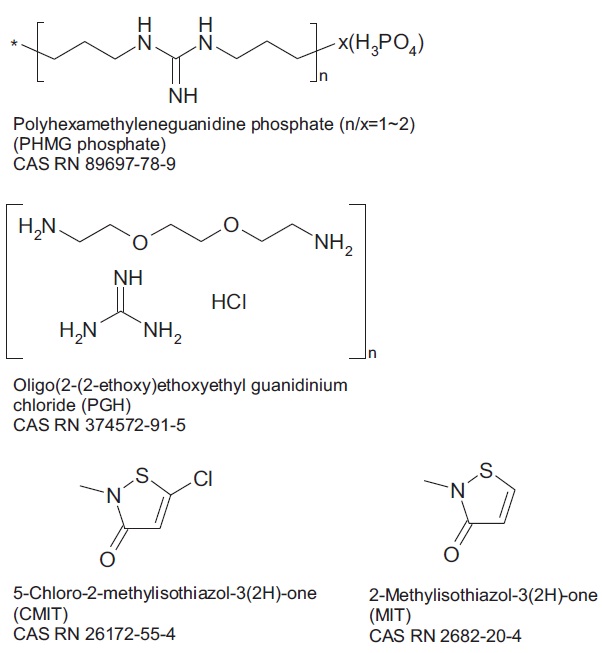
In 2011, the Korea Centers for Disease Control and Prevention reported that an unidentified fatal lung disease was likely to be caused by chemical disinfectants used with household humidifiers [1,2]. The causative active ingredients were identified as polyhexamethyleneguanidine (PHMG) and oligo(2-(2-ethoxy) ethoxyethyl guanidinium chloride (PGH), based on the epidemiological studies and in vivo histopathological readings, after instillation of those active ingredients to rats [3]. Chloromethyl/methyl isothiazolinone (CMIT/MIT) evaluated at the same time was not likely to cause fatal lung disease [3].
Although epidemiological evidences have revealed that those polymeric chemical disinfectants could be fatal when inhaled, quantitative risk assessment for the inhalation of those chemicals have not been conducted, except for a screening-level health risk assessment [4]. Lee et al. [4] assumed that the active ingredients of humidifier disinfectants are inert and homogeneously distributed in a bedroom (i.e., no chemicals react, precipitate, or were removed by other pathways). The resulting health risk quotients were calculated at 2,500, 10,500, and 9.41 for PHMG, PGH, and CMIT/MIT, respectively. These values at the screening level risk assessment are very high, indicating potentially significant health concerns, and requiring a refined risk assessment. Uncertainties with risk quotient at the screening-level lie in uncertainties with the predicted exposure concentration using simple steady-state modeling, as well as the reliability of the “read across” method used to predict the long-term toxicity data for the derivation of the reference concentrations.
In this study, we intended to measure the realistic exposure concentration, based on the human exposure scenarios under normal and excessive use conditions. Products of humidifier disinfectants containing three active ingredients (PHMG, PGH, and CMIT/MIT) were added to the humidifier water at the level recommended by the product suppliers and released to a room using a household humidifier. Aerosols containing active ingredients were sampled at a pumping rate close to the average breathing rate for Koreans, to estimate the human intake rate according to the exposure scenarios. Refined heath risk quotients were estimated, based on the measured intake rates.
2.1. Active Ingredients and Chemicals
Three active ingredients of the humidifier disinfectants used in this study were PHMG, PGH, and CMIT/MIT. Chemical structures of all three ingredients are shown in Fig. 1. Aqueous solutions of PHMG-phosphate (25% w/w) and PGH (25% w/w) were kindly provided by SK Chemical Industries, Inc. Analytical grade 5-chloro-2-methyl-4-isothiazolin-3-one (CMIT, 98%) was purchased from Dr. Ehrenstorfer GmbH (Augsburg, Germany), and analytical grade 2-methyl-4-isothiazolin-3-one (MIT, 98%) was purchased from Sigma-Aldrich (St Louis, MO, USA). Three commercial humidifier disinfectant products containing three active ingredients each were purchased from the market, before they were recalled after November, 2011.
Eosin Y solution (0.5% w/v), glycine (98.5%), magnesium nitrate (Mg(NO3)2, 99%) and magnesium chloride (MgCl2, 99%) were purchased from Sigma-Aldrich. Hydrochloric acid (35%) was purchased from Dae-Jung (Siheung, Korea). Methanol (high performance liquid chromatography [HPLC] grade) was purchased from Burdick & Jackson (Ulsan, Korea).
2.2. Sampling Site, Generation of Humidifier Aerosols, and Aerosol Sampling
A bedroom (area, 21 m2; volume, 47 m3) was rented for the experiments. A humidifier that uses a water boiling system combined with ultrasonic mist generation was used for the production of mists. This type of the humidifier was similar to those humidifiers used by patients who suffered the unidentified lung disease. The water tank size was 4.8 L and approximately 4 L evaporated during the 8 hr sampling period.
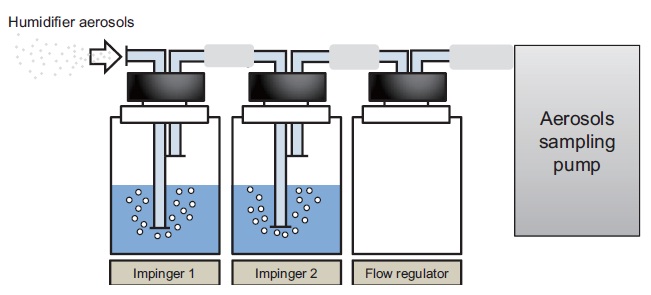
Air containing humidified aerosols was sampled using a custom- made air sampler, consisting of two serial 250 mL impingers containing 100 mL of aqueous solution and a constant flow sample pump (The QuickTake 30; SKC Inc., Eighty Four, PA, USA) (Fig. 2) [5]. Distilled water was used as the sampling medium for PHMG and PGH aerosols, and aqueous solution containing 2.5 g/L Mg(NO3)2 and 0.5 g/L MgCl2 was used for sampling CMIT/MIT, because PHMG and PGH are cationic polymers, and CMIT/MIT have high water solubility. Preliminary studies using a sampler with four serial impingers showed that two impingers were sufficient for quantifying the disinfectants in the sampled aerosols, since the trapped amount from the third impinger was below the detection limit. Air was sampled at the rate of 7.5 L/min, the mean breathing rate for Koreans at rest [6] with all windows and doors closed. The room was ventilated by opening all windows and doors, between independent exposure measurements, to minimize any potential carry-over effects from the previous measurement.
2.3. Instrumental Analyses of Active Ingredients
2.3.1. Spectrophotometric determination of PHMG and PGH
Quantitative analysis of polymeric active ingredients (PHMGphosphate and PGH) containing guanidine group was conducted, using the color-changing reaction of guanidine with tetrabromofluorescein (Eosin Y) [7,8]. Glycine buffer solution at pH 3.6 was prepared by adding 50 mL of 0.1 M glycine solution, and 2.5 mL of 0.2 M hydrochloric acid to 100 mL aqueous solution. Analytical standards were prepared at 1, 2, 4, 6, and 8 mg/L for PHMG-phosphate and PGH, for spectrophotometric determination of their concentration in the sampling solution. In case the sampled concentration exceeded the range of analytical standards, the solution was diluted appropriately before the analysis. Sample solution (10 mL) taken from the impingers was mixed with 10 mL of glycine buffer solution (pH 3.6) and 1.0 mL of 0.05% (w/v) Eosin Y solution. The mixture was vortexed briefly, and left for 5–10 min at room temperature for color development. Then, the absorbance of the mixture was measured at 549 nm, using a DR/4000U UV/Vis spectrophotometer (Hach Company, Loveland, CO, USA).
2.3.2. HPLC analysis of CMIT/MIT
Aqueous samples containing CMIT/MIT mixture and stabilizers (Mg(NO3)2 and MgCl2) were quantified, using an HPLC system equipped with a Waters 515 pump (Waters, Milford, MA, USA), an autosampler (Waters 717+), and a photodiode array detector (Waters 996). In order to exclude the potential degradation of CMIT and MIT by strong nucleophiles [9,10], the collected samples were analyzed within 8 hr. Samples were separated on an ODS HYPERSIL C18 column (4.6 × 150 mm, 5 μm particle size; Thermo Scientific, Hampton, NH, USA) at ambient temperature, under an isocratic condition (methanol:water = 15:85, v/v) with a flow rate of 1 mL/min. Injection volume was 10 μL and both compounds were monitored at 280 nm. The chromatographic retention times were 11.3 and 3.3 min for CMIT and MIT, respectively.
2.4. Modeling Exposure Concentration
2.4.1. Emission scenarios
According to the suppliers of humidifier disinfectants, 20 mL of the liquid product should be added to approximately 4 L water in a tank. Thus, we set the normal exposure scenario (scenario 1) that the humidifier runs for 8 hr during sleeping, and almost all water in the tank (approx. 4 L) is consumed during the sampling time. In order to estimate the level of exposure considering the worst-case, the amount of humidifier disinfectants added to water was assumed 2 times (scenario 2) and 10 times (scenario 3) that suggested by the suppliers.
2.4.2. Modeling the uptake of humidifier disinfectants
The behavior of humidifier disinfectants in the room could be explained by a simple mass-balance model:
where,
The total mass of humidifier disinfectant sampled (
Thus,
The time-weighted average concentration of humidifier disinfectants (
Method detection limits (MDL) of the analytical procedure were derived using an error distribution [11]. MDL for the spectrophotometric determination of PHMG-phosphate and PGH were 0.20 mg/L. For the quantification of CMIT and MIT using HPLC, MDL were 0.11 and 0.29 mg/L for CMIT and MIT, respectively. Calibration curves were obtained, using at least 5 levels of analytical standards. The relative standard deviation of the calibration factors was less than 15% for all measurements.
3.2. Estimation of Exposure Concentration
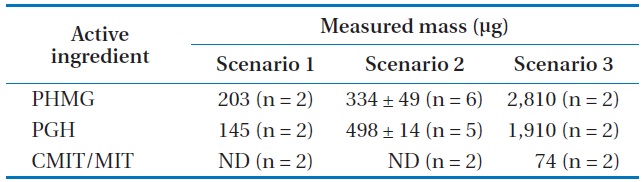
The masses of active ingredients collected by the sampler (M) are presented in Table 1. The time-weighted average concentrations in the bedroom were calculated using Eq. (4), as 0.06, 0.09, and 0.78 mg/m3 for PHMG, and 0.04, 0.14, 0.53 mg/m3 for PGH, according to the exposure scenarios 1, 2, and 3, respectively (Table 2). The concentration of CMIT/MIT in the absorbing solution was only measurable under exposure scenario 3. Because the value of
The possibility of potential deposition or decomposition of PHMG could not be excluded, although it has much lower
[Table 1.] Mass of active ingredients collected by the air sampler for 8 hr
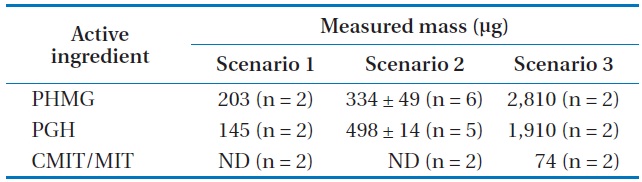
Mass of active ingredients collected by the air sampler for 8 hr
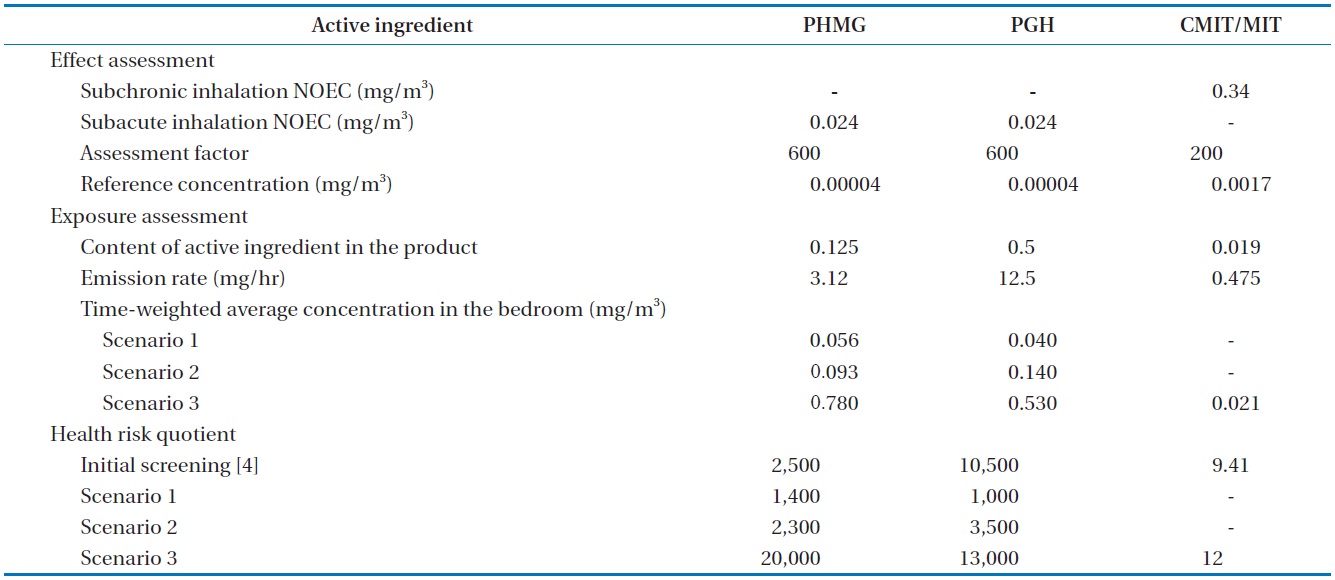
[Table 2.] Derivation of health risk quotients for the use of humidifier disinfectants
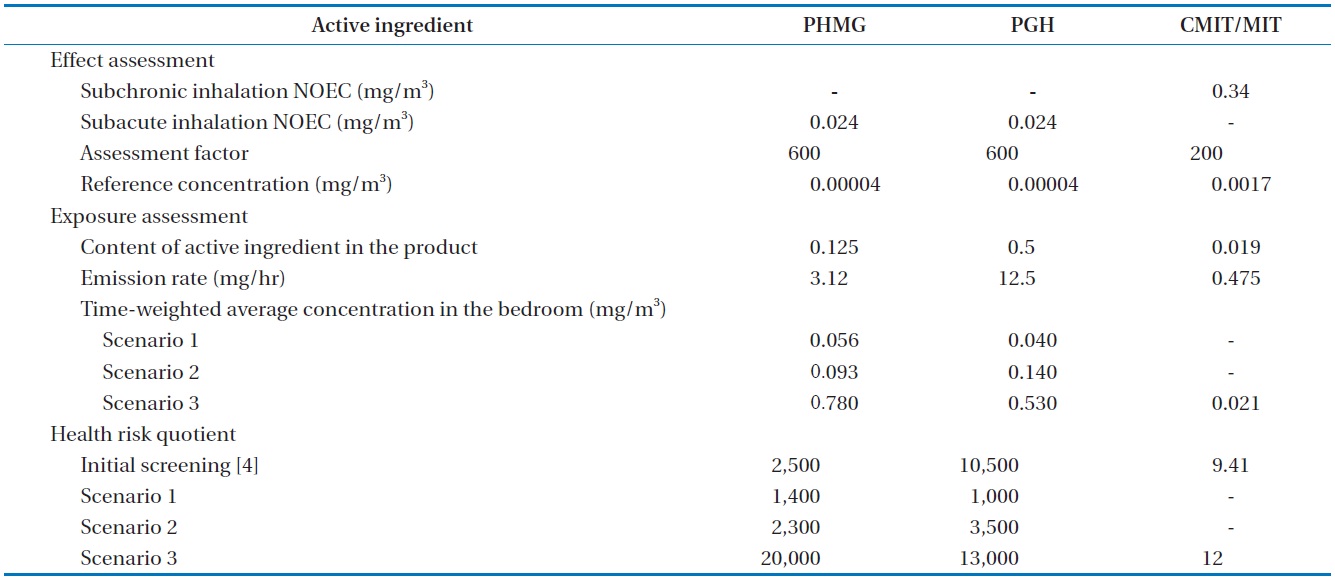
Derivation of health risk quotients for the use of humidifier disinfectants
3.3. Refined Health Risk Assessment Using Exposure Concentration and Indoor Air Modeling
Table 2 summarizes the steps in risk assessment for inhalation of the selected active ingredients of humidifier disinfectants. Whereas a subchronic inhalation no-observed effect concentration (NOEC) was available for CMIT/MIT (3:1) mixture [16], no values were reported for PHMG and PGH. Thus, we adapted a “read across” method, for the derivation of the reference concentrations for those chemicals. The EU harmonized classification and labeling report on polyhexamethylene biguanide (PHMB) reported the subacute inhalation NOEC of 0.24 μg/L using rats [17]. Assessment factors for the derivation of reference concentrations for the three active ingredients in air were 600 for PHG and PHMG and 200 for CMIT/MIT, according to the guideline of the European Chemical Agency [18]. The steady-state concentrations obtained in this study were used for the refined risk assessment. Experimental exposure concentrations for PHMG and PGH in this study were lower by factors of 1.8 and 10.5, respectively, than those calculated in the risk assessment at screening level using paper-and-pencil [4]. However, the corresponding risk quotients were higher than 1,000 for both PHMG and PGH under the normal use condition (scenario 1), indicating significant health concerns. For CMIT/MIT mixture, risk quotient was not calculated under the normal-use condition (scenario 1). Based on the risk quotient of 12 in scenario 3, it could be extrapolated to about unity under the normal-use condition.
3.4. Implications for Risk Assessment
In spite of the uncertainties with inhalation toxicity data, the resulting risk quotients indicated that PHMG and PGH used as humidifier disinfectants are of significant concerns, because measured intake rates and exposure concentration significantly exceeded the reference concentrations for PHMG and PGH, under our exposure scenarios of normal and heavy uses of humidifier disinfectants. The experimental determination of the exposure levels in this study also strengthened the utility of a low-cost initial screening risk assessment [4], because measured intake rates did not decrease significantly for PHMG and PGH.
Because
A refined risk assessment conducted for three active ingredients of humidifier disinfectants by measuring intake rates indicated that the exposure concentrations under plausible exposure scenarios were significantly higher, than the reference concentrations for PHMG and PGH. This confirmed that those chemicals may pose severe health concerns, when they are used as humidifier disinfectants. Unlike PHMG and PGH, low molecular weight biocides, CMIT and MIT seem to undergo significant decomposition in the indoor environment, resulting in the reduced exposure concentrations. Results in this study imply that reactions with indoor surfaces may be important in the evaluation of the atmospheric concentration and corresponding risks for heavily applied spray-type biocides.