The objective of this study is to investigate the effects of Salviae Miltiorrhizae Radix hot aqueous extract on nitric oxide (NO) and prostaglandin E2 (PGE2) production and on 1,1-diphenyl-2-picryl hydrazyl (DPPH) free-radical scavenging in macrophages.
Salviae Miltiorrhizae Radix (300 g) was heated at 100℃ with distilled water (2 L) for 4 hours. The extract was filtered and concentrated to 100 mL by using a rotary evaporator, was frozen at -80℃, and was then freeze-dried by using a freezing-drying system. The RAW 264.7 macrophage was subcultured by using 10-㎍/mL lipopolysaccharide (LPS). In order to evaluate cytotoxicity, we performed (3-(4,5-dimrthylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) (MTT) assays and measured the cell viability. The NO production was measured by using Griess assays, and the PGE2 production was measured by using enzyme immunoassays. The antioxidant activity, the DPPH free-radical scavenging capability, was measured by using the DPPH method.
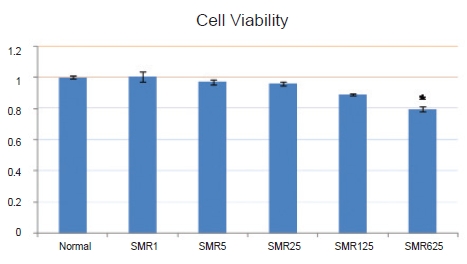
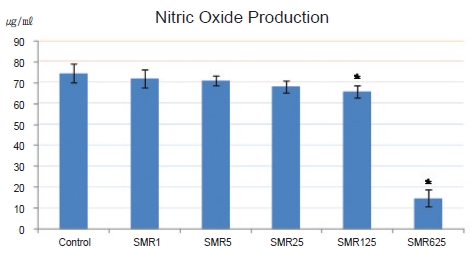
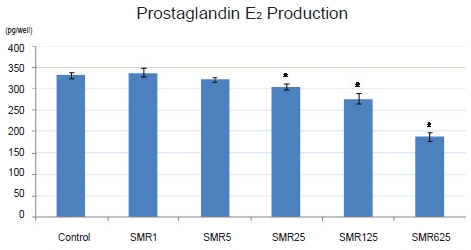
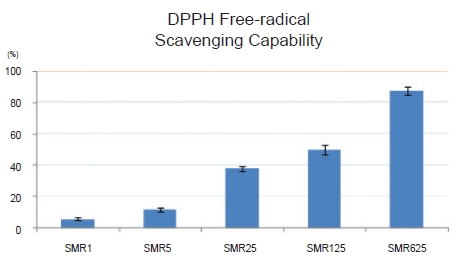
Cell viability with the 1-, 5-, 25-, 125- and 625-㎍/mL Salviae Miltiorrhizae Radix hot aqueous extract was not significantly decreased compared to the cell viability without the extract. When 125 and 625 ㎍/mL of Salviae Miltiorrhizae Radix hot aqueous extract were used, NO production in LPS-stimulated RAW 264.7 macrophages was significantly inhibited compared to that in the control group. When 25, 125, and 625 ㎍/mL of Salviae Miltiorrhizae Radix hot aqueous extract were used, PGE2 production in LPS-stimulated RAW 264.7 macrophages was significantly inhibited compared to that in the control group. The 125- and 625-㎍/mL Salviae Miltiorrhizae Radix hot aqueous extracts had high DPPH free-radical scavenging capabilities in RAW 264.7 macrophages.
This study indicates that Salviae Miltiorrhizae Radix hot aqueous extract suppresses NO and PGE2 production and improves DPPH free-radical scavenging capability. Thus, it seems that Salviae Miltiorrhizae Radix hot aqueous extract may have an anti-inflammation effect and antioxidant activity.
Inflammation is one of the self-protective abilities against tissue injury, and it has clinical symptoms like pain, heat, redness, and loss of function [1]. If severe inflammation occurs, organs and cells may be destroyed, and changes such as decomposition of proteins and suppression of DNA synthesis may be caused by oxidative stress [2].
The study of
For experimentation, we purchased
The RAW 264.7 macrophages that were used in this experiment were parceled out from ATCC: The Global Bioresource Center (Manassas, USA). Afterward, we cultured the cells by using Dulbecco’s modified Eagle’s medium (DMEM) including 10% fetal bovine serum (FBS) and 1% antibiotic (100-U/mL penicillin and 100-㎎/mL streptomycin: GIBCO). The macrophages were kept in the culture medium at 37℃, with 5% CO2 sustained.
We measured the cell viability by using 3-(4, 5-dimrthylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) assays. First, a 96-well plate that included 1×105 cells in each well was stabilized in culture medium at 37℃, with 5% CO2 sustained. We cultured stabilized macrophages by using
A 96-well plate that included 1×105 cells in each well was stabilized in culture medium at 37℃, with 5% CO2 sustained. We cultured stabilized macrophages by using 10- ㎍/mL LPS and
We measured the PGE2 concentration by using commercial competitive enzyme immunoassay kits purchased from R&D Systems (Minneapolis, USA). A 96-well plate that included 1×105 cells in each well was stabilized in culture medium at 37℃, with 5% CO2 sustained. We cultured stabilized macrophages by using 10-㎍/mL LPS and
A 96-well plate that included 1×105 cells in each well was stabilized in culture medium at 37℃, with 5% CO2 sustained for 16 hours. At first, we diluted
We used the SPSS Windows program (Ver. 10.0) for statistical analyses. All variables are presented as means ± standard deviations. The student’s
When the cell viability of the group not treated with the extract was set at 1.00 ± 0.01, the cell viabilities for the groups treated with 1-, 5-, 25-, 125- and 625-㎍/mL
The NO production of the control group, the group treated solely with 10-㎍/mL LPS, was 74.49 ± 4.64 ㎍/mL whereas the NO productions for the groups treated with 10-㎍/mL LPS and 1-, 5-, 25-, 125-, and 625-㎍/mL
PGE2 production of the control group, the group treated with 10-㎍/mL LPS only, was 31.52 ± 7.68 pg/well, but its values for the groups treated with 10-㎍/mL LPS and 1-, 5-, 25-, 125-, and 625-㎍/mL
The DPPH radical-scavenging capabilities of the groups treated with 1-, 5-, 25-, 125-, and 625-㎍/mL Salviae Miltiorrhizae Radix hot aqueous extract were measured as 5.58 ± 0.83, 11.71 ± 1.35, 37.84 ± 1.52, 50.23 ± 3.11 and 87.59 ± 2.58 percent, respectively. Thus, the values for the groups treated with 125- and 625-㎍/mL Salviae Miltiorrhizae Radix hot aqueous extract were higher than 50% (Fig. 4).
The nature of
Inflammation is an immune response by activated immunocytes. When macrophages are stimulated by using LPS of a gram-negative bacterium, virus, microbe, et cetera, an inflammatory factor is activated to protect the host from the bacterium. However, if pro-inflammatory cytokines, such as IL-6, IL-1ß, TNF-α, NO and PGE2 activated from macrophages by using LPS, are overproduced [19], the inflammation response continues, finally causing arterioles, capillaries and veinlets to dilate, plasma to exude, and leukocytes to move to inflamed sites [1].
Oxidative promoter and suppressor are balanced in the body. When they are unbalanced and oxidative stress occurs, organs and cells may be harmed [2]. Oxidative stress radicals are reactive oxygen species (ROS) and reactive nitrogen species (RNS), such as superoxide (O2-), nitric oxide (NO), hydroxyl radical (˙HO), singlet oxygen (1O2) and hydrogen peroxide (H2O2) [20]. NO is a free radical related with inflammation and is a transporter of the cardiovascular system, the nervous system, and the immune system [21]. Its maintains homeostasis, transports neurotransmitters, serves as an anticancer substance, controls blood pressure and serves as an antibiotic, but excessive NO causes vasodilation, cytotoxicity and tissue damage [22].
PGE2 is also related with inflammation and with pain and fever in damaged tissue. It is synthesized by cyclooxygenase- 2 (COX-2). However, excessive PGE2 causes vasodilation, edema, fever and pain [23]. DPPH, which is used for measuring antioxidant activity, is a very stable free radical and violet compound presenting specific absorption at 517 ㎚. The DPPH radical is very stable in an organic solvent such as alcohol, and discoloration by a proton-radical scavenger can easily be seen, so we used the DPPH free radical to measure antioxidant activity. Generally, an unstable DPPH free radical changes to a stable DPPH-H by gaining an electron or an atom of hydrogen from the antioxidant [24].
In this study we wanted to determine the anti-inflammation effect and the antioxidant activity of
The cell viability of the group not treated with the extract was 1.00 ± 0.01 whereas the cell viabilities of the groups treated with 1-, 5-, 25-, 125- and 625-㎍/mL
NO production of the control group, the group treated with 10-㎍/mL LPS only, was 74.49 ± 4.64 ㎍/mL, but for the groups treated with 10-㎍/mL LPS and 1-, 5-, 25-, 125- , and 625-㎍/mL
PGE2 production of the control group, the group treated with 10-㎍/mL LPS only, was 331.52 ± 7.68 pg/well, but for the groups treated with 10-㎍/mL LPS and 1-, 5-, 25-, 125- , and 625-㎍/mL
The DPPH radical-scavenging capabilities of the groups treated with 1-, 5-, 25-, 125-, and 625-㎍/mL
Based on the above findings, we conclude that
We investigated whether
Based on the results, we could conclude that