Low-rank coal with high water content (32.3 wt%) was dried by fry drying, and the fuel characteristics of the dried coal from which the oil was separated by using a high-speed centrifugal separator were analyzed. After fry drying for 6 min and 10 min, the water content decreased to 5.0 wt% and 4.2 wt% respectively. The higher calorific value (HCV) of the coal increased remarkably after fry drying, from 11,442.0 kJ/kg-wet. The oil content of the fry-dried coal was 15.0 wt% and it decreased with an increase in the reheating temperature: 9.7 wt% at 80 ℃ to 9.3 wt% at 100 ℃, and then to 8.5 wt% at 120 ℃. The recovered oil could then be reused. According to of thermogravimetric analysis (TGA), there was no difference in the weight loss patterns of the coal samples with different coal diameters at a reheating temperature of 120 ℃. This was because the amount of oil separated by the centrifugal separator was affected by the reheating temperature rather than the coal diameter. And derivative thermogravimetry (DTG) curves of raw coal before the fry-drying process, a peak is formed at 400 ℃ in which the volatile matter is gasified. In case of the fry-dried coal, the first peak is generated at 350 ℃, and the second peak is generated at 400 ℃. The first peak is caused by the oil that is replaced with the water contained in the coal during the fry-drying process. Further, the peaks of the coal samples in which the oil is separated at a reheating temperature of 80 ℃, 100 ℃, 120 ℃, respectively are smaller than that of the coal in which the oil is not separated, and this is caused by that the oil is separated by the centrifugal separator.
본 연구에서는 함수율 32.3 wt%인 저등급 석탄을 유중증발 건조한 후 고속 원심분리 장치에서 기름을 분리하여 연료 특성 을 분석하였으며, 유중증발 건조시간이 6, 10분일 때 석탄의 함수율은 각각 5.0, 4.2 wt%까지 감소하였다. 유중증발 건조 후 석탄의 고위발열량은 11,442.0 kJ/kg-wet에서 27,816.0 kJ/kg-wet로 증가하였다. 유중증발 건조한 석탄을 상온에서 원심분리 장치로 기름을 분리한 석탄의 기름 함량은 15.0 wt%이나 건조 석탄을 80, 100, 120 ℃로 재가열하여 기름을 원심분리하면 각 각 9.7, 9.3, 8.5 wt%로 줄어들었으며 석탄 직경별 기름 함량차이는 미약하였다. 원심분리 장치에서 회수한 기름은 유중증발 건조용 기름으로 재사용한다. 저등급 석탄을 대상으로 열중량분석(thermogravimetric analysis, TGA)을 수행한 결과, 건조 후 120 ℃로 재가열한 석탄은 석탄입자 크기에 따른 무게변화는 거의 없었으며 이것은 원심분리 장치 성능이 석탄입자의 크 기에는 영향을 받지 않기 때문이다. 또한, 미분열중량 분석(derivative thermogravimetry, DTG)을 수행한 결과에 의하면 원시 료 석탄은 400 ℃에서 휘발분이 가스화되면서 피크가 나타났고 유중증발 건조한 석탄의 경우 350 ℃에서 1차 피크, 400 ℃에 서 2차 피크가 발생하였다. 1차 피크는 유중증발 건조과정 중에 석탄내부의 수분과 치환된 기름 때문으로 사료되었다. 또한, 80 ℃, 100 ℃, 120 ℃로 재가열하여 기름을 분리한 석탄시료들의 피크가 기름을 분리하지 않은 석탄의 피크보다 작은 것은 원심분리 장치에 의해 기름이 일정량 분리되었기 때문이다.
Global energy crisis is currently becoming a serious threat because of the exhaustion of fossil fuels. In order to overcome the energy crisis, it is important to develop alternative energy sources that ensure a stable supply of energy. In addition, various clean coal technologies for securing the existing energy sources have been proposed, such as removal of ash from coal with an ash content of 20.0-30.0 wt% and drying low-rank coal with a water content of 30.0-40.0 wt%[1-4]. Despite its low ash content (5.0 wt%), low-rank coal has a low heating value, which is attributed to the high water content, and hence, this type of coal is inexpensive. To enhance the quality of low-rank coal, it is necessary to develop a new technique that not only facilitates the removal of water from coal, but also aids in the facile transport and storage of coal by affording coal that does not show spontaneous combustion and water reabsorption.
Conventional coal-drying methods based on convection heat transfer and conduction heat transfer require a long drying time and have low energy efficiency (80.0-85.0%), because of which they cannot be applied to low-rank coal with a high water content (30.0-40.0 wt%). Recently, fry drying, which is based on boiling heat transfer, has been proposed to improve the low heat transfer ratio and energy utilization efficiency on the basis of the characteristics of the material to be dried. Fry drying has a very high energy efficiency of about 95.0%[5-7]; further, lowrank coal (which has high water content) dried by this method does not reabsorb any notable amount of moisture and hence can be safely shipped or stored for a long time. In fry drying, highwater- content coal is brought into direct contact with hot oil (130-150 ℃) to induce boiling heat transfer. During this heat transfer, steam bubbles are generated, which give rise to strong turbulence. In addition, the hole in the coal is locally maintained under the ambient pressure, and the resulting strong diffusion causes the moisture contained in the coal to be replaced with oil, thereby effecting rapid dehydration[8-12]. By fry drying, the water content of coal can be reduced to 5.0 wt% or lesser within 10 min, and the rate of moisture reabsorption after the drying process is very low. However, a large amount of oil may be transferred to the coal dried using this method. Since dried lowrank coal is used as fuel, the presence of oil in it is advantageous, but liquid fuels are more expensive than coal; hence, drying by conventional methods is economically infeasible as recovery of oil, a liquid fuel, is not possible.
To address these issues, we performed an experiment aimed at the separation of oil from low-rank coal that was dried in a fry-drying apparatus to reduce its water content to 5.0 wt% or lower. We conducted the experiment for various coal particle diameters and reheating temperatures and then measured the amount of oil recovered in each case. Moreover, we evaluated the fuel characteristics of the deoiled coal by thermogravimetric analysis (TGA), derivative thermogravimetry (DTG), and differential thermal analysis (DTA)[13,14].
2. Experimental Apparatus and Methods
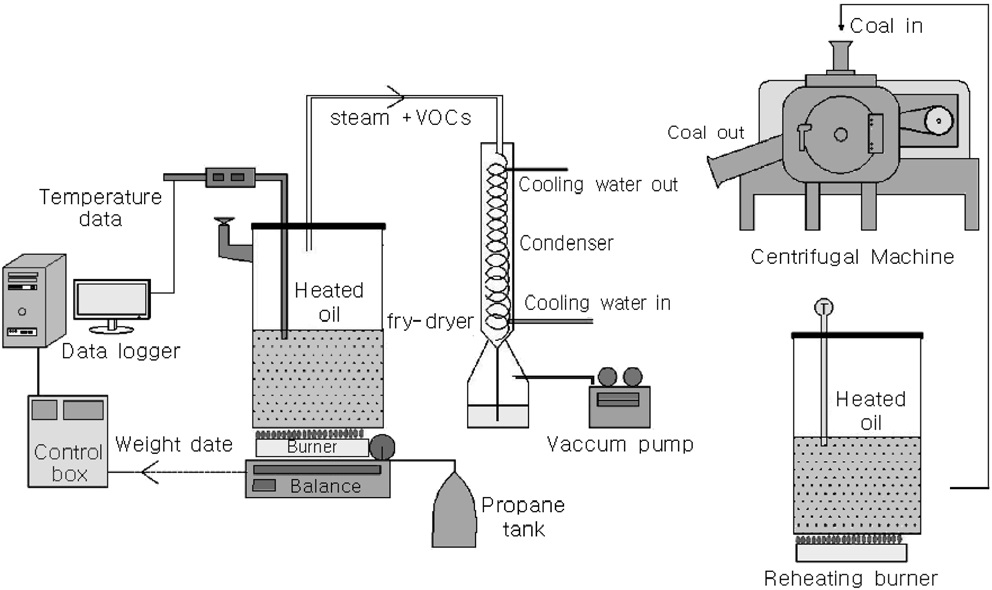
Figure 1 shows the batch-type coal-fry-drying apparatus and oil separation apparatus used in this study. For fry drying, the coal was placed in a rectangular mesh and immersed in oil (2.0 L) filled in a rectangular stainless-steel container.
An electronic scale was installed at the bottom of the apparatus for automatically measuring the rate at weight changes occurred in the apparatus, and a T-type thermocouple was installed to maintain a constant temperature in the evaporator. During the experiments, the oil temperature in the evaporator was
adjusted to 150 ℃, and the error was within ± 3 ℃. A notebook computer was connected to the measuring devices for real-time monitoring of the temperature and weight change rates and storage of these data. When the coal sample was introduced into the oil, a violent reaction occurred between oil and coal, and vapour containing small amounts of oil was generated. The vapour was allowed to pass through a condenser and an oil-water separator for oil recovery. Because the fry-drying apparatus and the oil-water separator were not arranged in series, the fry-dried coal was segregated into three groups on the basis of the particle diameter, and each group was reheated for 5 min at 80, 100, and 120 ℃ in a reheating burner. Subsequently, the oil in the reheated coal samples was separated in a high-speed centrifugal separator.
The experiment performed in this study can be divided into two parts.
Part I: Fry drying of low-rank coal (water content: ≥30.0 wt%) to reduce the water content to 5.0 wt% or lesser
Part II: Separation of oil contained in the fry-dried coal
In the coal dehydration experiment, 100.0 g of coal was dried for 10 min using oil heated to 150 ℃. From the viewpoint of economic feasibility, refined oil was used for the drying process. During fry-drying, a part of the water in the raw coal was replaced by oil. The oil content of the fry-dried coal was about 15.0 wt%, and it was recovered by a centrifugal separator (Contabex: 9,000 rpm). The fry-dried coal was reheated for 5 min at 80 ℃, 100 ℃, and 120 ℃ for 5 min each and then added to the centrifugal separator rotating at about 9,000 rpm to effect oil separation. The water content of the coal after the fry drying was measured by using a Karl Fischer titrator (V20) equipped with a DO308 drying oven. The use of the Karl Fischer titrator helps in preventing measurement errors due to oil fraction contained
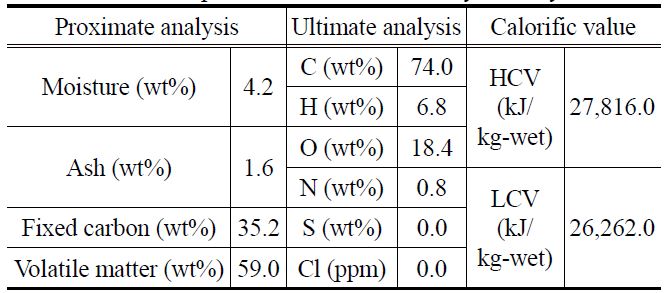
[Table 1.] Results of proximate and ultimate analyses of raw Indonesian brown coal
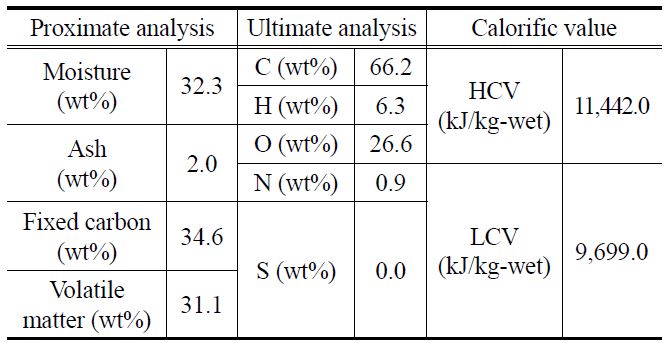
Results of proximate and ultimate analyses of raw Indonesian brown coal
in the sample. Furthermore, the amount of oil in the frydried coal was analysed in a specialized analysis agency.
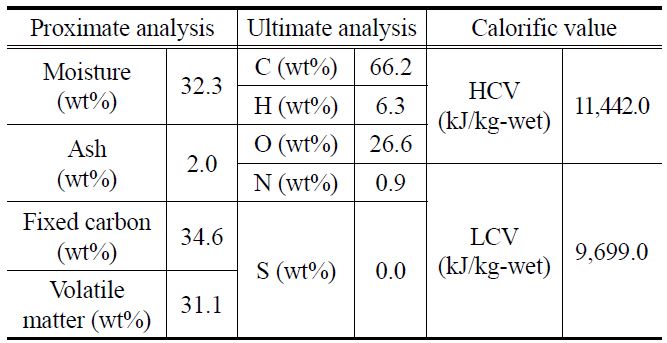
Table 1 shows the analysis results for the raw sample, lowrank Indonesian brown coal, used in this study. The proximate and ultimate analysis equipment are TGA-701 Proximate Analyzer, Truspechn Elemental Analyzer (LECO), SC-432DR Sulfur Analyzer (DIONEX), Analyzer (LECO) and IC-2000 Analyzer (LECO).
The water, fixed-carbon, and volatile matter contents of this coal are 32.3 wt%, 34.6 wt%, and 31.1 wt%, respectively. The calorific value of this coal is very low, as indicated by the higher calorific value (HCV, 11,442.0 kJ/kg-wet) and lower calorific value (LCV, 9,699.0 kJ/kg-wet). For investigating the degree of replacement of water with oil, the abovementioned Indonesian brown coal, which was originally in the form of lumps, was crushed and separated into three groups with particle diameters of 2-3 mm, 4-5 mm, and 6-7 mm.
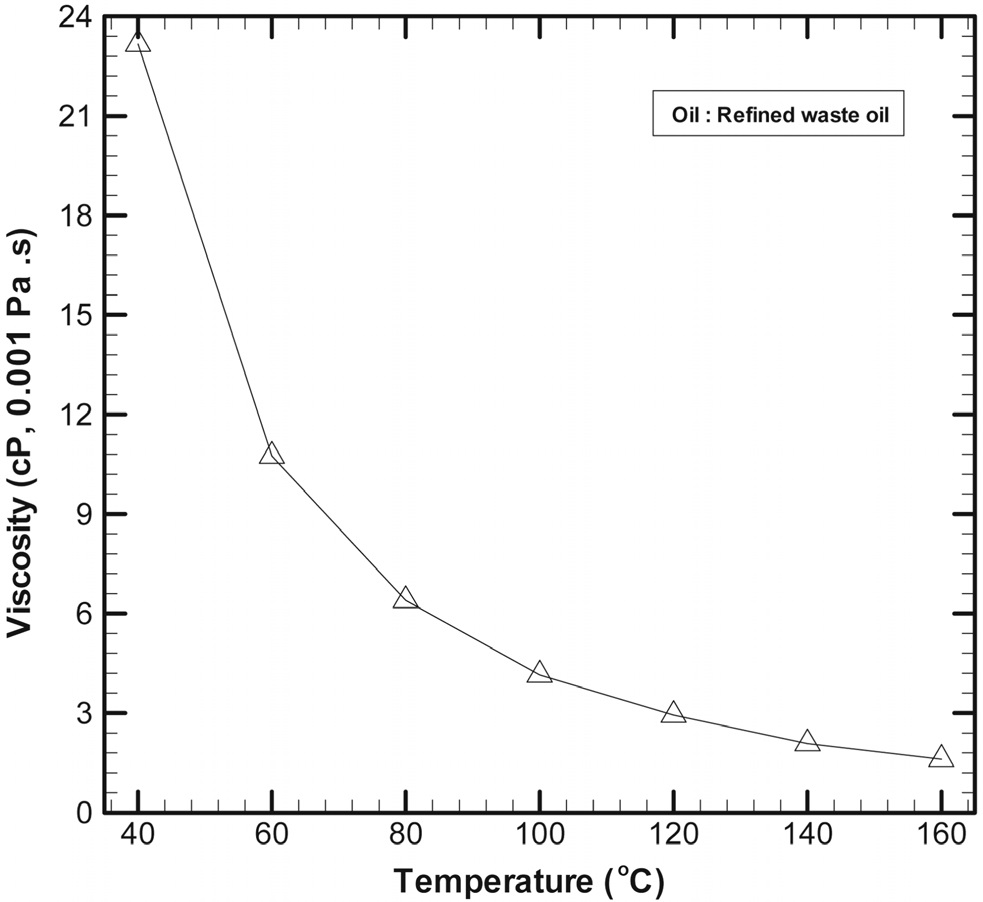
Refined oil, which is recycled from waste engine oil, obtained by removing moisture, ash, heavy metals, and other foreign substances from waste mineral oil, was used in this study. Oil viscosity is a very important factor affecting the degree of replacement of water with oil during fry drying of low-rank coal and the oil separation from the reheated fry-dried coal. In previous fry-drying of organic sludge tests, the higher oil viscosity the higher temperature of fry-drying. Hence, in the present study, the oil viscosity was measured as a function of temperature by using a Brookfield viscometer. The spindle in the viscometer was immersed in the target fluid and rotated at a predetermined speed, and the torque on the spindle was measured. The measurement range of the viscometer was 0.1-800 million cp (cp: centipoise; 1 cp = 0.01 g/(cm s)), and the corresponding speed range was 0.01-250 rpm. The temperature range employed for the viscosity measurement was -100 ℃ to 300 ℃.
Figure 2 shows the viscosity of the refined oil as a function of temperature. At 40 ℃, the viscosity was 23.2 cp, which decreased to 10.8 cp at 60 ℃. Further, the viscosities at reheating temperatures of 80 ℃, 100 ℃, and 120 ℃ were 6.4 cp, 4.2 cp, and 2.9 cp, respectively. The viscosities at 140 ℃ and 160 ℃ were 2.1 cp and 1.6 cp, respectively, indicating that there was only a small difference between the viscosities at 120 ℃, 140 ℃, and 160 ℃.
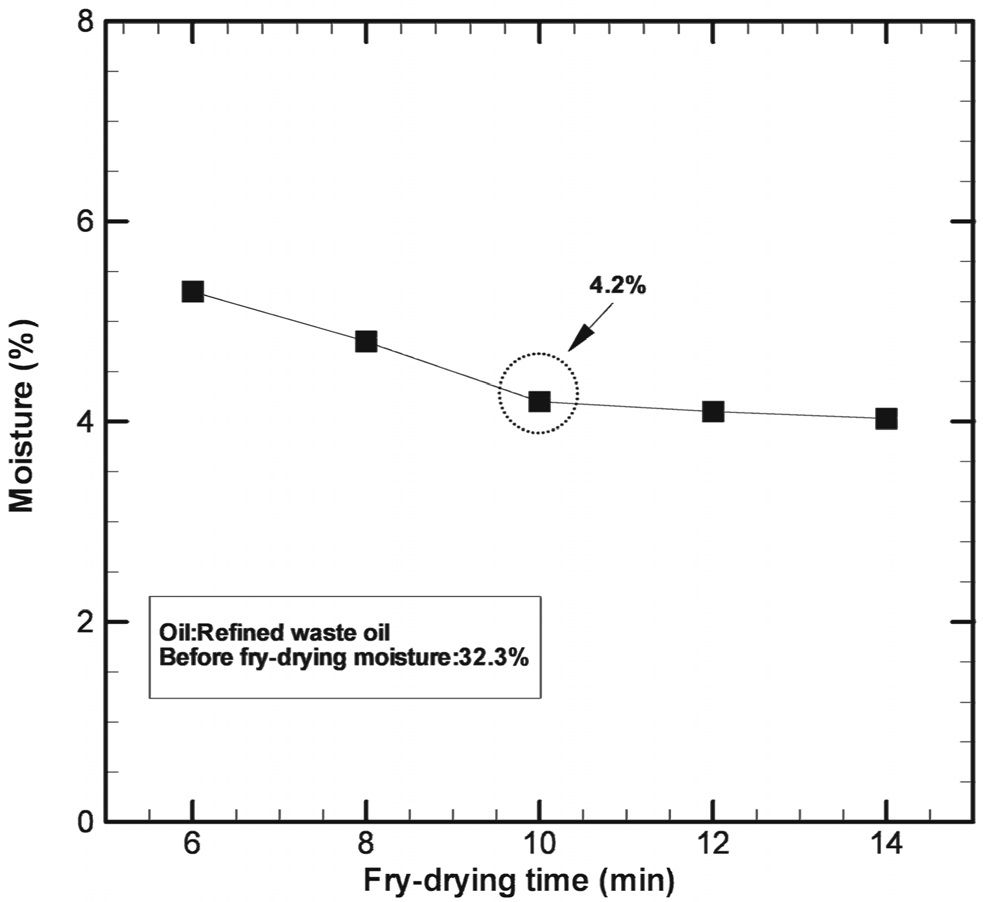
Figure 3 shows the water content of the fry-dried coal as a function of drying time. The parameters used for the analysis were as follows: oil temperature, 150 ℃; weight of the coal sample, 100 g; coal particle diameter, 2-3 mm; drying time, 6, 8, 10, 12, and 14 min. The water content of the coal sample dried for 6 min was reduced to 5.0 wt%. When the drying time was 10 min, the water content was reduced to 4.2 wt%. Since no notable change in the water content was observed when the drying time was extended beyond 10 min, the optimum drying time was set to 10 min. The observations indicated that the drying time required in fry drying is much shorter than that in other direct or indirect drying methods and that the dried coal obtained in the former case has a very low water content. The boiling heat transfer coefficient in fry drying is very high, 500.0 W/m2 ℃. The drying process can be further accelerated (i.e. the drying time can be reduced) if an oil whose molecular weight is lower than the refined oil is used, as doing so would aid the facile replacement of water with oil.
[Table 2.] Results of proximate and ultimate analyses of fry-dried coal
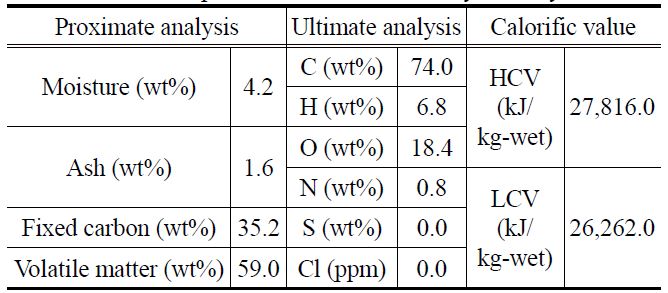
Results of proximate and ultimate analyses of fry-dried coal
Table 2 shows the results of the proximate and ultimate analysis of the coal samples after the fry drying. The water content before fry drying was 32.3 wt%, which rapidly reduced to 4.2 wt% after fry drying. While there was no notable change in the ash and fixed-carbon contents before and after fry drying, the volatile matter content increased considerably from 31.1 wt% to 59.0 wt%. The latter increase was caused by the replacement of water in the coal samples with oil, which contains a large number of volatile components. These observations were confirmed by elemental analysis, the results of which indicated that the carbon and hydrogen contents increased remarkably after fry drying, and calorific value analysis, which showed that the HCV increased considerably from 11,442.0 kJ/kg-wet to 27,816.0 kJ/ kg-wet after fry drying.
Therefore, we concluded that fry dried coals have a desired calorific value proper to conventional coal combustor. However, the drying cost and oil consumption would increase if coal with very high oil content is to be prepared. Therefore, it is economical to separate the maximum possible amount of oil from the fry-dried coal and reuse it.
3.2. Results of oil separation at different reheating temperatures
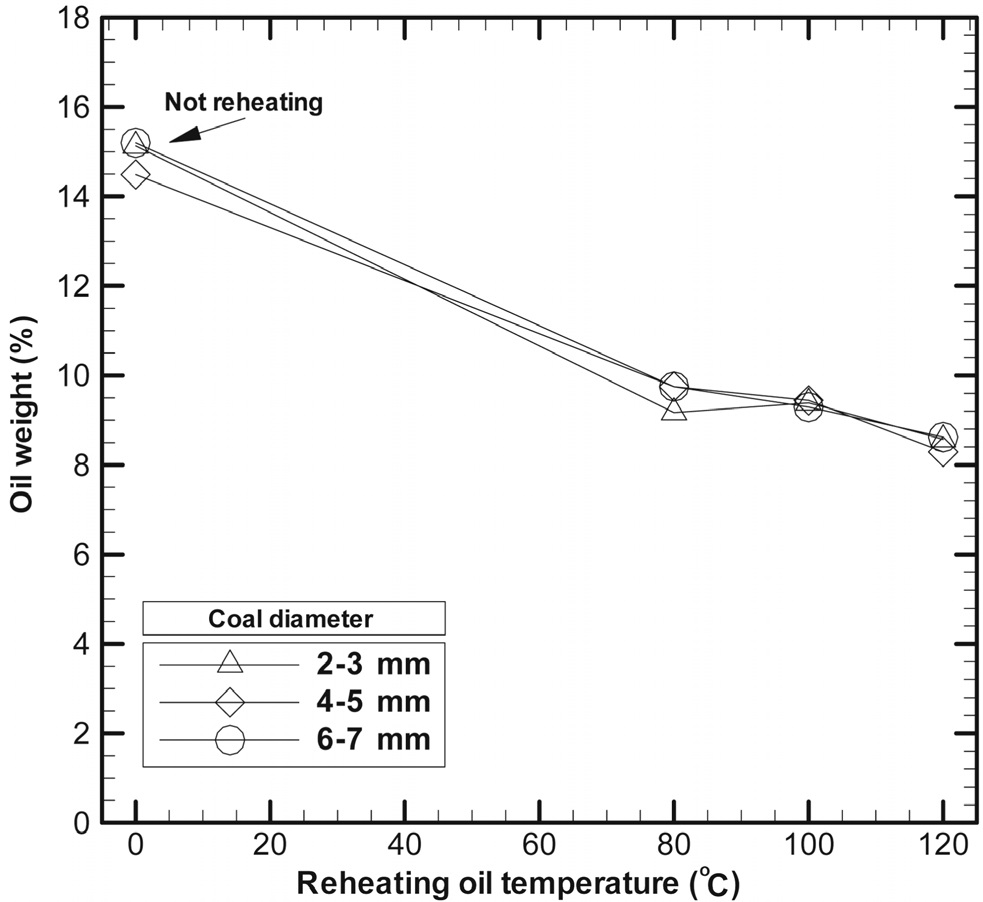
Figure 4 shows the results for oil separation from reheated coals and non-reheated dried coals. The oil content of fry-dried coal was about 15.0 wt% before oil recovery, and this amount differed slightly with the coal particle diameter. Coal samples having an oil content of 15.0 wt% were reheated for 5 min at 80 ℃, 100 ℃, and 120 ℃, and then, the oil was separated by using the centrifugal separator. In this study, the degree of oil separation from the fry-dried coal was tested by a mechanical separator after reheating.
In tests of oil separation from the fry-dried coal after reheating, the rotational speed of the centrifugal separator was set at 9,000 rpm, and the amounts of the separated oil from the coal were within a margin of error of ± 0.5% in 3 times of measurement. The amount of oil was 9.0 wt% at reheating temperatures of 80 ℃ and 100 ℃ and 8.0 wt% at 120 ℃. In this case, the separated oil from fry-dried coal would be adherent oil on the outside surfaces of the coal. Therefore, it was apparent that a large amount of oil can be recovered if the centrifugal separator is directly connected to the rear end of the frydrying apparatus, in which case oil separation from high-temperature coals would be possible.
When the centrifugal separator was used for oil recovery from the fry-dried coals, the amount of recovered oil changed remarkably with the reheating temperature. This was because the cohesive force of oil is related to the viscosity of oil and the viscosity of oil is affected by oil temperature. Therefore, if the oil temperature is increased to 80 ℃, for instance, the viscosity is reduced by 10.0%, thus making oil separation relatively easy.
3.3. Results of TGA at various reheating temperatures
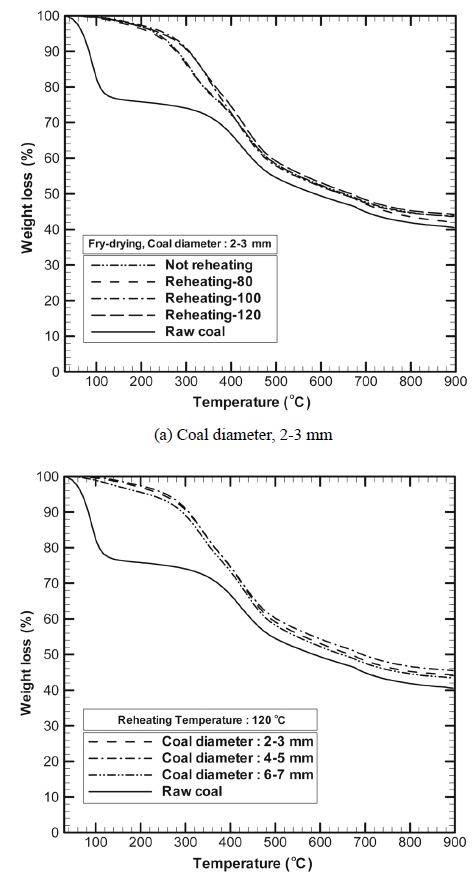
Figure 5(a) shows the TGA curves of coal samples with particle diameters of 2-3 mm at different reheating temperatures, while Figure 5(b) shows the weight loss curves obtained at 120 ℃ for different coal particle diameters. Since the aim of TGA (Mettler Toledo, TGA/SDTA 851e) was to evaluate the efficiency of oil incorporation in the coal samples during fry drying and that of oil separation in the centrifugal separator, nitrogen was used as the carrier gas, so that combustion was prevented and only pyrolysis occurred. The heating rate was 10 ℃/min. Various reactions occurred during pyrolysis. First, unbound water and surface water from the coal samples evaporated at 100-105 ℃, that is, most of the water present evaporated; then, even bound water evaporated at about 300 ℃, thereby effecting complete drying.
After the aforementioned pyrolysis reaction, the high-molecularweight components in the coal samples decomposed. From the curve for the non-dried raw coal shown in Figure 5(a), it could be seen that evaporation occurred at 100 ℃ and gradual weight loss occurred in the range 100-300 ℃. This trend was probably due to moisture loss and discharge of volatile matter. Beyond 300 ℃, drastic weight loss occurred owing to the depolymerization of coal and discharge of volatile matter[15]. Combustion did not occur because the carrier gas used was nitrogen.
The TGA curves of the fry-dried coals indicated that there was hardly any weight loss at 100 ℃ since most of the water had evaporated. In the case of the non-dried raw coal, gradual weight loss occurred at 100-300 ℃. On the other hand, in case of fry drying, oil permeated into the coal, replacing the water present in the coal, and volatilized at 100-300 ℃; consequently, there was increased weight loss. After 300 ℃, the permeated oil and the volatile matter generated by the decomposition of coal decomposed and volatilized simultaneously, resulting in sharp weight loss. As seen in Figure 5(b), there was no difference in the weight loss patterns of the coal samples with different coal diameters at a reheating temperature of 120 ℃. This was because the amount of oil separated by the centrifugal separator was affected by the reheating temperature rather than the coal diameter.
3.4. Results of DTG at various reheating temperatures
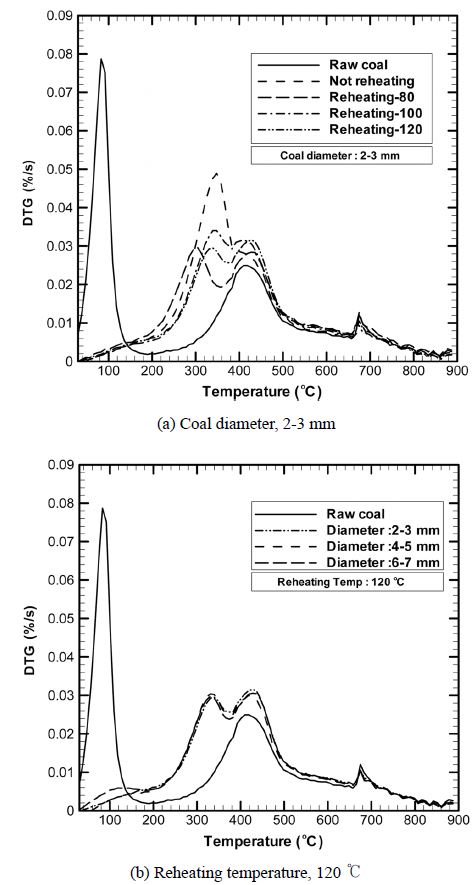
Figure 6(a) shows the DTG curves obtained for coal samples with particle diameters of 2-3 mm at various reheating temperatures and those for samples with diameters of 2-3 mm, 4-5 mm, and 6-7 mm at a reheating temperature of 120 ℃. The pyrolytic characteristics of the coal samples can be understood more clearly from the rate of weight loss with temperature in DTG curve. The peak height is found to be directly proportional to the weight loss.
In general, the conversions occurring during the pyrolysis of coal can be expressed as below[13-15].
The thermogram of the non-dried raw coal shows a large peak
at 100 ℃ attributed to moisture loss, as indicated in reaction (1). The corresponding weight loss is very high because of the loss of the large amount of water (32.3%) present in the raw coal. The peak height increases from 300 ℃, reaching a maximum at 400 ℃. This change is due to the large weight loss resulting from the devolatilization reaction triggered by decomposition of the polymeric materials in the coal (reaction (2)). The small peaks appearing at around 670 ℃ are due to tertiary volatile matter such as hydrocarbons and hydrogen-containing compounds generated when the semicokes formed as per reaction (4) are decomposed at a relatively high weight loss rate. In the thermogram of the fry-dried coals, the large raw-coal peaks at 100 ℃ disappear, indicating a remarkable decrease in the water content after fry drying.
According to the results of TGA and DTG analysis, the peaks at around 670 ℃ do not indicate any dramatic change in the weight loss but are attributable to the thermal decomposition of coal particles into cokes and volatiles, which increases the rate of weight loss change. This phenomenon is probably caused by the separation of volatile matter from the fine pores of the coals, which in turn is due to the destruction of coal particles and polymeric materials such as tar.
The DTG curves shown in Figure 6 indicate that the primary peaks appear at around 300-350 ℃, i.e., at a temperature range lower than that in the case before fry drying, probably because of the influence of oil permeated into the coals during fry drying. In case of the fry-dried coals, primary volatile matter whose molecular weight is lower than that of the volatile matter generated from raw coals are generated from oil, and secondary volatile matter from the coals is generated at about 400 ℃. Further, the secondary peak has the maximum height in case of the non-reheated fry-dried coal but is relatively smaller in case of the reheated, deoiled coals. This is because a large amount of oil is separated in the centrifugal separator from coals reheated at 80 ℃, 100 ℃, and 120 C. The height of the secondary peak appearing at 400 ℃ is small in the thermograms of the nondried raw coals as well as the fry-dried, deoiled coals. The small peak height is due to (1) the fact that volatile matter is generated only from the coal, regardless of the fry-drying conditions employed, and (2) oil separation after reheating. Therefore, it can be understood that the fatty acid components of the refined oil used for fry drying high-water-content coals is mostly volatilized at around 350 ℃. According to the DTG curves for each diameter of coals which are reheated at 120 ℃, as shown in Figure 6(b), particle size of the coals separated oil makes no difference.
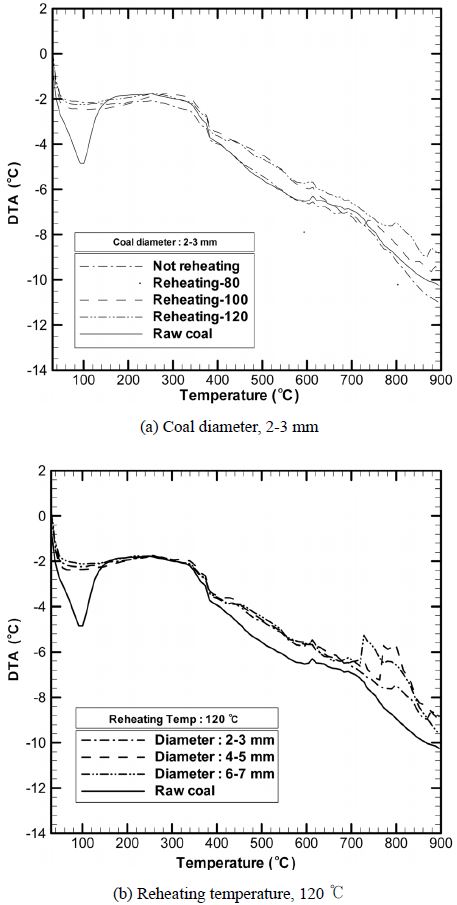
3.5. DTA results at different reheating temperatures
Figure 7 shows the DTA curves for coal samples with different particle diameters at a reheating temperature of 120 ℃ and those for the coal sample with a particle diameter of 2-3 mm at different reheating temperatures. DTA is carried out to measure the temperature difference between a thermally stable standard material and the analytical sample, both of which are heated to the same temperature at the same heating rate. In this study, since coal pyrolysis is analysed using nitrogen as the carrier gas, the entire process involves endothermic reactions. In the case of the non-dried raw coal, the peak at 100 ℃ indicates that a large amount of energy is consumed for the evaporation of moisture. The peaks appearing at 100 ℃, 300-400 ℃, and around 700 ℃ (Figure 5 and 6) are indicative of evaporation,
devolatilization, and pyrolysis, respectively, all of which are endothermic reactions.
In particular, the peaks in the temperature range 700-800 ℃ indicate that with an increase in the reheating temperature and coal diameter, energy consumption is reduced at around 730 ℃. Moreover, with a decrease in the reheating temperature and coal diameter, energy consumption is reduced at around 770 ℃. As indicated in the previous TGA and DTG analysis results, the coal particles are destroyed at around 670 ℃ and decomposed into coke and volatile matter, while the pressure of water and oil in coal is increased by external heat; consequently, energy consumption is reduced.
In this study, low-rank coal with a water content of 32.3 wt% was dried by fry drying, and an oil separation test was carried out using a centrifugal separator. The results are summarized below.
After fry drying for 6 min and 10 min, the water content reduced to 5.0 wt% and 4.2 wt%, respectively. However, there was no notable change in the water content when the fry-drying time was prolonged beyond 10 min, indicating that the optimum drying time was 10 min.
Analysis of the coal samples before and after fry drying indicated that the water content decreased from 32.3 wt% to 4.2 wt% but the volatile matter content increased considerably from 31.1 wt% to 59.0 wt%. There was little difference between the ash and fixed-carbon contents before and after drying, but the results of elemental analysis revealed that the amount of carbon components increased from 62.2 wt% (before drying) to 74.0 wt% (after drying). The HCV of the coal increased remarkably from 11,442.0 kJ/kg-wet (before drying) to 27,816.0 kJ/kg-wet (after drying).
Although the oil content of the fry-dried coal was 15.0 wt% before oil recovery, oil separation by a centrifugal separator resulted in a decrease in the oil content from 9.7% at a reheating temperature of 80 ℃ to 9.3% and 8.5% when the reheating temperature was increased to 100 ℃ and 120 ℃, respectively. The oil content varied slightly with the coal particle diameter.
TGA and DTG results for non-dried raw coal revealed the appearance of two peaks at 100 ℃ and 400 ℃, attributable to the evaporation of moisture and devolatilization of volatile matter, respectively. In case of the fry-dried coals, the first peak was generated at 350 ℃, and the second peak was generated at 400 ℃. The former was due to the replacement of oil with the water contained in the coal during fry drying. Further, the peaks in the thermograms of the coal samples for which the oil separation was effected at reheating temperatures of 80 ℃, 100 ℃, and 120 ℃ were smaller than those in the case of non-reheated coal; this difference was due to oil separation by the centrifugal separator. The DTA results indicated that the observed peaks were due to endothermic reactions only.
cp : centipoise; 1 cp = 0.01 g/(cm s)
m : mass
r : radius of circular motion
w : angular velocity of motion