Acne vulgaris is a common skin disease affecting children and adolescents. The pathogenesis of acne is multifactorial and complex. There are four important factors that cause acne in humans, such as an increase in sebum secretion, keratinization of the follicle, bacteria, and inflammation (Farrar and Ingham 2004). Propionibacterium acnes, Staphylococcus epidermidis, S. aureus, and Pseudomonas aeruginosa are often involved in the development of abnormal follicular keratinization and inflammation (Yamaguchi et al. 2009). P. acnes and S. epidermidis have been recognized as pus-forming organisms that trigger inflammation in acne. Especially, P. acnes, one of the commonly isolated skin organisms, induces an inflammation of the sebaceous glands in human face, neck, chest or back (Park et al. 2004). The currently available therapeutic option for acne (antibiotic treatment) is usually used to treat acne vulgaris to inhibit inflammation or kill the bacteria. In the case of antibiotic therapy, triclosan, benzoyl peroxide, azelaic acid, retinoid, tetracycline, erythromycin, macrolide, and clindamycin are the most commonly prescribed (Gollnick et al. 2003, Ravenscroft 2005, Han et al. 2010). However, these antibiotics are often associated with several side effects, such as the emergence of resistant bacteria, organ damage, and immune hypersensitivity if these medicines are used for a long period (Kim et al. 2008). Therefore, many researchers have tried to develop targeted therapeutic agents with no side effects and high antibacterial activity.
To overcome the problem of side effects, medicinal plants and marine organisms have been investigated for the treatment of acne. Therefore, we investigated the possibility that they may be effective acne treatments based on the previously known anti-methicillin-resistant S. aureus effects of phlorotannin isolated from brown alga Eisenia bicyclis. E. bicyclis is a common perennial phaeophyceae (brown alga) and generally inhabits the region of Ulleung Island in the East sea of Korea. This seaweed has been added to appetizers, casseroles, muffins, pilafs, and soups (Maegawa 1990, Yoon et al. 2011). The antioxidant activity of E. bicyclis phlorotannins, such as eckol (a trimer), phlorofucofuroeckol A (a pentamer), dieckol and 8,8′-bieckol (hexamers) have been previously described (Okada et al. 2004). This brown alga has also been reported to exhibit several medicinal functions, such as anti-tumor (Ermakova et al. 2013), anti-Alzheimer’s disease (Ahn et al. 2012), anti-atherosclerosis (Kang et al. 2006), anti-inflammatory (Jung et al. 2013), anti-coagulant activities (Jeong et al. 2009), anti-allergic disease and anti-cancer activities (Shibata et al. 2003, Yoon et al. 2013). In addition, phlorotannins have been known to show potent antimicrobial activity against several microorganisms (Eom et al. 2013).
However, there is no obvious report on the antibacterial activity of phlorotannins from brown alga against acne-related bacteria. Therefore, we demonstrated that phlorotannins isolated from E. bicyclis have high antibacterial effects against acne-related bacteria and may act as alternative and therapeutic agents for acne in this study.
In late September 2010, E. bicyclis was purchased from Ulleung Trading Co. (Ulleung-gun, Korea). A voucher specimen has been deposited in the author’s laboratory. Dried E. bicyclis was finely ground and powdered with a food mixer (HMF-1000A; Hanil Electronics, Seoul, Korea). The dried powder was vacuum-packed and kept at -20°C until use. The dried E. bicyclis powder (1.0 kg) was extracted with methanol (MeOH; 10 L × 3) at 70°C for 3 h (3 times) and the solvent was evaporated in vacuo with a rotary evaporator (N-1001S-W; Eyela, Tokyo, Japan). The crude MeOH extract of E. bicyclis was suspended in 10% MeOH (1.0 L) and then partitioned in turn with n-hexane (Hexane), dichloromethane (DCM), ethyl acetate (EtOAc), and n-butanol (BuOH) in sequence. The concentration of each extract was adjusted to 200 mg mL-1 by dissolving in dimethyl sulfoxide under sterile conditions and stored at -70°C until used.
The following bacterial strains obtained from the Korean Collection for Type Cultures (KCTC; Daejeon, Korea) were used as indicator microorganisms in the study: P. acnes (KCTC 3314), S. aureus (KCTC 1927), S. epidermidis (KCTC 1370), P. aeruginosa (KCTC 1637), which were used for evaluation of anti-acne-related bacterial effect. Two strains of P. acnes clinical isolates were provided by the Gyeongsang National University Hospital (Jinju, Korea), a member of the National Biobank of Korea.
P. acnes strains were anaerobically cultivated in brain heart infusion broth (BHI; Difco Inc., Detroit, MI, USA) supplemented with 1.0% glucose, and incubated at 37°C for 24 h in a CO2 incubator (NAPCO 5400; General Laboratory Supply, Pasadena, TX, USA), in a 10% CO2 humidified atmosphere. As for determining the growth curves of bacterial cells under optimal growth conditions, cultures of P. acnes were diluted and plated on BHI-agar. The plates were incubated at 37°C for 24 h in a 10% CO2 incubator and the number of colony-forming units (CFU) was determined. S. aureus, S. epidermidis, and P. aeruginosa were grown aerobically at 37°C in tryptic soy broth (TSB; Difco Inc.). The disk diffusion assay was prepared in Mueller-Hinton agar (MHA; Difco Inc.) and the broth dilution method was carried out in Mueller-Hinton broth (MHB; Difco Inc.) according to the Clinical and Laboratory Standards Institute (CLSI) guidelines (formerly National Committee for Clinical Laboratory Standards [NCCLS]).
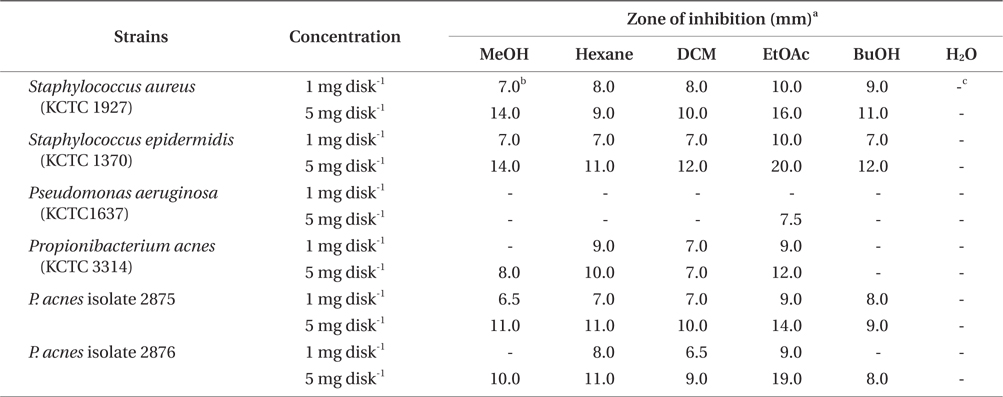
The antibacterial efficacy was evaluated by disk diffusion assay described by the CLSI (2009). In brief, bacterial strains were cultured in TSB at 37°C until cells reached at an OD 600 nm of 0.5. One hundred microliter of bacterial culture containing approximately 104-105 CFU mL-1 was spread on MHA agar plates. A paper disc (6 mm in diameter) containing 1 mg and 5 mg of each extract was placed in the above MHA plate. After incubating for 24 h at 37°C, the diameter of the inhibition zone was measured on bacterial culture plates. The experiment was carried out three times and the mean values were presented.
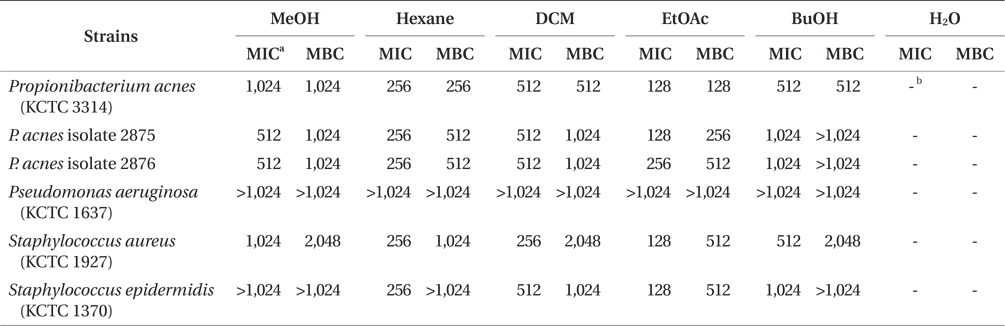
The concentration of MeOH extract and its solvent fractions of E. bicyclis was 200 mg mL-1. Each extracts was diluted with MHB to obtain a stock solution of 2,048 μg mL-1. To determine the MIC values of the MeOH extract and its solvent fractions of E. bicyclis, a stock solution of extracts was prepared in microbial culture medium for each microbial species. The MICs are the lowest concentration of MeOH extracts and its solvent fraction of E. bicyclis to inhibit the visible growth of microorganisms after overnight incubation using MHB, which was modified from the methods described for antimicrobial susceptibility testing by the CLSI (2006). MIC was defined as the lowest concentration of crude extract that inhibited the visual growth after incubating the aerobic bacteria for 18 h and the anaerobic bacteria for 48 h. MICs of the solvent-soluble extracts were determined by the two-fold serial dilution method in 96-well flat-bottomed microtitration plates at final concentration of 7 × 105 CFU mL-1. The microtitration plates were read visually and the MIC of the extracts that exhibited no turbidity was recorded as the MIC. For MBC testing, an aliquot of inoculum was taken with a MIC test well that did not show turbidity, and was poured onto nutrient agar (Difco Inc.) plates for each bacterial species. The agar plates were incubated for 2 days for aerobic bacteria and 5 days for anaerobic bacteria. The MBC value was read as the lowest concentration of the solvent-soluble extracts at which 99.99% or more of the initial inoculum was killed. The MIC and MBC experiments were repeated in triplicate.
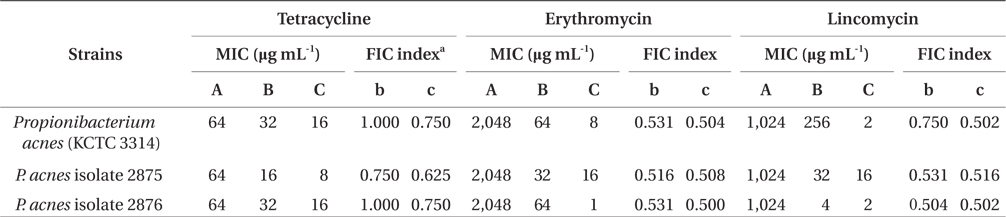
The interaction between FF and antibiotics including tetracycline, erythromycin, and lincomycin (Sigma Chemical Co., St. Louis, MO, USA) against P. acnes was tested by the checkerboard method (Weig and Müller 2001, Perea et al. 2002). The synergistic effect was evaluated as a fractional inhibitory concentration (FIC) index. With the checkerboard test, the FIC was calculated as the MIC values of an antibiotic or FF in combination divided by the MIC of the antibiotic or FF alone. The FIC was then summed to derive the FIC index, which indicated synergy when index values were determined using the following formulae:
FICA = MICA in combination / MICA,FICB = MICB in combination / MICB,FIC index = FICA + FICB
The interaction was defined as synergistic if the FIC index was <1, additive if the FIC index was 1.0, subadditive if the FIC index was between 1.0 and 2.0, indifferent if the FIC index was 2, and antagonistic if the FIC index >2. Synergy was further subclassified as marked (FIC index, ≤0.50) and weak (FIC index, between 0.50 and 1.0).
In all cases analyses were performed in triplicate and data were averaged over the three measurements. The standard deviation (SD) was also calculated. Significance of differences between average MICs for each individual microorganism were determined by Student’s t test at the 95% significance level using SPSS version 12.0 (SPSS Inc., Chicago, IL, USA).
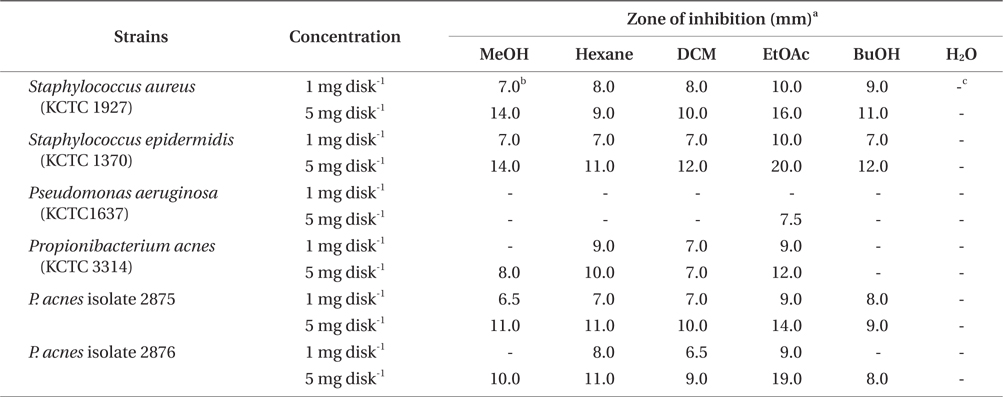
The antibacterial activity of methanol extract and its solvent fractions are presented in Table 1. The MeOH extract of E. bicyclis exhibited an antibacterial activity against acne-related bacteria, suggesting that the extract contains antibacterial substances against acne-related bacteria. For P. acnes, the clear zones of treatment with the EtOAc-soluble fraction had a diameter of 9.0 mm with a concentration of 1 mg per disc and 12.0-19.0 mm with concentrations of 5 mg per disc. For S. aureus and S. epidermidis, the antibacterial activity of the EtOAc-soluble fraction were noted as 1.0 mg per disc (clear zone, 10.0 mm) and 5.0 mg per disc (clear zone, 16.0 and 20.0 mm, respectively). However, the antimicrobial effects on gram-negative P. aeruginosa were less effective than those on gram-positive bacteria in all the tested extract and fractions. Regardless of solvent fractionation, the water-soluble fraction of the MeOH extract did not exhibit the antibacterial activity against all of the bacteria tested.
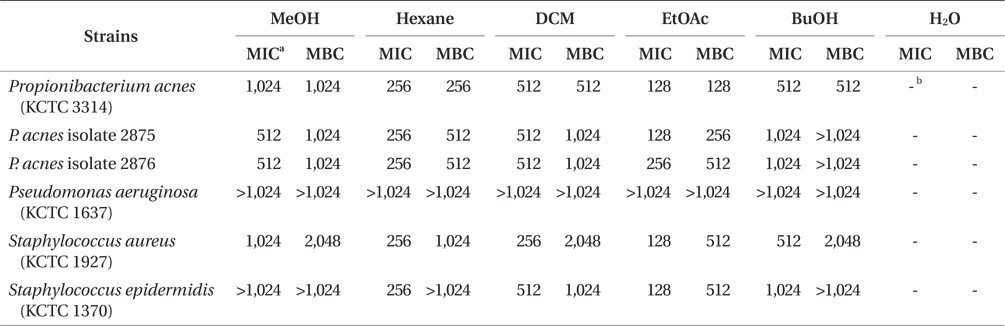
The MIC values of solvent fractions against acne-related bacteria varied depending on the polarity of the solvent. Among solvent-soluble fractions, the EtOAc-soluble fraction showed the lowest MIC values against acne-related bacteria. The EtOAc-soluble extract could completely inhibit the growth of P. acnes strains at 128 and 256 μg mL-1 concentration. The antibacterial activity of the EtOAc-soluble extract against acne-related bacteria was higher than those of other-soluble fractions. Eom et al. (2011) reported that the EtOAc-soluble extract of E. bicyclis exhibited the highest antibacterial activity against methicillin-resistant Staphylococcus aureus (MRSA) and food-pathogen bacteria. The MBC values of EtOAc-soluble fraction against P. acnes strains were determined from 128 to 512 μg mL-1 (Table 2). However, no antibacterial activities of E. bicyclis extracts against P. aeruginosa were observed. It was also observed that the gram-negative bacteria P. aeruginosa showed higher MIC values for all extracts compared to other gram-positive bacteria.
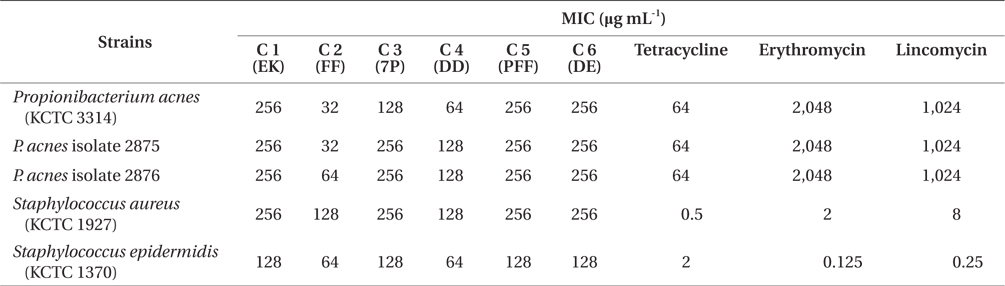
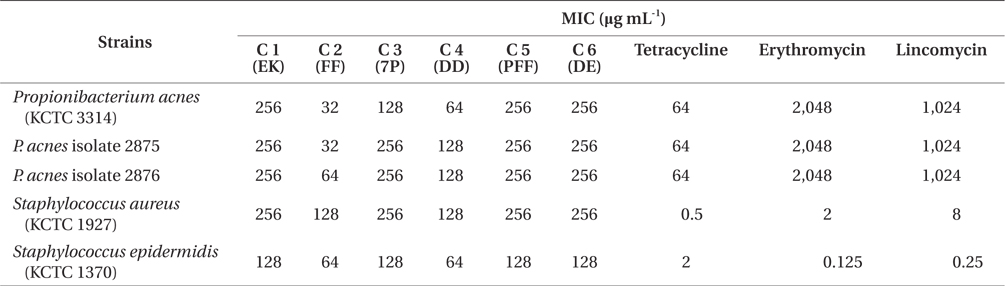
According to above results, the EtOAc-soluble fraction of E. bicyclis showed the strongest antibacterial activity against acne-related bacteria. In order to identify an anti-acne substance from the EtOAc-soluble fractions of E. bicyclis, we screened the antibacterial activity of previously isolated phlorotannins against acne-related bacteria. It is reported that marine-derived polyphenols (phlorotannins) are believed as the active components of E. bicyclis (Eom et al. 2013). Recently, we have reported that successive column chromatographic purification of the EtOAc-soluble extract led to the isolation and characterization of six phloroglucinol derivatives: eckol (1), FF (2), 7-phloroeckol (3), dioxinodehydroeckol (4), phlorofucofuroeckol-A (PFF) (5), and dieckol (6) (Eom et al. 2013). In this report, we demonstrate the antimicrobial activities of these compounds against acne-related bacteria. The MIC values of these phlorotannins were in the range of 32 to 64 μg mL-1 and were at least two-folds less than those of tetracycline, erythromycin and lincomycin against P. acnes (Table 3). Among the isolated compounds, compound 2 (FF) had the highest antibacterial activity against acne-related bacteria (32 to 128 μg mL-1) (Table 3). Other compounds had also similar antibacterial activities with a MIC values from 64 to 256 μg mL-1. P. acnes strains were considered to be highly resistant to antibiotics (tetracycline, erythromycin, and lincomycin) in this study. In particular, P. acnes strains with high-level resistance to erythromycin and lincomycin were detected with the MIC values of 2,048 and 1,024 μg mL-1, respectively. The MIC values of FF against P. acnes (32 to 64 μg mL-1) were equal or lower than that of tetracycline against P. acnes (32 μg mL-1). S. epidermidis and S. aureus were found to be sensitive for tetracycline, erythromycin, and lincomycin (0.125 to 8 μg mL-1).
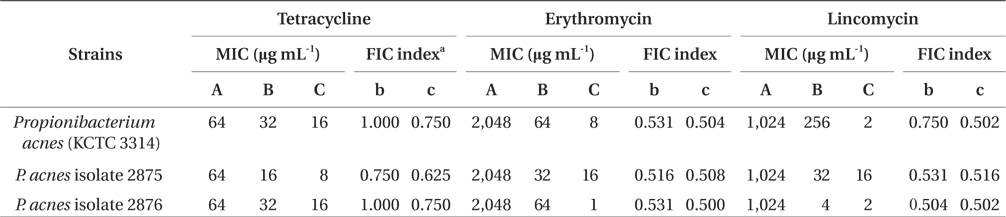
As an alternative way, natural materials such as plantderived or marine-derived compounds in combination with traditional medicines against drug-resistant bacteria may be used as an effective approach for restoration of antibiotic activity (Eom et al. 2013). Taylor et al. (2005) reported that green tea components may display synergy with conventional antibiotics against gram-negative bacteria. In addition, the catechin fraction of green tea acted synergistically with ciprofloxacin in a chronic bacterial prostatitis model in the rat (Lee et al. 2005). Based on these reports, the synergistic effect of marine-derived polyphenol on P. acnes was assessed in combination with commercial antibiotics to treat acne. As shown in Table 4, the MIC values of tetracycline against P. acnes were reduced from 16 to 8 μg mL-1 when administered in combination with 64 μg mL-1 of FF. The MIC values of erythromycin and lincomycin against P. acnes were also dramatically reduced when administered in combination with FF. The FIC indices of antibiotics were in a range from 0.500 to 0.750 in combination with the concentration of FF (64 μg mL-1) against P. acnes strains, thereby indicating the marked or weak synergy effect of FF-antibiotics (tetracycline, erythromycin, and lincomycin) combination, suggesting that FF-tetracycline, FF-erythromycin, and FF-lincomycin synergistically inhibited the growth of P. acnes.
Tetracycline, erythromycin, and lincomycin have been used for decades to treat acne. However, these antibiotics can cause undesirable side effects, including vomiting diarrhea, sore mouth, and skin redness. In addition, antibiotic-resistant P. acnes is a growing problem in many countries due to the overuse of antibiotics (Davies and Davies 2010). Therefore, there is a need to develop new medicines or alternative therapies for acne.
In an effort to decrease usage of antibiotics and discover an alternative therapeutic agent for treating acne infection, we have screened MeOH extract and its soluble extract from a brown alga E. bicyclis to find out anti-acne agents. The relative susceptibility of acne-related bacteria to the potential antimicrobial agent was measured by a clear zone of growth inhibition around the disc. A previous study reported the antimicrobial activities of MeOH extract and its solvent-soluble extract from E. bicyclis against MRSA and food-pathogen bacteria (Eom et al. 2011). The EtOAc-soluble extract showed the highest antibacterial activity against MRSA and S. aureus in disc diffusion (15-24 mm) with the highest total polyphenolic contents (Eom et al. 2011). Choi et al. (2011) reported that the MeOH extracts of brown algae Ecklonia cava, Ecklonia kurome, and Ishige sinicola exhibited antibacterial activity against P. acnes, with 5.3, 5.7, and 6.3 mm at 5 mg per disc, respectively. Thus, E. bicyclis has a similar antibacterial activity against P. acnes when compared with other brown seaweeds.
The present study showed significant correlations between anti-MRSA activity and anti-acne-inducing bacterial activity. In general, gram-negative bacteria are more resistant than gram-positive bacteria to treatment with natural anti-bacterial extracts (Afolayan 2003). The higher resistance of gram-negative bacteria than gram-positive bacteria against different antibacterial compounds is generally attributed to differences in their cell wall and outer membrane (Nikaido 1996, McDonnell and Russell 1999). In this study, the EtOAc-soluble extract of E. bicyclis exhibited a similar antibacterial activity against acne-related bacteria in comparison to those from E. cava, E. kurome, and I. sinicola. Therefore, the MIC values indicated that the anti-acne-related bacteria activity of E. bicyclis was almost equal to the anti-MRSA activity of E. bicyclis.
According to Eom et al. (2011), the antibacterial activities of brown algae are related to their total phenolic contents. Marine-derived polyphenols (phlorotannins) are the predominant EtOAc-soluble compound in brown algae (Choi et al. 2010). Among EtOAc-soluble compound, the polyphenol polymers (eckol, PFF, dieckol, and 8,8′-bieckol) exhibited potent antibacterial activities (Nagayama et al. 2002, Isnansetyo and Kamei 2009). We also reported the isolation of six phloroglucinol derivatives from EtOAc-soluble extract of E. bicyclis with successive column chromatographic purification: eckol (1), FF (2), 7-phloroeckol (3), dioxinodehydroeckol (4), PFF (5), and dieckol (6) (Eom et al. 2013).
Eckol (1), PFF (5), and dieckol (6) have previously been reported to exhibit potential antibacterial activity against MRSA (Lee et al. 2008, Choi et al. 2010, Eom et al. 2013). These results are in accordance with those of our study, which found that eckol-type phlorotannins might possess potential anti-bacterial activities. Although several anti-P. acnes agents such as xanthonoid (α-mangostin, MIC = 1.95 μg mL-1), tannins (terchebulin, MIC = 250 μg mL-1; ellagic acid, MIC = 125 μg mL-1; flavogallonic acid dilactone, MIC = 250 μg mL-1), flavonoid (kaempferol and quercetin, MIC = 32-64 μg mL-1), and terpenoids (rosthornins, MIC = 3.17-25 μg mL-1) (Kubo et al. 2004, Lim et al. 2007, Pothitirat et al. 2010, Muddathir and Mitsunaga 2013) from natural sources have been identified, there has been no scientific report on anti-P. acnes activity of phlorotannins.
From our results, it appears that P. acnes is resistant against antibiotics. Because of its resistance to many commonly used antibiotics, there is a need for searching more effective anti-acne agents. As an alternative method, the FIC test for the combination of isolated phlorotannins and antibiotics, a commonly ineffective antibiotic to P. acnes due to resistance, was assessed using the checkerboard test. It has been previously reported that dieckol from E. stolonifera and PFF from E. bicyclis exhibited the synergistic effect in combination with β-lactam antibiotics against MRSA (Lee et al. 2008, Eom et al. 2013). The results of the checkerboard assay revealed the restoration of antibacterial activity of antibiotics used in this study against the antibiotic-resistant P. acnes in combination with FF. FF (MIC = 64 μg mL-1) can remarkably reduce the MIC values of the antibiotics against P. acnes, suggesting that FF may have potential for use as an adjunct in the treatment of antibiotic-resistant P. acnes. Several studies have previously reported synergistic effects between catechin and β-lactam antibiotics occurred at the cell wall (Zhao et al. 2001, Taylor et al. 2005). Therefore, further investigations are needed to fully elucidate the underlying antimicrobial mechanisms of FF against P. acnes.
In conclusion, the antibacterial activity of the phlorotannins from edible marine brown algae E. bicyclis against anti-acne-related bacteria was evaluated. Since the ethyl acetate fraction showed the strongest antibacterial activity against acne-related bacteria among solvent fractions, antibacterial activity of E. bicyclis extracts against acne-related bacteria may also correlate with their phlorotannins or marine-derived polyphenolic contents. Therefore, previously isolated phlorotannins such as eckol, FF, 7-phloroeckol, dioxinodehydroeckol, PFF, and dieckol were evaluated for antibacterial activity against acne-related bacteria. Among them, FF exhibited the highest antibacterial activity against acne-related bacteria. In addition, FF in combination with other antibiotics is expected to have a therapeutic effect for relieving symptoms against P. acnes with synergy effects. Collectively, phlorotannins can be used as natural antibiotic agents and food supplement as functional ingredients. The results of the present investigation are expected to contribute to the development of an alternative phytotherapeutic agent against antibiotic-resistant P. acnes.
To our knowledge, this is the first report on the antibacterial activity of phlorotannins against acne-related bacteria. The results of the present investigation are expected to contribute to the development of an alternative phytotherapeutic ingredient without any adverse side effects to human body.