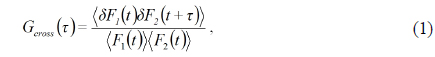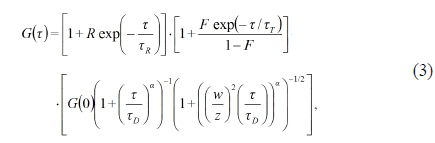Fluorescence correlation spectroscopy (FCS) was developed to study molecular diffusion dynamics and the photochemical properties of various fluorescent probes. FCS is based on the analysis of time-dependent fluorescence fluctuations from diffusing fluorescent probes. Since the late 1990s FCS has progressed due to advances in confocal systems and correlators, and due to the development of detectors with high quantum efficiency. It is well known that the diffusivity of a small particle in solution depends on the size of the particle and the viscosity of the solution. In most studies employing FCS, the relative size of the particle and viscosity of the sample solution have been determined using the Stokes-Einstein equation, based on the diffusion coefficient of a well-known probe in water [1-5]. These studies were unable to confirm particle shape, but the translational diffusion of fluorescent probes has often been investigated assuming that particles are spherical [6, 7].
The timescale of rotational diffusion for nanometer-sized probes introduces technical challenges, because the detectable time ranges of the correlator become expanded. However, photon antibunching and triplet formation occur on a timescale similar to that of rotational diffusion [8]. Fortunately, larger fluorescent probes (nanometer- or submicrometer-sized fluorescent particles such as quantum dots (Q-dots) and Q-rods) have different time scales for antibunching and lifetime, and they have no triplet state to complicate the analysis. Quantum particles are useful as probes due to their broad absorption bands, narrow emission bands, and high quantum yields [9, 10]. In particular, Q-rods of well-defined size and shape can be used as a standard to verify experimentally the diffusion properties of various rodlike molecules and particles.
In this study we built a simple confocal-microscope-based polarization FCS system using CW-laser excitation to investigate the rotational and translational diffusion of CdSe/CdS quantum rods. By autocorrelation analyses of various combinations of polarization and viscosity, we characterized the dependence of the rotational and translational diffusion of Q-rods on polarization by using the amplitude and characteristic time of the correlation function [11].
In a confocal microscope system, the movements of fluorescent particles into and out of the focal volume cause the detected fluorescence to vary in intensity, and this intensity variation inherently contains information about the size, number, shape, and speed of the particles. It is difficult to confirm a large number of total-time series data varying over less than a few milliseconds, so instead these data may be analyzed via the following correlation function:
where
This equation can be expanded as follows:
where
The third term of Eq. (3) has been well established by various experiments showing that the parameters of the translational diffusion term carry quantitative information about the average particle number contained in the focal volume during
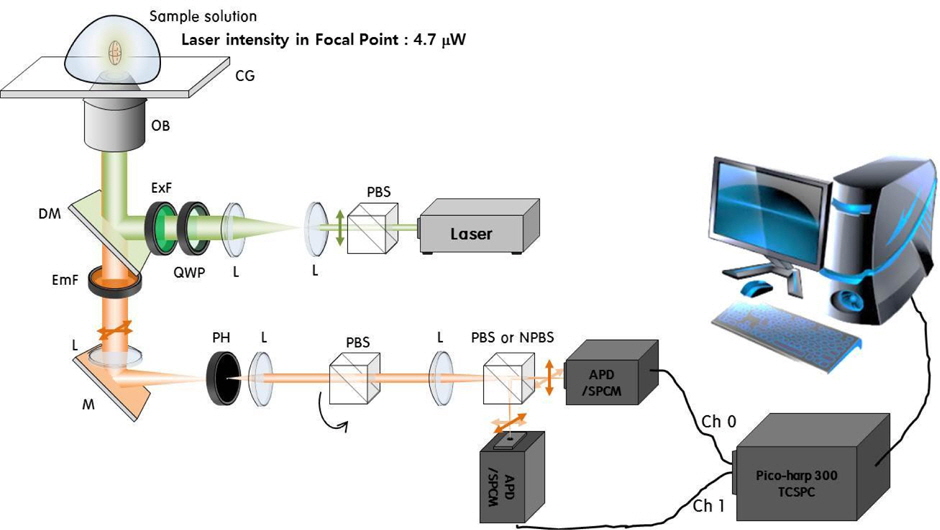
The FCS setup is illustrated schematically in Fig. 1. A 532-nm DPSS laser (Cobolt, Samba; 25 mW) and a single-photon counting module (SPCM; Id Quantique, id100-50) were used as the excitation source and detector respectively. Alignment of the optical components was accomplished using a commercial optical microscope (Olympus IX71). The optical components in the FCS setup were a polarization beam splitter (PBS), nonpolarized beam splitter (NPBS), excitation filter (ExF), dichroic mirror (DM), objective lens (OB), cover glass (CG), emission filter (EmF), lens (L), mirror (M), and pinhole (PH).
In this setup, the laser beam from the light source passes through the PBS and enters the optical microscope. The beam then passes through the ExF (Semrock, FF01-531/40-25) and reflects off the DM (Semrock, FF545/650-Di02). The reflected beam passes through the OB (Olympus, 60×, 1.2 NA), the immersion water, and the CG, and then focuses on a very small volume of a few fL inside the solution. The fluorescent particles in the focal volume are excited by the laser beam and then emit fluorescent light; a portion of this light enters the OB. After the light passes through the DM, EmF, and L, it escapes the microscope by the inner mirror. The light emerging from the microscope passes the PH, which is 30 μm in diameter, and the PBS. The PH and PBS are used to make a confocal conjugate point for the confocal volume, and to pass only the polarized fluorescence light, respectively. The light is split into two beams by the NPBS, and these beams are detected by two SPCMs. The fluorescent light detected at the SPCMs is converted into TTL signals, which are then converted into a correlation function by using a time-correlated single-photon counting system (PicoQuant, Picoharp300). The correlation function is monitored as the cross-correlation function of the responses of the two SPCSMs, using the FluoFit program (PicoQuant) [16], the delay time of the correlation being limited to the range from 0.15 μs to 0.1 s.
3.2. Preparation of Probe Sample
To investigate the rotational diffusion of rodlike particles, we used Q-rods (Q-rod591) of length 35 nm and width 5 nm (aspect ratio of 7) as the fluorescent particles. SEM analysis showed that the Q-rods were of uniform high quality, and TEM analysis showed that they were of uniform size (Fig. 2). The Q-rods were of a core/shell configuration, with CdSe cores and CdS shells. Q-rod growth is described in detail in Ref. [17]. The Q-rods had a maximum emission peak at 591 nm and could be sufficiently excited by the 532-nm laser beam.
To characterize the rotational diffusion of the Q-rod591 particles, we diluted them to a concentration of a few nM. At this concentration the number of Q-rods in the effective focal volume of our system would be less than 10. Therefore, we prepared samples of two different concentrations such that the average particle number in the focal volume would be approximately 4 (the “high-concentration” solution) or 0.5 (the “low-concentration” solution). In addition, Q-rod solutions with various viscosities were prepared, to study the dependence of rotational diffusion on viscosity, and to allow a comparison of that result to the effect of translational diffusion on viscosity. Glycerin was used to adjust the viscosity of the solutions with volume percentages of 0, 10, 20, 30, 40, and 50%; the corresponding viscosities of these solutions at 23 ℃ were 0.9, 1.2, 1.7, 2.6, 4.0, and 6.9 cP, as calculated using a formula for the viscosity of a glycerin-water mixture [18].
3.3. Combination of Polarization States
To examine the dependence of excitation and fluorescence on polarization states, two linear polarizers were used, one near the laser and one between the PH and the NPBS. The fluorescent light was split into two beams for cross correlation. The fluorescent light split by the NPBS was polarized into two states,
3.4. Effective Focal Volume of FCS System
Before analyzing the diffusion of Q-rods, we confirmed the effective focal volume of the FCS system based on polarization states using a rhodamine 6G (R6G) solution of about 10 nM concentration. At our laser intensity the correlation curve of the R6G solution revealed a triplet state and a translational diffusion state. Thus, the curve was fitted by Eq. (3) assuming the triplet state and linear translational diffusion (
To investigate the polarization dependence of the rotational diffusion of Q-rods, we built the polarization FCS system and measured correlation functions of the Q-rods’ fluorescence responses. Measured functions were fitted by Eq. (3) to determine parameters related to the rotational and translational diffusion of the Q-rods. In Eq. (3) we neglected the triplet state because it does not appear in semiconductors such as Q-dots and Q-rods [19]. However, because the diffusion of Q-rods cannot be expected to be linear like that of Rhodamine 6G, due to the small differences in particle sizes and the irregular blinking in the full duration of the experiments, the anomalous diffusion exponent α was fitted by Eq. (3) using other parameters.
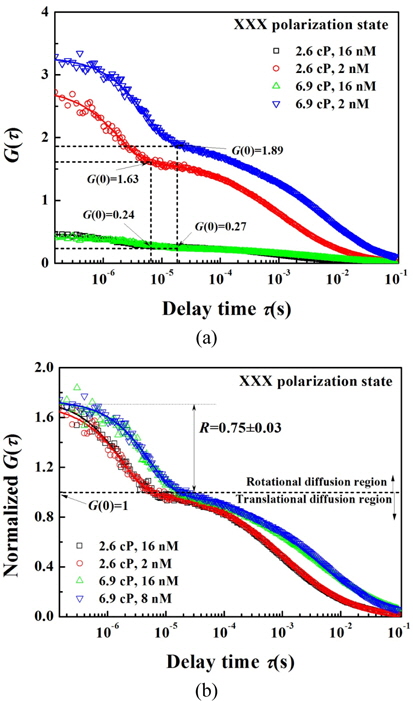
Before analyzing the influence of polarization, we examined the change in correlation function caused by varying the Q-rod concentration. Figure 3 shows the correlation functions obtained from Q-rod solutions of two different concentrations and the two solvent viscosities of 2.6 and 6.9 cP, in the
As shown in Fig. 3(a), in samples of concentration 2 nM the
According to the results in Fig. 3(a), the correlation functions obtained based on measurements of the Q-rod solutions were affected by the solutions’ concentration and viscosity. As mentioned previously, the goal in this study is to investigate the rotational diffusion of Q-rods in combination with their polarization states. To carry this out, the influences of the concentration and the viscosity in the translational diffusion of the correlation function were eliminated by using a normalized correlation function. The normalization was achieved by dividing
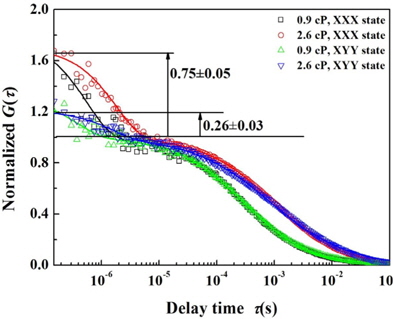
To analyze the rotational diffusion of Q-rods in combination with the polarization state, we measured and normalized the correlation function using viscosities of 0.9, 1.2, 1.73, 2.57, 4.06, and 6.9 cP and the polarization states
Figure 4 illustrates the normalized
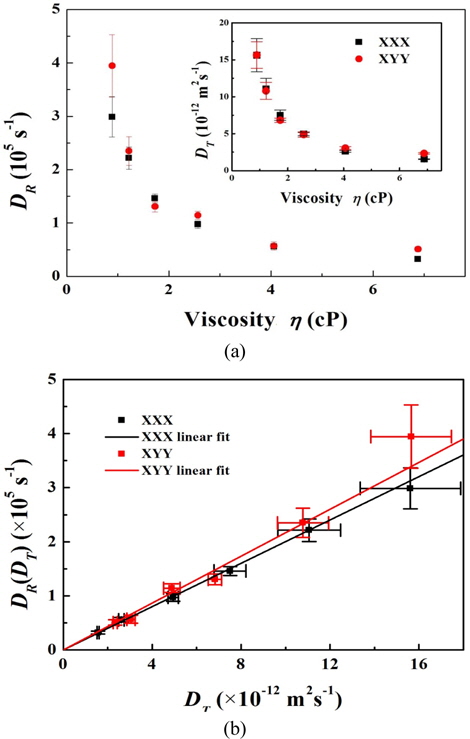
The rotational and translational diffusion coefficients were calculated by Eq. (4) using the diffusion time; the results are shown in Fig. 5(a) and Table 1. The results agree with the trend in diffusion time. As shown in Fig. 5a, the translational and rotational diffusion coefficients in 0.9-cP samples were (1.6±0.2)×10-11 m2 s-1 and (2.9±0.4)×105 s-1 for the
[TABLE 1.] Translational and rotational diffusion coefficients for various viscosities

Translational and rotational diffusion coefficients for various viscosities
The relationship between
We built a polarized fluorescence correlation spectroscopy system based on a confocal microscope, and measured and analyzed the correlation functions of CdSe/CdS Q-rods in solution for various concentrations and viscosities, and for two polarization states. In our analysis of the correlation functions with respect to polarization states, translational diffusion did not depend on the polarization state, whereas rotational diffusion depended strongly on polarization. The rotational fraction was 0.75±0.05 in the