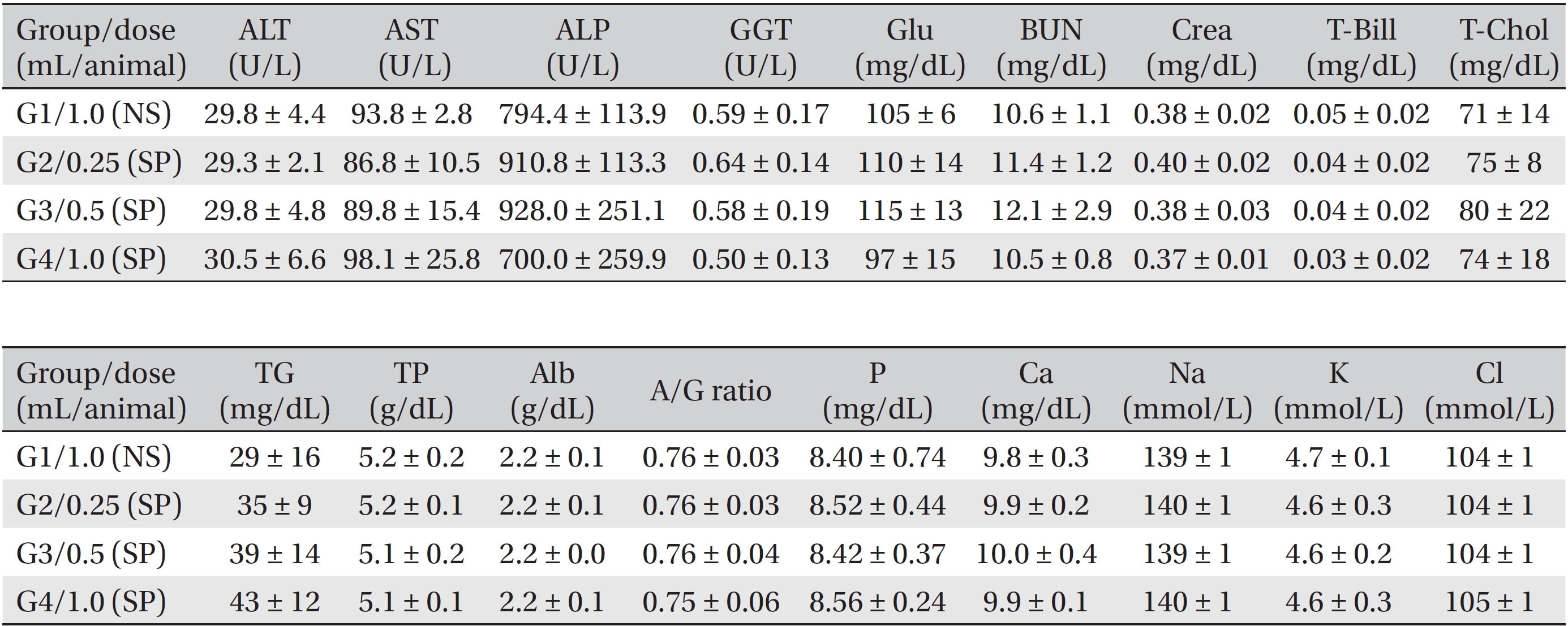
Summary of blood biochemistry data (male)
[Table. 5] Summary of blood biochemistry data (female)
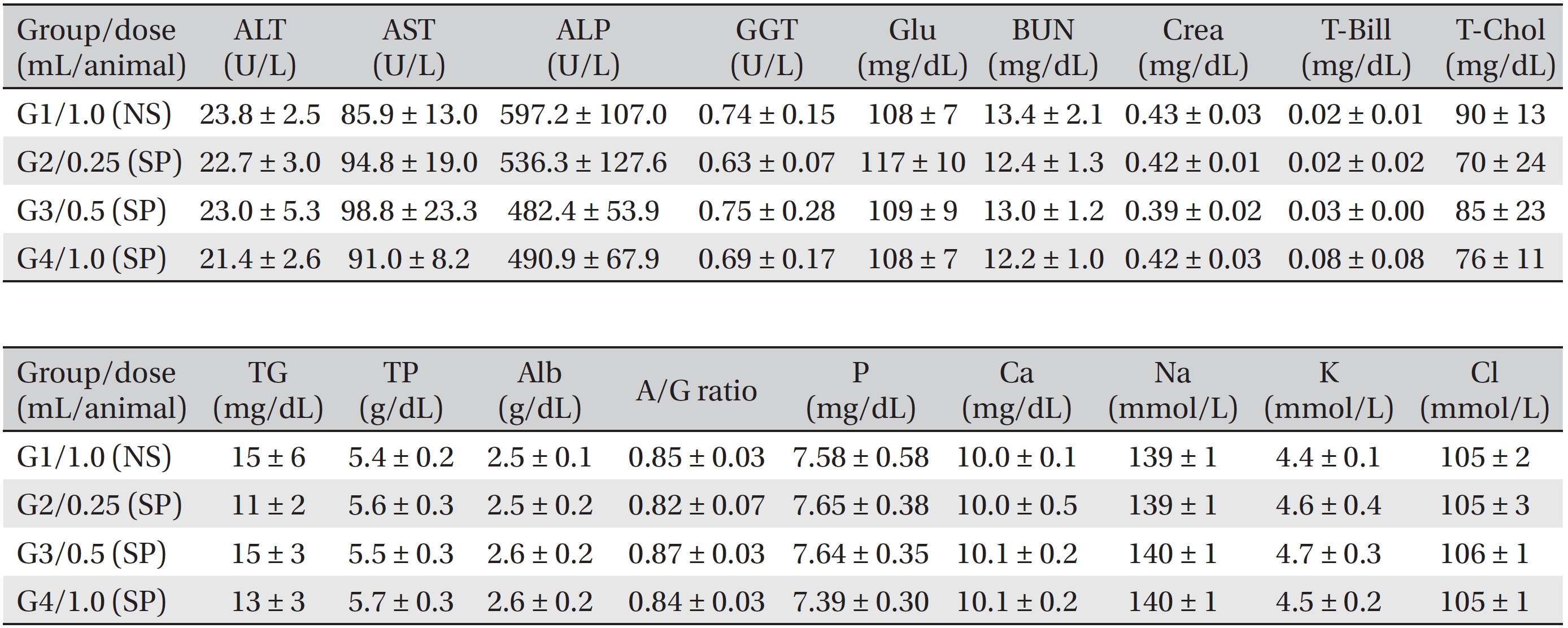
Summary of blood biochemistry data (female)
[Table. 6] Summary of necropsy findings
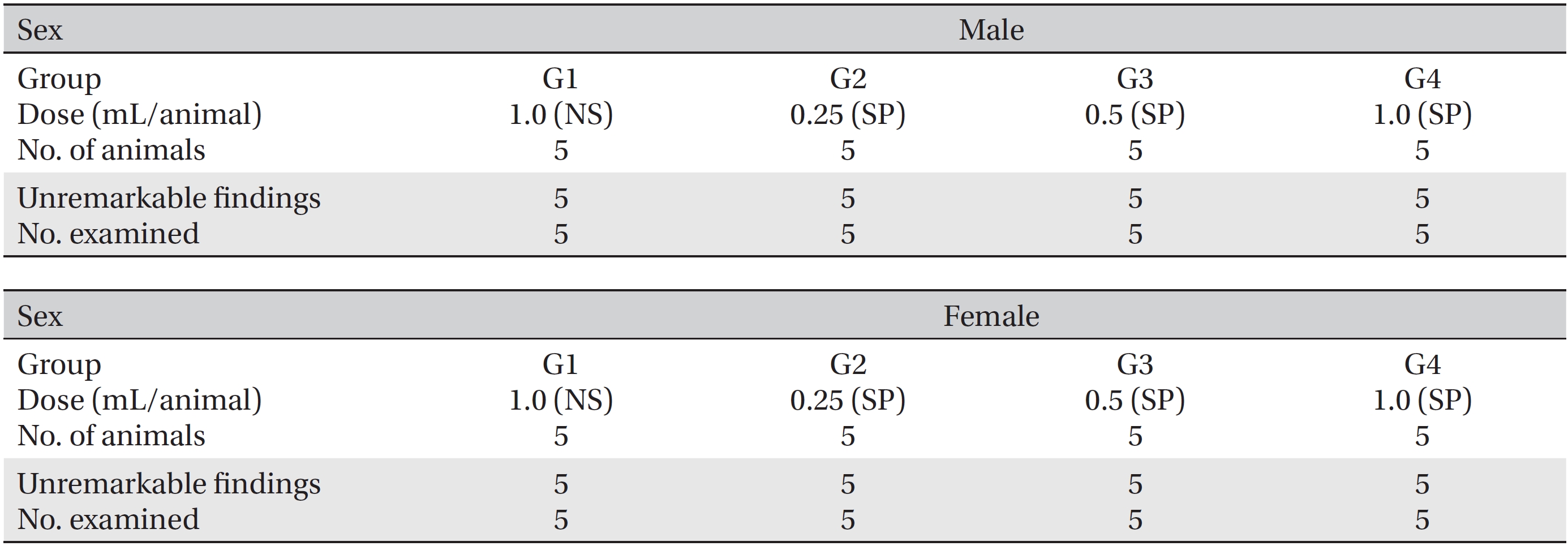
Summary of necropsy findings
[Table. 7] Summary of histopathological findings
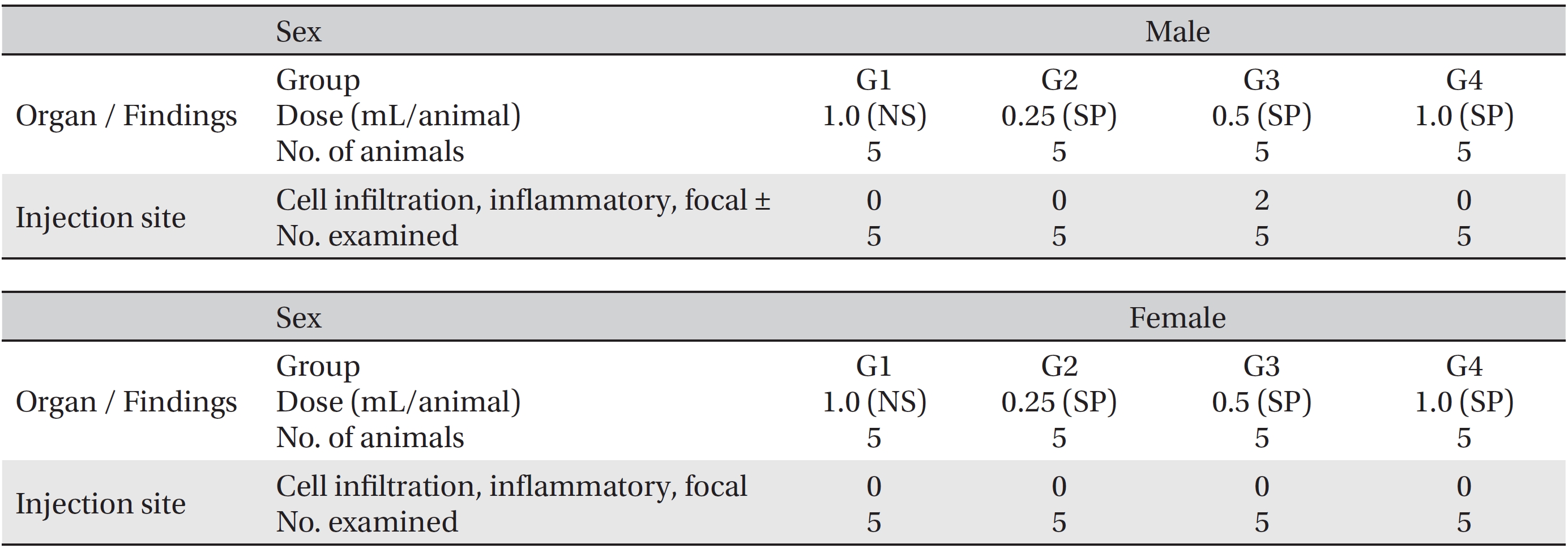
Summary of histopathological findings