Water temperature is an environmental factor in aquaculture that affects the growth and survival of aquatic animals (Wang et al., 2006). Recently, the results of an analysis of observed temperature data by Seong et al. (2010) showed that sea surface temperatures in the East, West, and South seas of Korea were increasing. In fish, changes in temperature can affect their ability to obtain oxygen from the environment and may promote changes at the physiological level that alter the rate of oxygen consumption and its delivery to tissues (Powell and Watts, 2006). For example, from July to early September 2012, there was mass mortality in fishes, particularly the black rockfish
Accumulation of heavy metals in fish can also lead to redox reactions, generating free radicals, and especially ROS. Among them, copper is a pollutant widespread in surface waters at concentrations of up to 100 μg/L (Roy, 1997). Copper sulfate (CuSO4) has been used widely to control algae and some pathogens in fish culture ponds, further increasing copper concentrations in water (Carvalho and Fernandes, 2006). Copper is highly toxic to fish, so the concentrations used to control algae or pathogens must be below the toxicity threshold for fish. Copper toxicity varies with water chemistry, temperature, and fish species, and induces various forms of damage that can lead to the death of the fish (Sanchez et al., 2005). The effects of copper on the aquatic environment are complex and depend on the physiochemical properties of the water (Takasusuki et al., 2004). High temperatures tend to increase the diffusion rate, accelerating chemical reactions and changing the rates of various processes that may become uncoordinated, leading to imbalances (Reynolds and Casterlin, 1980). Thus, a combination of temperature and copper stress is an environmentally relevant situation for populations of intertidal poikilotherms in polluted environments.
At the cellular level, copper has been described as a promoter of oxidative stress, catalyzing the formation of ROS, such as the OH. radical, through the Haber-Weiss reaction, and causing damage to DNA, proteins, and lipids (Matés, 2000; Linde et al., 2005). Cells have a well-developed antioxidant defense system to protect against oxidative stress. Glutathione (GSH)-associated metabolism is a major protective mechanism against agents that cause oxidative stress. GSH participates in detoxification at several levels, and may scavenge free radicals, reduce peroxides, or become conjugated to electrophilic compounds. Several antioxidant enzymes, including superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx), convert ROS into less-noxious compounds. Collectively, these enzymes represent the first line of defense against superoxide and H2O2. However, such enzymes are not 100% effective under normal conditions. Because some of the chemicals generated following reactions between ROS and macromolecules are themselves highly reactive, these secondary oxidation products must also be detoxified to prevent them damaging DNA, proteins, and lipids. This second line of defense against ROS is provided by enzymes such as glutathione-
Thus, in the present study, we sought to evaluate the combined effects of elevated temperature and copper on antioxidant profiles (GSH, GST, GPx, GR, and SOD) in the liver, gill, and kidney of the black rockfish.
>
Experimental animals and water conditions
Black rockfish
[Table 1.] The chemical components of seawater and experimental condition used in the experiments
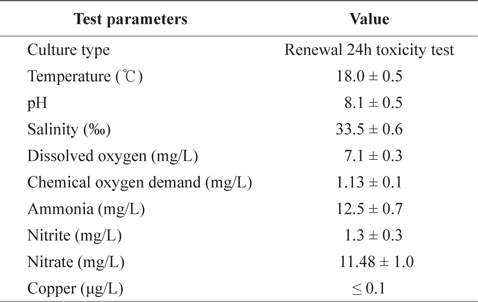
The chemical components of seawater and experimental condition used in the experiments
Prior to the temperature acclimation, fish were held for 4 weeks in aerated tanks containing 18°C seawater to ensure a common thermal history between the acclimation groups. Temperatures were then adjusted from 18°C in 1°C steps on alternate days to final temperatures of 23 or 28°C using a water heater (Electronic thermostat, MS701‑H, Mink, Korea).
Toxicity tests with two replicates (12 fishes at each copper concentration and controls) were carried out in static systems with continuous aeration, at constant temperatures (18, 23, and 28°C) for 96 h in a constant-temperature room (18°C). Experimental fish were exposed to waterborne treatment of 0, 100, and 200 μg/L CuSO4 (copper (II) sulfate, minimum 99%, Sigma, USA) concentrations for 96 h at each temperature. Following the toxicity test, fish were deeply anesthetized using benzocaine, and the liver, gills, and kidney were removed and stored at -80°C until analysis.
Liver, gill, and kidney tissues for determination of enzyme activity were rinsed in 0.1 M KCl (pH 7.4) and homogenized (099CK4424, Glass-Col, Germany) in homogenization buffer (0.1 M K2HPO4, 0.15 M KCl, 1 mM DTT, 1 mM EDTA, 1 mM PMSF). The homogenate was centrifuged (12,000
The total glutathione (GSH) contents of tissues were quantified using the method of Richardson and Murphy (1975). Briefly, the working solution containing 0.01 M 5,5’-dithiobis, 2‑nitrobenzoic acid (DTNB, Sigma, USA) and 0.1 M PBS buffer (pH 8.0) was added to the sample. The GSH level was evaluated at 412 nm using a spectrophotometer (Zenyth 200, Anthos Labtec Instruments GmbH, Austria) and determined using a reduced glutathione standard curve. The GSH level was expressed as nmol GSH/mg protein.
Glutathione
Glutathione peroxidase (GPx) activity was measured by determining the decrease in NADPD (Sigma, USA) during the formation of reduced glutathione via glutathione reductase at 340 nm by a modified method of Bell et al. (1985). In this procedure, 2.5 mM H2O2 in 50 mM potassium phosphate buffer (1 mM GSH, 0.1 M NADPH, 0.5 U/mL glutathione reductase, 1 mM EDTA, 2 mM sodium azide, pH 7.4) was used as the substrate.
Glutathione reductase (GR) activity was determined using NADPH and oxidized glutathione (GSSG) as substrate, as described by Beutler (1984). One unit of GR was defined as the NADPH consumed per min, which catalyzed the reduction of 1 mmol GSSG.
Superoxide dismutase (SOD) activity was measured using a SOD assay kit (Dojindo Co., Japan) to determine the 50% inhibition rate of the reduction of 2-(4-lodophenyl)-3-(4- nitrophenyl)-5-(2,4-disulfophenyl)-
Protein concentrations were determined using the Bradford (1976) method, with bovine serum albumin as the standard.
Data are presented as means ± standard error. For statistical analysis, a one-way analysis of variance (ANOVA) was used, followed by Duncan’s multiple range test. We used the SPSS software (SPSS Inc., Chicago, IL, USA). Differences were considered statistically significant when
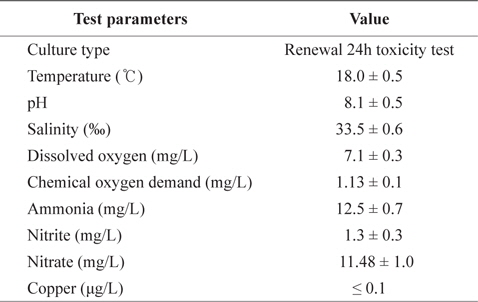
Fig. 1 shows the GSH levels in liver, gills, and kidney of black rockfish
>
Glutathione S-transferase (GST)
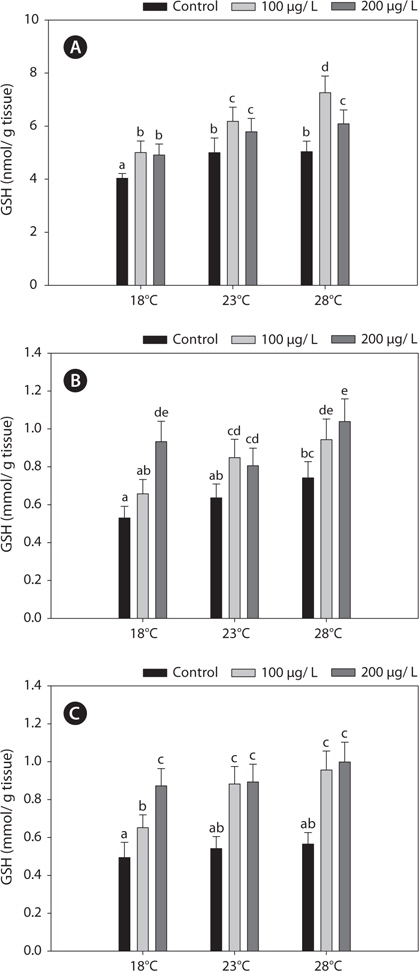
Fig. 2 shows the GST activities in the liver, gills, and kidney of
>
Glutathione peroxidase (GPx)
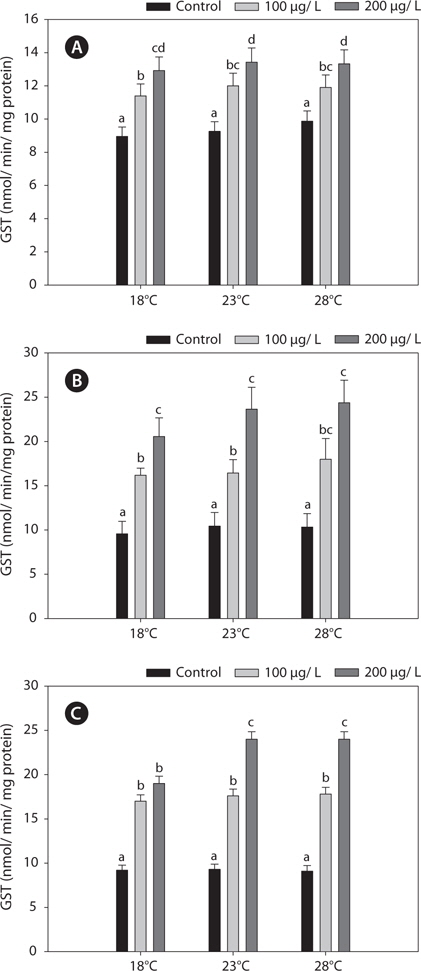
Fig. 3 shows the GPx activities in the liver, gills, and kidney of
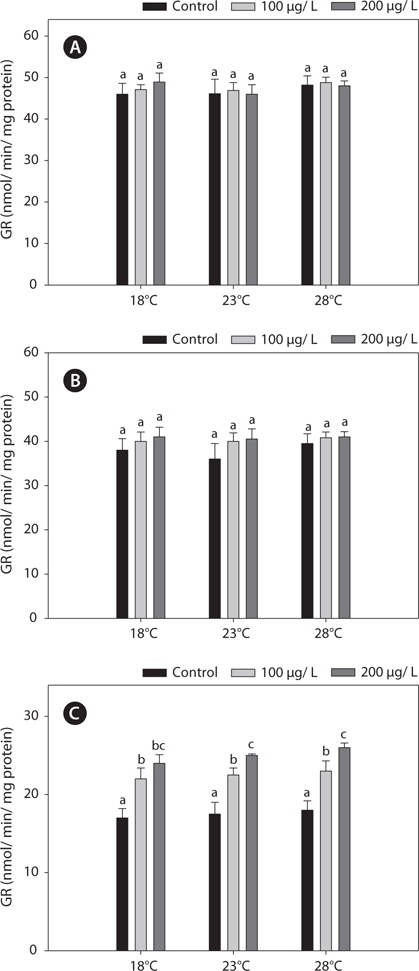
The GR activities in the liver and gill were similar between 18°C and warm (23 and 28°C)-acclimated fish (Fig. 4). Also, the GR activities in the liver and gill of non-exposed and copper-exposed fish did not differ (
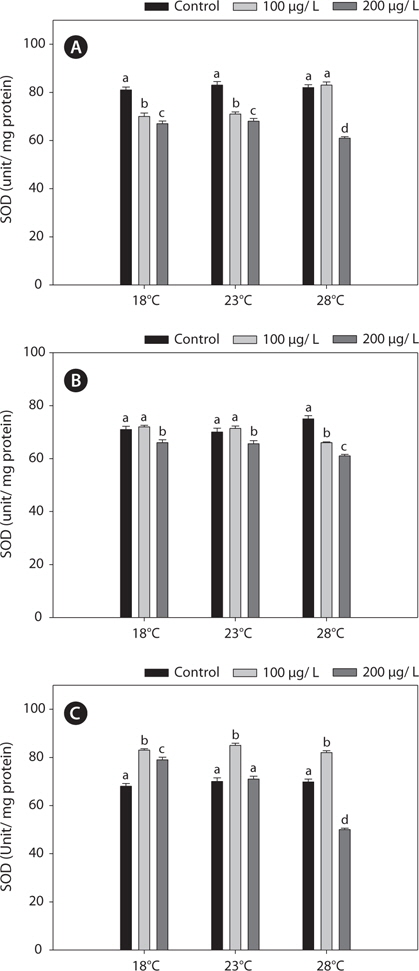
In this study, SOD levels did not differ significantly between 18°C- and warm (23°C and 28°C)-acclimated fish. SOD activities in liver and gills decreased significantly after copper treatment, compared with the non-exposed group, and these reductions were enhanced by acclimation to 28°C (Fig. 5).
Dissolved copper—as CuSO4—at low molecular concentrations has been reported to be toxic to zebrafish
Anti-oxidant enzyme activities are significantly increased in copper-exposed fish, indicating that copper causes oxidative stress in fish (Florence et al., 2002; Sanchez et al. 2005; Varo et al., 2007; Vieira et al., 2009). However, we suggest that after temperature acclimation heavy metals have no toxic effects on the components of the glutathione antioxidant defense system. Indeed, in the present study, we demonstrated the combined effects of thermal and copper stress on five antioxidant enzymes in black rockfish
Acute copper toxicity is believed to result primarily from an imbalance in ion regulation in the gill (Wood, 2001). Temperature affects ion regulation processes in fish and may increase copper toxicity at higher temperatures (Lemus and Chung, 1999). Changes in membrane permeability and increased breathing frequency, which may accelerate copper absorption, are the main reasons for the increased toxicity of copper at higher temperatures. Several mechanisms have been proposed to explain copper-induced cellular toxicity. Most commonly, the basis of these theories is the propensity of free copper ions to participate in the formation of ROS. Copper ions participate in oxidation and reduction reactions. In the presence of superoxide or reducing agents, such as GSH, Cu2+ can be reduced to Cu+, which is capable of catalyzing the formation of hydroxyl radicals from hydrogen peroxide via the Haber-Weiss reaction (Kadiiska et al., 1993; Bremner, 1998).
Endogenous GSH plays a role in cells as an antioxidant, as a co-factor of GPx, and participates in the reduction of peroxides, with concomitant formation of oxidized glutathione disulfide (GSSG). Under normal physiological conditions, GSSG is reduced to GSH by GR at the expense of reduced NADPH, thereby forming a redox cycle (Parke and Piotrowski, 1996). GSH is central to the detoxification of ROS, but in the absence of an enzyme system to catalyze the multiple detoxification reactions, GSH cannot function as an intracellular antioxidant. The production of GSSG by the reduction of GPx or as a consequence of free-radical scavenging is potentially highly cytotoxic. Subsequently, GSSG is reduced by GR, using NADPH as a reductant, or exported by the multidrug resistance-associated protein (Dringer, 2000). Accordingly, the activity of GR represents one of the most important determinants of cellular protection against oxidative stress (Hayes and McLellan, 1999). In this regard, our study showed that both increased GSH and GR and decreased GPx activities are of importance in protecting against copper-induced oxidative stress. Furthermore, fluctuations in GSH activities in organisms exposed to metals are generally accompanied by variations in GST, which conjugates GSH to various xenobiotic compounds (Sheweita et al., 1998). Also, the S-conjugation reaction by GSH is accelerated significantly by GST (Strange et al., 2000). These findings support the results of our study; i.e., the interactions between GSH and glutathione-related enzyme activities through redox cycles in the black rockfish. Specifically, changes in the activities of GPx, GR, and GST can alter the GSH concentration.
In this study, copper alters the activities of these enzymes, resulting in modulated antioxidant capacity. Also, this effect of copper was particularly pronounced at elevated temperatures for 96 h. In detail, GST and GR activities were significantly higher in copper-exposed fish than control, non-exposed fish (Figs. 2, 4), and remained elevated even upon exposure to copper, with the exception of GR activity, in which no significant difference was observed between fish acclimated to temperatures of ≥18°C.
Regarding the basis of the enzymatic activities in the liver and kidney, the increases in the antioxidant capacity in copperexposed fish were probably linked to the increased metabolic activity in warmer temperatures. This activation was related to an adaptive mechanism against stress (Schreck, 1981). In this study, while copper modulated other antioxidant enzyme activities in liver and gills, it had no effect on GR activity in the liver or gill. However, kidney GR activities were increased significantly, in a copper-concentration-dependent manner (Fig. 4). This discrepancy between the liver and kidney antioxidant capacities highlights the central position of the liver in the antioxidant responses of fish. Although copper is an essential micronutrient, it can accumulate and overload hepatocytes, causing liver injury (Pourahmad and O’Brien, 2000).
Kaur et al. (2005) showed that 3-h heat treatment (from 20 to 32°C) of
In the present study, GST and GR activities were increased, and that of SOD was decreased, by copper in the liver, gill, and kidney, regardless of the water temperature (Figs. 2, 4, 5). Although acclimation to warm conditions had no effect on GST, GR, and SOD activities, both GSH and GPx were affected by temperature (Figs. 1, 3). Grim et al. (2013) suggested that because activities of glutathione-dependent enzymes were unaffected by temperature acclimation, the protein levels of glutathione-dependent antioxidants may limit the activity of the enzymatic system at lower temperatures. Similarly, in other studies, SOD and CAT activities were unchanged in fishes following temperature acclimation (Leggatt et al., 2007; Grim et al., 2010). Furthermore, there is no consensus as to whether changing body temperature impacts oxidative stress in fishes (Grim et al., 2010; Kammer et al., 2011; Mueller et al., 2012). However, Cherkasov et al. (2007) reported that Cd-induced oxidative stress in oyster mitochondria was particularly pronounced at 30°C versus 20°C. They suggested that antioxidants conferred adequate protection against the Cd-induced increase in ROS production at 20°C. In contrast, at 30°C, antioxidant systems appeared to be incapable of coping with Cd-induced ROS generation, and considerable oxidative damage resulted. Lanning et al. (2006) also suggested that elevated ROS production in response to Cd might be the mechanism underlying the increased lipid peroxidation at 24 compared to 28°C. These findings are consistent with the results of the present study, in that exposure of black rockfish to copper resulted in significant modulation of GSH and GPx activities at elevated temperatures (23 and 28°C). This study also supports the notion that elevated temperatures can exacerbate copper-induced oxidative stress in the black rockfish, and emphasizes the importance of the thermal context when assessing metal toxicity.
In conclusion, our data indicate that a higher water temperature may increase the toxicity of copper, so that antioxidant defenses in various tissues become overwhelmed, leading to elevated oxidative stress. This provides another mechanism for the synergistic effects between environmental temperature and pollutants, which may have important implications for the survival of fish in polluted environments during seasonal warming and/or the temperature increase caused by global climate change.