Small red algae, especially those previously referred to as ‘primitive’ are often overlooked, but can be quite abundant. These ‘primitive’ red algae are now placed in several classes distinct from the Florideophyceae, for example the Stylonematophyceae. A brownish-red filamentous alga was collected from a sandy tide pool at Cape Tribulation, Queensland, Australia. Cultured specimens were identified as Bangiopsis and conformed to the morphological characters of the genus (multicellular base, erect filaments branched or unbranched, uniseriate to multiseriate-tubular, single multilobed purple-red to red-brown plastid with central pyrenoid, vegetative cells released directly as spores). Molecular data of two plastid genes (rbcL, psbA) support placement of the Australian isolate and isolates from India in Bangiopsis. The genetic variation between these isolates and isolates from Puerto Rico previously attributed to B. subsimplex indicates that these should be considered as a separate species. As the type locality is in the Atlantic Ocean, French Guiana, and not far from Puerto Rico, and the Puerto Rican isolate has been used often in phylogenetic analyses, we propose that the Indian and Pacific Ocean isolates be designated a new species, B. franklynottii, to acknowledge Ott’s many years of research on inconspicuous freshwater and marine red algae. Our research also highlights the lack of careful descriptions in many of the records of this genus and the lack of morphological characters to distinguish species. Especially within the morphologically simple red algae, morphological distinctness does not necessarily reflect evolutionary divergences.
Many red algal morphospecies comprise a grouping of several genetic entities, which are usually referred to as cryptic species (Le Gall and Saunders 2010). Our ability to distinguish these genetic entities using traditional morphological diagnoses, while commendable, may not be achievable (Verbruggen 2014). This is especially true for the more simply constructed red algae (e.g., Zuccarello et al. 2011).
The ubiquity and taxonomy of the ‘simple’ red algae once considered in the class Bangiophyceae is slowly being resolved (e.g., Zuccarello et al. 2008, 2010, 2011, Necchi et al. 2013). The old Bangiophyceae is now subdivided into six separate classes (Yoon et al. 2006), one of which is the Stylonematophyceae. The Stylonematophyceae is comprised of 14 microscopic genera that are mostly marine but two are freshwater and one is terrestrial (Zuccarello et al. 2008). Most of the genera are filamentous and two are unicellular (
Joly (1965) recorded
Krishnamurthy (1957) obtained
The basic problem with species and generic identification in the Stylonematophyceae is that they are not easily discernible from nomenclatural descriptions, and without molecular data are even more uncertain. Taxonomic progress requires careful field collections, culture and molecular observations. Even though
We collected an isolate of
Pale brown / red microscopic filamentous specimens were collected by Richard Wetherbee in a shallow sandy tidal pool on Jul 6, 2013 just north of Noah Beach car park (ca. 16˚07.896′ S, 145˚27.302′ E), Cape Tribulation, Queensland, Australia. Initially sand samples were placed in culture flasks with seawater from the site and held at about 20 ± 2℃ on a rotary shaker at approximately 25 rpm and 30 ℃mol photons m-2 s-1 cool white LED lighting at 10 : 14 LD daily cycle. Multiseriate filaments were checked for epiphytes and isolated by excising short segments about 1 mm long. These were placed in 50 × 70 mm culture dishes with 1/4 strength Modified Provasoli’s Medium (20 mL enrichment per litre sterile seawater) to supress epiphyte growth. GeO2 and sodium penicillin G were added to inhibit diatoms and cyanobacteria, respectively (West 2005). For slower growth light was reduced to about 6-7 µmol photons m-2 s-1. Other conditions were maintained as stated above. The culture was designated as JAW 4835.
Molecular analyses were done on the Australian specimen and two other isolates: UTEX LB 2854 from Guajataca, Puerto Rico (Ott MO 7041) and Ott MO 7045 (isolated Nov 27, 2004 from specimens forming distinct bands on rocks from Fisherman’s Cove, Chennai, India and obtained by V. Krishnamurthy, JAW 4567).
Total DNA was isolated from silica gel-dried cultured material using a modified CTAB procedure (Zuccarello and Lokhorst 2005). Amplification and sequencing of the plastid-encoded large subunit of the ribulose bisphosphate carboxylase / oxygenase gene (
Sequences were edited, assembled and aligned using the Geneious software package (Biomatters, available from http://www.geneious.com/). Alignment was straight forward as no gaps were found in the data set and the two genes were combined. The program Modeltest version 3.7 (Posada and Crandall 1998) was used to find the model of sequence evolution that best fit the data set by an Akaike Information Criterion (AIC) (Posada and Crandall 2001). Maximum likelihood (ML) was performed with RAxML 7.2.8 (Stamatakis 2006). RAxML was performed with all threes codons partitioned and the GTR + gamma model and 500 non-parametric bootstrap replicates (Felsenstein 1985).
Bayesian inference was performed with MrBayes v3.1.2 (Ronquist and Huelsenbeck 2003). Analyses consisted of two independent simultaneous runs of one cold and three incrementally heated chains, and 3 × 106 generations with sampling every 1,000 generations. Codons were partitioned. The log files of the runs were checked with Tracer v1.5 (Rambaut and Drummond 2007) and a burn-in sample of 100 trees was removed from each run before calculating the majority rule consensus tree.
A field specimen grown in culture showed a uniseriate filament branching and forming multiseriate upper sectors that became tubular with a single peripheral layer of cells (Fig. 1A & C). Thalli were up to 1.5 cm long and red coloured (Fig. 1A). Cell divisions in the uniseriate section were initially transverse and then longitudinal to irregular in orientation forming biseriate and multiseriate sectors (Fig. 1B). Older tubular branches were composed of a single peripheral layer of cells containing multicellular packets of a polyhedral appearance with a thick colorless matrix (Fig. 1C) and some of these vegetative cells were released directly as spores (Fig. 1B). Each cell had a single nucleus laterally displaced by a single multilobed plastid with a large central pyrenoid (Fig. 1C). In this
The spherical spores were 9-10 ℃m in diameter and displayed slow rotation combined with gliding movement before settling on a substrate. No amoeboid movement was seen. Germination was initiated by a series of cell divisions, with second divisions usually at right angles to the first divisions and then dividing in various planes to form a multicellular aggregate enclosed in a thin matrix (Fig. 1D). Erect shoots normally developed from aggregates of 12 or more cells (Fig. 1D, two arrowheads) but sometimes an erect filament formed directly from an undivided spore (Fig. 1D, single arrowhead). In low light (6-7 ℃mol photons m-2 s-1) the basal system was extensive and erect filaments formed late (Fig. 1D). In brighter light (20-22 ℃mol photons m-2 s-1) erect branching from the basal system was enhanced (Fig. 1E).
Isolate MO1560 (JAW 4283) was analysed for low molecular weight carbohydrates (sorbitol 1,102 mmol kg-1 DW and digeneaside 20 mmol kg-1 DW, floridoside was not detected). Isolate MO70450 (JAW 4509) had sorbitol 485 mmol kg-1 DW, digeneaside 14 mmol kg-1 DW and floridoside 2 mmol kg-1 DW (Ulf Karsten, personal communication).
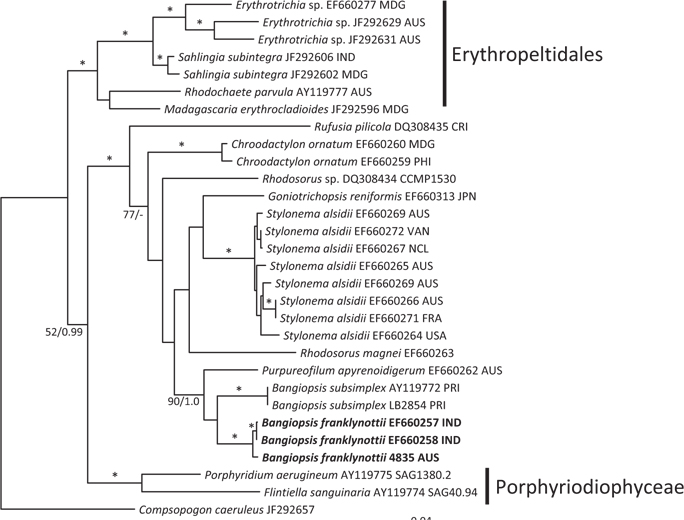
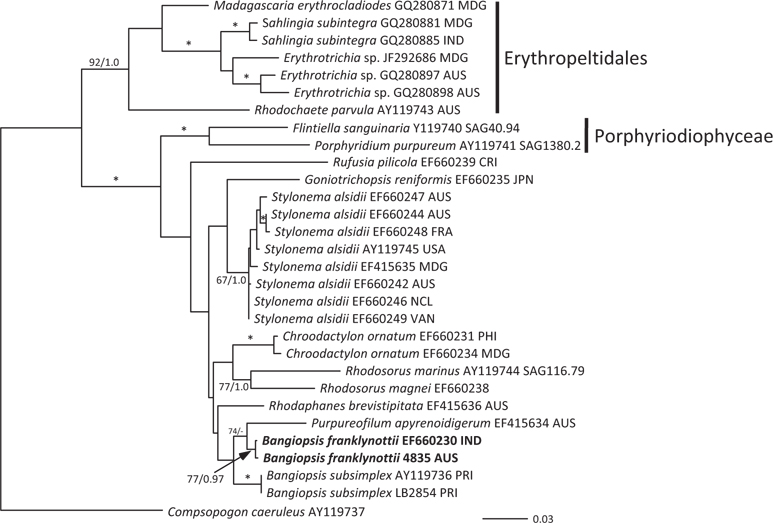
The topologies of the two gene trees are similar to previous phylogenies of the class Stylonematophyceae, and also similar between the ML and Bayesian analysis. The class Porphyridiophyceae and order Erythropeltidales are both well supported, and
Pairwise differences between the two species also support this species distinction. Inter-species pairwise differences are over six times greater than the intra-species variation. For
>
Bangiopsis franklynottii sp. nov. J. A. West, de Goer et Zuccarello
Description. Cells with single nucleus and a single central multilobed plastid containing a central pyrenoid. Basal attachment of thallus by a small multicellular disc giving rise to uniseriate and then multiseriate erect shoots by intercalary cell divisions. Mature thallus often becoming a hollow cylinder with a clear thick mucilaginous matrix in which cells are solitary or in packets. Monospores are discharged directly from vegetative cells along the axis.
Culture 4835 (CCMP 3416) was deposited in the National Center for Marine Algae and Microbiota (NCMA, https://ncma.bigelow.org/), 60 Bigelow Drive, PO Box 380, East Boothbay, Maine 04544, USA. A herbarium specimen was deposited at the National Herbarium of Victoria, Royal Botanic Gardens, Birdwood Ave, South Yarra VIC 3141, Australia (MEL 2371924). Genbank accession Nos:
Our data clearly show that the isolates from Australia and the isolates from India are different from the Atlantic Ocean isolates from Puerto Rico. These genetic analyses clearly show that these isolates are distinct and probably have a common ancestor a long time ago. Based on
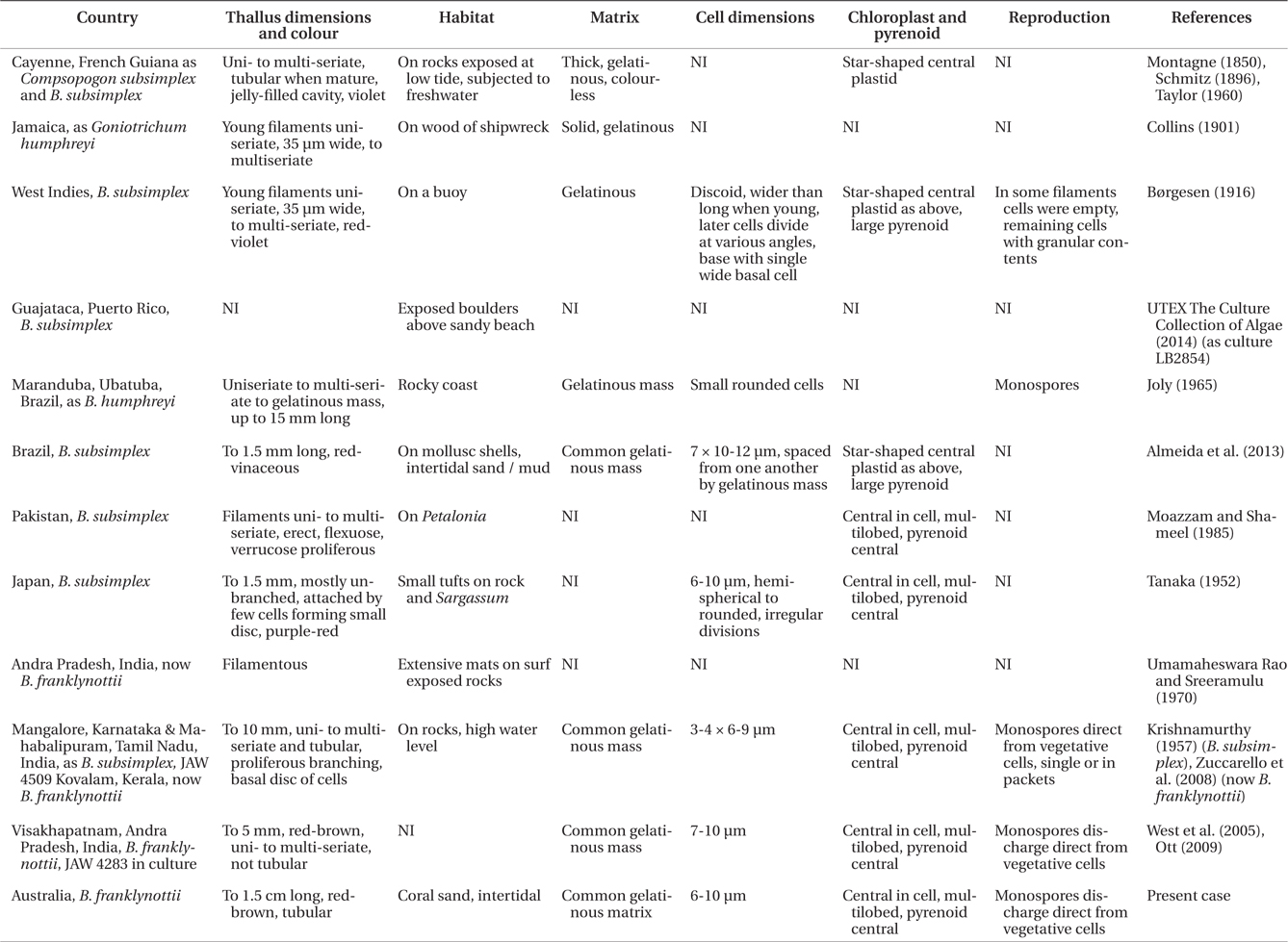
Comparison of morphological and anatomical features of Bangiopsis from the literature and this study
West et al. (2005) compared
Spore formation and germination are very similar in cultured specimens from Australia and India although the mature thalli in the Australian isolate are clearly tubular with a peripheral single-celled layer. The tubular thalli were not evident in the culture of the Indian isolate (West et al. 2005) but Krishnamurthy (1957) observed tubular thalli in field specimens from India.
As with many of these small red algae their distribution and diversity has been poorly explored. Careful culture observations are needed, along with molecular analysis to confirm species identity (e.g., Zuccarello et al. 2010). Critical evidence on the geographic distribution and genetic variation of