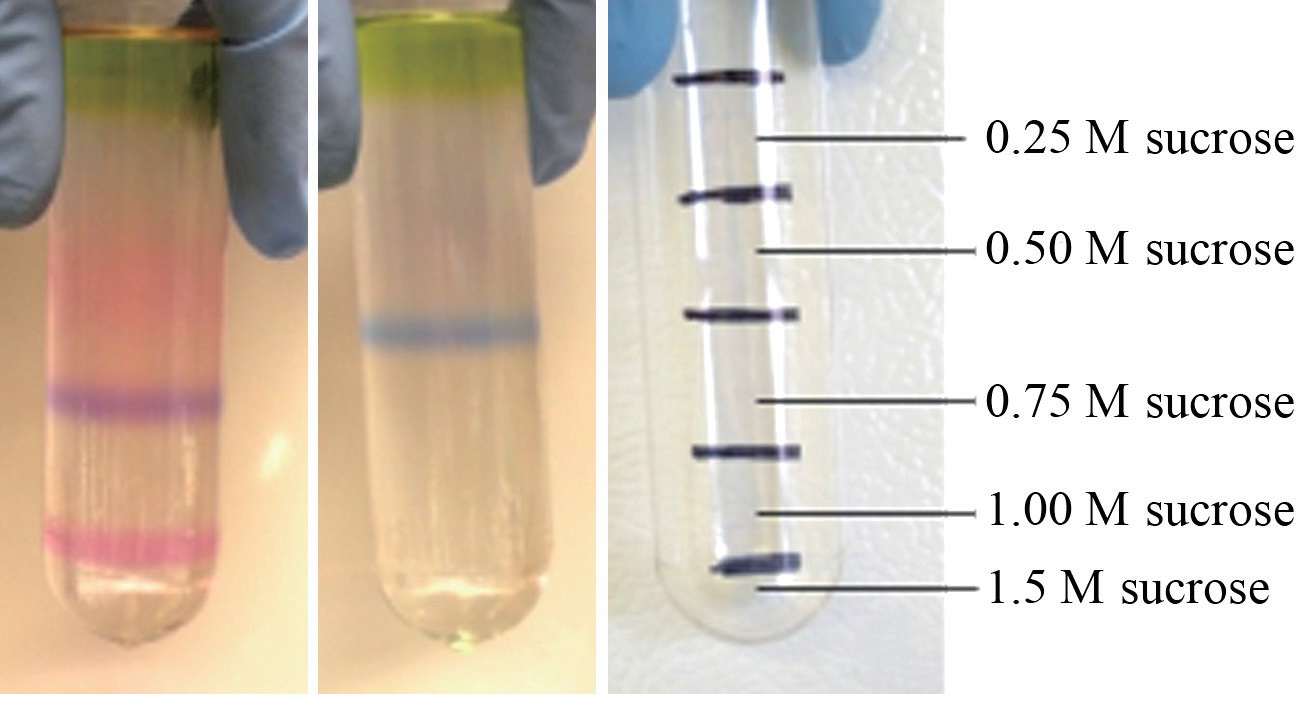
Intact phycobilisomes from a wild-type red Chondrus crispus and its vegetatively derived green mutant were isolated by centrifugation through a discontinuous sucrose density gradient. Pigment composition was subsequently characterized by spectrophotometry. Vegetative thalli of the two strains grown together for six months in the laboratory resulted in different pigment profiles. Two pigmented phycobilisome bands appeared in the sucrose gradient of the wild-type alga, a purple coloured one, and a pink one, whereas only a single blue band appeared in the gradient of the green mutant. Spectrophotometric and fluorescence analyses identified the phycobiliprotein composition of the purple band as the typical phycoerythrin?phycocyanin-allophycocyanin complement in the wild-type, but there was no detectable phycoerythrin present in the blue band of the green mutant. Sodium dodecyl sulphate, preparative polyacrylamide gel electrophoresis analysis confirmed the presence of allophycocyanin subunits in all extracts, but firm evidence of an R-phycoerythrin linker polypeptide in the blue band was missing. These results highlight the ability of C. crispus to adapt to a phycoerythrin deficiency by adjusting light harvesting pigment ratios.
Phycobilisomes (PBS), the supramolecular structures of pigment-protein complexes of red algae and cyanobacteria, serve as integral light harvesting modules (e.g., Arteni et al. 2008, Shi et al. 2011). The functional versatility of PBS is critical to the evolutionary fitness of their hosts. For over a billion years they have enabled the red algae and cyanobacteria to adapt to fluctuations in life sustaining irradiance, effectively enhancing the organisms’ repertoire of adaptation mechanisms. The full complement of phycobiliproteins (PBP) contained in a typical red algal PBS includes R-phycocyanin (R-PC), allophycocyanin (APC), and three forms of phycoerythrin: B-phycoerythrin (B-PE), present in the more primitive red algae, R-phycoerythrin (R-PE), occurring primarily in the higher red algae, and C-phycoerythrin (Lee 2008). The PBP are each composed of two subunits, α and β, and their associated structural components (Grossman et al. 1993).
In nature, well-nourished
Phycobiliprotein complexes are actively formed, dismantled, and rearranged in PBS in response to light conditions and nutrient status. As the most peripheral phycobiliprotein to the APC core, the uncoupling of energy transfer often begins with PE as it becomes dissociated from the PBS complex. Under higher ionic conditions however, the initial uncoupling can occur between PC and APC. These dissociation patterns are thought to be species specific (Gantt et al. 1979, Grossman et al. 1993). Phycoerythrins are the most abundant PBP in many red algae and they display rapid binding and dissociation abilities (Algarra et al. 1990, Glazer 1994, Talarico 1996). The presence of two different PBS structures in some red algae may enhance their ability to acclimate quickly to changing light environments by maintaining a pool of PE PBP in a state of metabolic flux (Algarra et al. 1990, Luder et al. 2001).
In the present study, intact PBS were isolated from
This study compared the PBP complement of a wild type red alga to that of a green colour mutant that arose vegetatively on a frond of the original wild type plant. Ascertaining that the green mutant was PE deficient, and quantifying the resultant ratios of PC and APC would be instructive in establishing commercial value of the alga as a crop. Certain algal pigments have specialized applications, such as in clinical and immunological fluorescence analyses, as natural colorants, and as therapeutic agents for diseases associated with chronic oxidative imbalances (Farooq et al. 2006, Sekar and Chandramohan 2008, Ivanova et al. 2010). Unique pigment ratios contained in the commercially cultivated green mutant should be identified and characterized for potential market application. The potential for improving growth rate by understanding the light harvesting capabilities of the green alga is also of commercial interest.
>
Algal material and culture conditions
A green colour mutant was derived vegetatively from a red (wild-type) strain of
Irradiance was then reduced to 75-80 μmol m-2 s-1 at 16 : 8 L : D for another six weeks, to encourage pigment development.
The PBS extraction procedure was adapted from Algarra et al. (1990), and Gray and Gantt (1975). The algal samples (20 g fresh weight) were washed in pasteurized seawater and rinsed twice in 0.65 M phosphate buffer before placing them into a -80℃ freezer for approximately 24 h. After removal from the freezer, 5.0 g of each algal sample was ground in liquid nitrogen and approximately 2 g of the ground material was added to 50.0 mL extraction solution consisting of 49.0 mL Sorensens’ phosphate buffer (Bozzola and Russell 1999) at pH 7.0, 7.0 μL β-mercaptoethanol, 10 protease deactivator tablets (Ref. #11 836 170 001; Roche Diagnostics GmbH, Mannheim, Germany) and 1.0 mL Triton X-100 in a 100 mL beaker. The samples were stirred gently in the dark at 4℃ for 30 min. Cell debris was removed by centrifugation at 20,000 rpm (33,600 ×g) for 15 min at 4℃ (Beckman J2-M1 centrifuge with fixed angle rotor JA20; Beckman Inc., Palo Alto, CA, USA), and 4-5 mL aliquots of the pigmented supernatant were layered on top of a discontinuous sucrose step gradient (similar to Gantt and Lipschultz 1972) in 0.65-M phosphate buffer (4.0 mL 1.5 M, 7.0 mL 1.0 M, 8.0 mL 0.75 M, 7.0 mL 0.5 M, and 7.0 mL 0.25 M sucrose). The remaining supernatants were retained as whole crude extracts and analyzed separately. Discontinuous sucrose density centrifugation was performed at 27,000 rpm (96,508 ×g) for 17.5 h (Beckman Coulter Le-Bok “Optima” Ultracentrifuge with SW28 swinging bucket rotor) at 4℃. After centrifugation, each of the pigmented PBS bands isolated from the algal samples were withdrawn from the sucrose gradient using a B-D 1M11/2 23GTW needle (Becton Dickinson and Co., Rutherford, NJ, USA) and a 5.0 mL syringe, and collected into 15 mL Fisherbrand plastic vials, covered with aluminum foil to prevent light penetration and frozen at -20℃.
PBS samples were thawed and diluted with 0.65 M phosphate buffer solution at pH 6.97 and centrifuged at 3,300 ×g through an Amicon Ultra Centrifugal Filter Devices in a 1EC Central MP4R SB centrifuge (Millipore, Billerico, MA, USA) for 20 min at 4℃. The PBS were retained on the filter while buffer and sucrose passed through. Retained PBS were re-suspended in fresh phosphate buffer and the centrifugation step was repeated three times to further concentrate the pigment proteins and to minimize the interference effects of the sucrose and some of the other compounds in the samples, such as Triton-X.
A 12% polyacrylamide resolving gel was prepared with a 4% stacking gel and 10 μL samples of whole crude PBS extracts from red and green thalli as well as concentrated PBS samples isolated from sucrose density gradient bands were diluted 1 : 1 in SDS-PAGE sample buffer and loaded into separate lanes in the stacking gel for protein subunit analysis. Samples were loaded by volume and were not normalized for equivalent protein concentrations. Precision Plus (Bio-Rad Laboratories Inc., Richmond, CA, USA) protein standards were resolved in adjacent lanes for molecular mass determination. SDS-PAGE was conducted as per standard operating protocol (Laemmli buffer system). When the majority of the samples reached the bottom of the resolving gel, the gel was removed and washed three times in double distilled (dd) H2O then placed in Biosafe Coomassie stain (Bio-Rad Laboratories Inc.) overnight, and subsequently destained with dd H2O.
The absorption spectra for the pigment samples were recorded at room temperature with a SpectraMax Plus Spectrophotometer (Molecular Devices, Sunnyvale, CA, USA). The fluorescence emission spectra were carried out with a SpectraMax GeminiXS Spectrafluorometer (Molecular Devices) at room temperature with an excitation of 450 nm (similar to Algarra et al. 1990). Absorbance values at 498.5, 614, and 651 nm of both the crude PBS extracts and of the pigmented PBS bands isolated from the sucrose gradient were used to calculate phycobiliprotein content by the following equations (Kursar et al. 1983, Beer and Eshel 1985):
PE (mg mL-1) = [(A564 - A592) - (A455 - A592) × 0.20] 0.12
PC (mg mL-1) = [(A618 - A645) - (A592 - A645) × 0.51] 0.15
APC (μg mL-1) = 181.3 × A651 - 22.3 × A614
PBS isolation profiles resolved by discontinuous sucrose density gradient centrifugation of crude extracts from each algal strain were clearly different from one another. The wild type
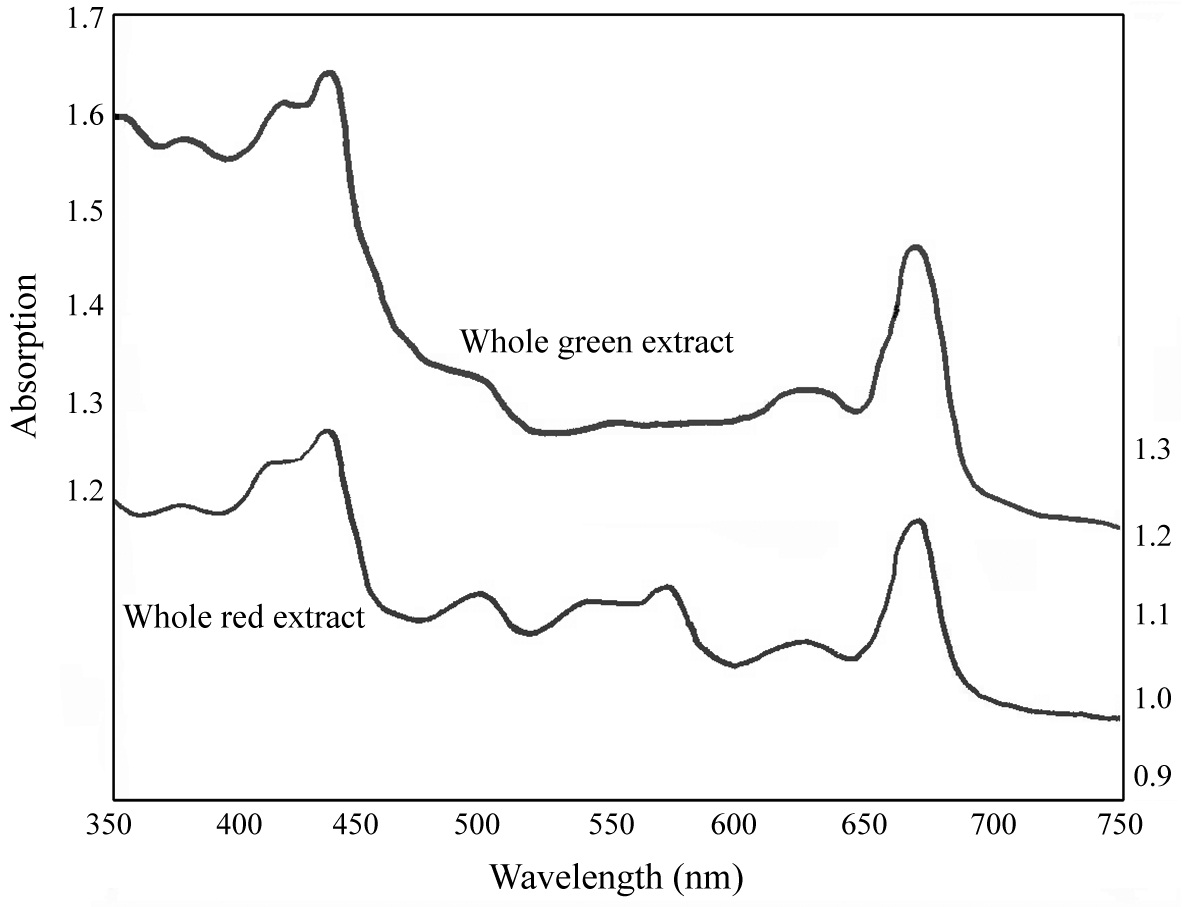
The absorption spectra of the extracts before separation in the sucrose density gradient indicated the presence of phycoerythrin (565 nm) in the wild type red plant (Fig. 3). There was no apparent absorbance maximum for PE in the green mutant (Fig. 3), compared to the red strain. The whole red extract also displayed absorption peaks at 495 nm and 540 nm, suggesting the presence of R-phycoerythrin type I (Gantt and Lipschultz 1972). In both the red and green whole extracts, there was an obvious absorbance maximum at ~650 nm, indicative of APC.
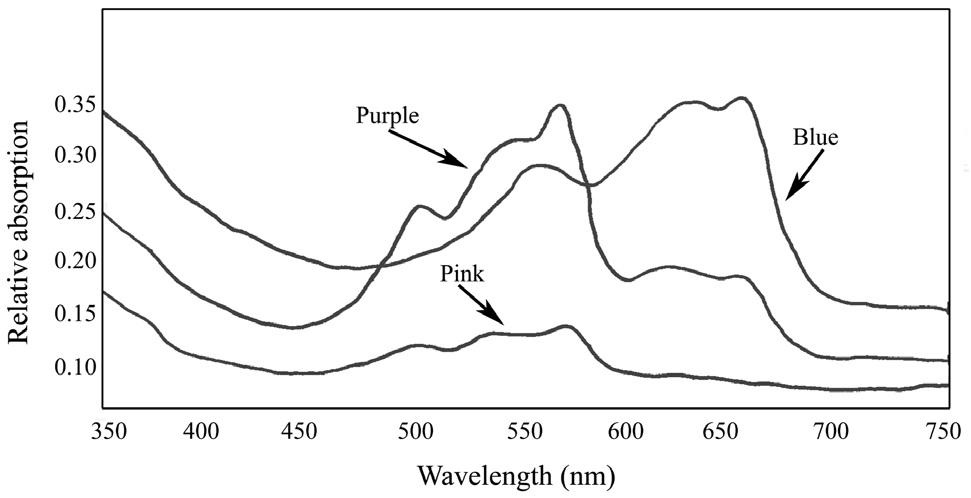
Absorbance spectra of the pigment proteins isolated by the sucrose density gradient showed that the purple band from the wild type
R-PE type I (3 peaks, 498, 540, and 565) (Su et al. 2010
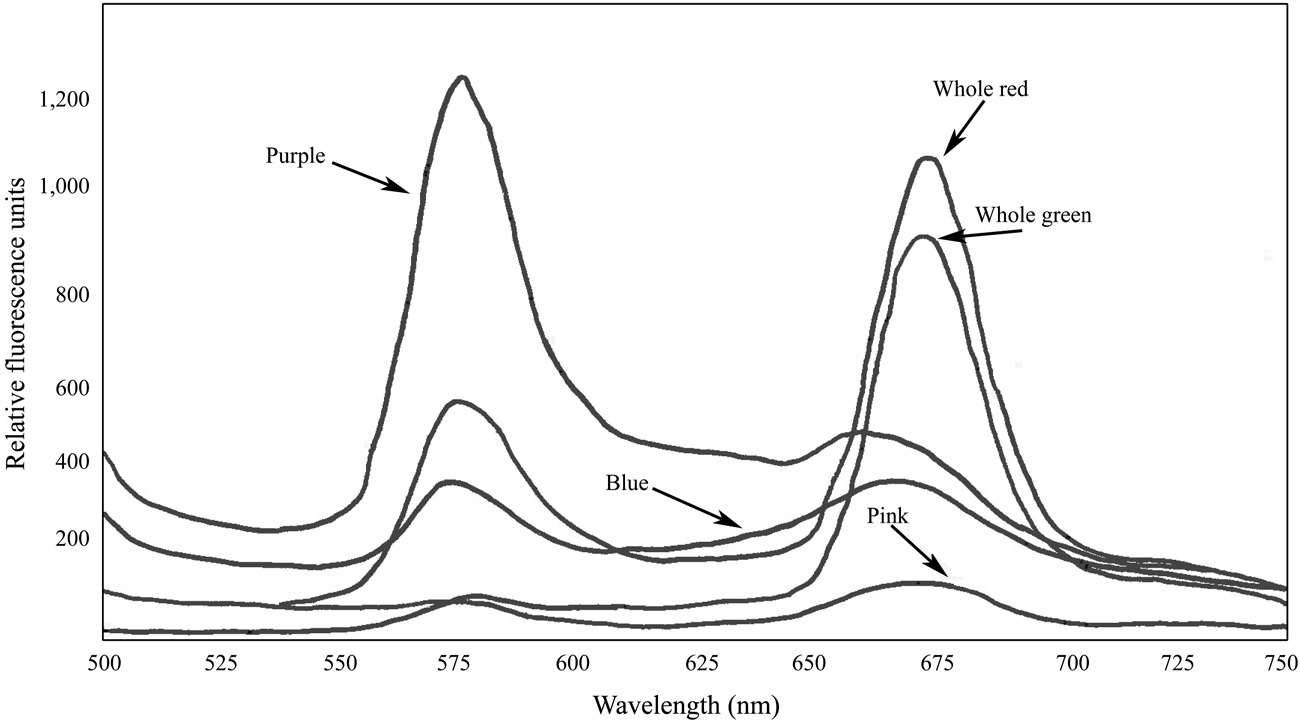
Fluorescence spectra of the whole crude extracts indicated the presence of functional PBS in the samples of both
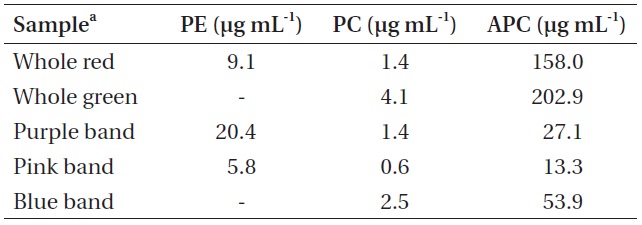
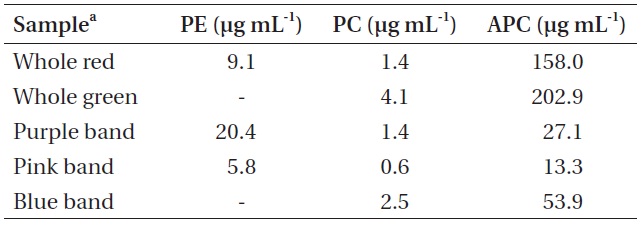
Pigment concentrations in whole crude Chondrus crispus extracts and in samples of each isolated pigment band
which can cause PBS to dissociate. Fluorescence spectra of the pigmented bands indicate that the isolated PBS were mostly dissociated, although there was some fluorescence around 670 nm in all cases, suggesting that a relatively small number of intact PBS were still present (Fig. 5). While phycoerythrin content at 9.10 μg mL-1 appeared to be present in a substantial amount in the whole extract from the red
Pigment content calculated for the coloured bands indicated that the purple band extracted from the red
The whole red extracted samples had a thin pool of carotenoids on top of the sucrose gradient, followed immediately below by a green chlorophyll zone. Below this was a narrow section of clear area, followed by a zone of pale pink, concentrated mostly in the 0.5 M sucrose segment. It is expected that the primary constituents of this pinkish zone were bits of PBS debris and dissociated PE components. The purple band isolated from the red
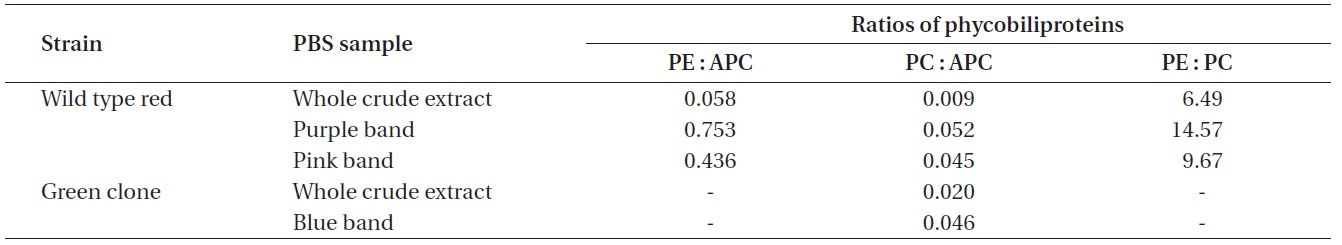
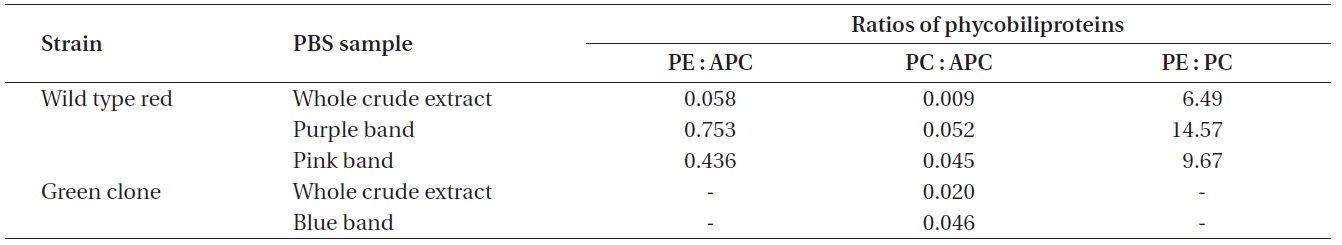
Ratios of the phycobiliproteins isolated from wild type red Chondrus crispus and its green mutant
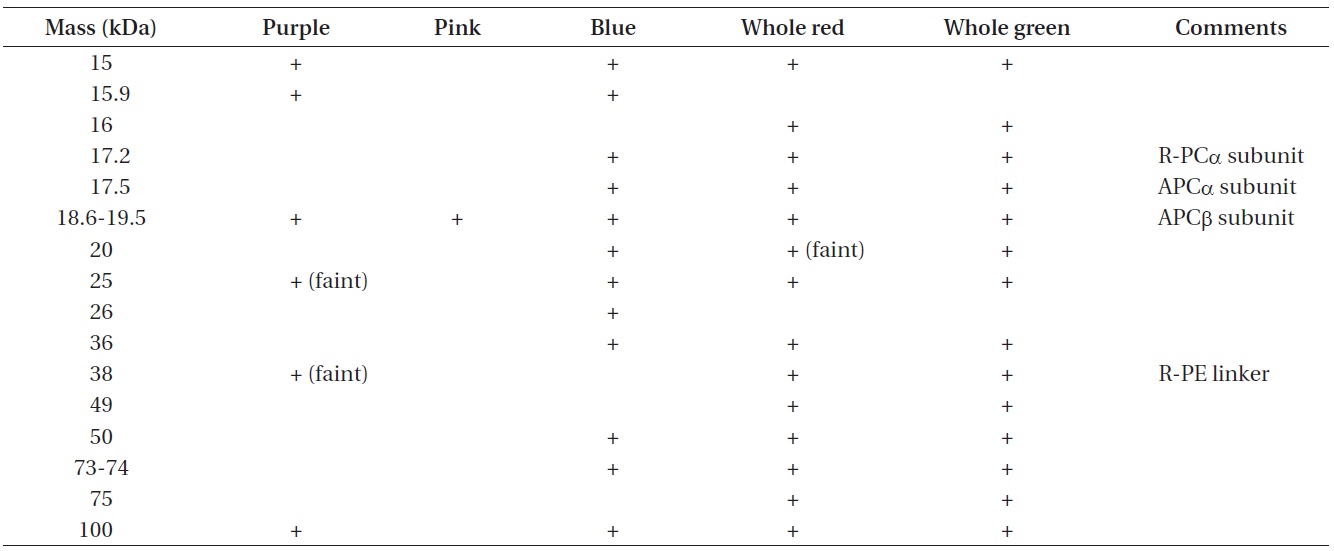
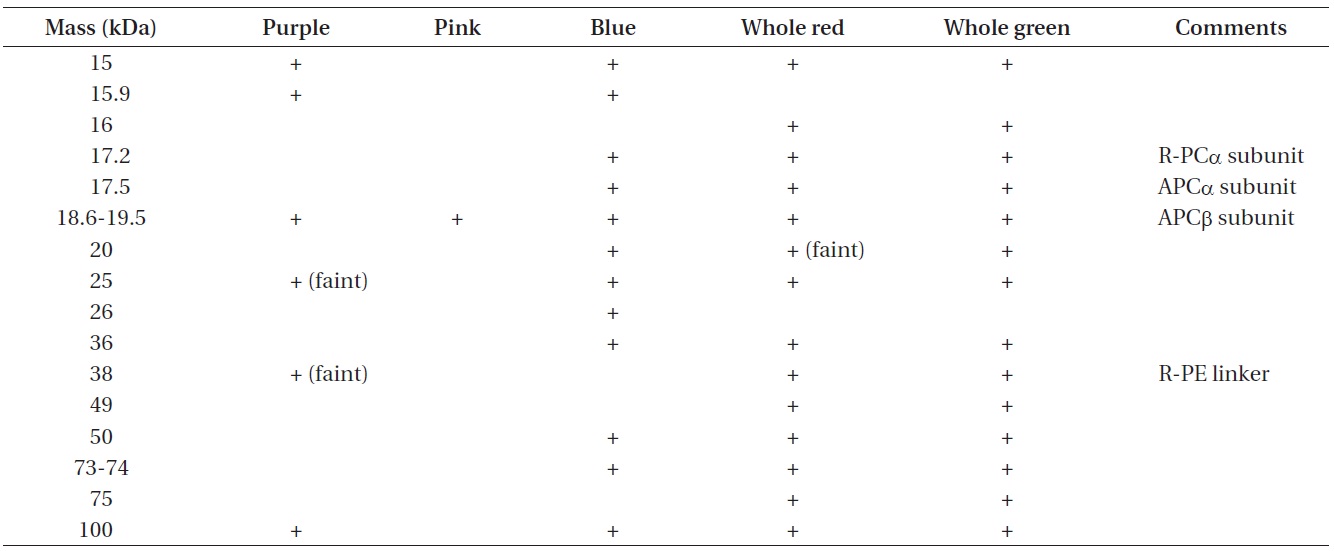
Apparent molecular masses of phycobiliprotein subunit complexes from phycobilisome bands isolated from wild type red Chondrus crispus and its vegetative green mutant
whole red extract at 6.490 PE to PC. With the exception of the whole extracts, all the pigment samples had a PC to APC ratio between 0.045 and 0.052. Dry matter content of the frozen algal samples was not measured and the PBS extractions were not quantitative, therefore the pigment measurements cannot be related back to the original tissue concentrations.
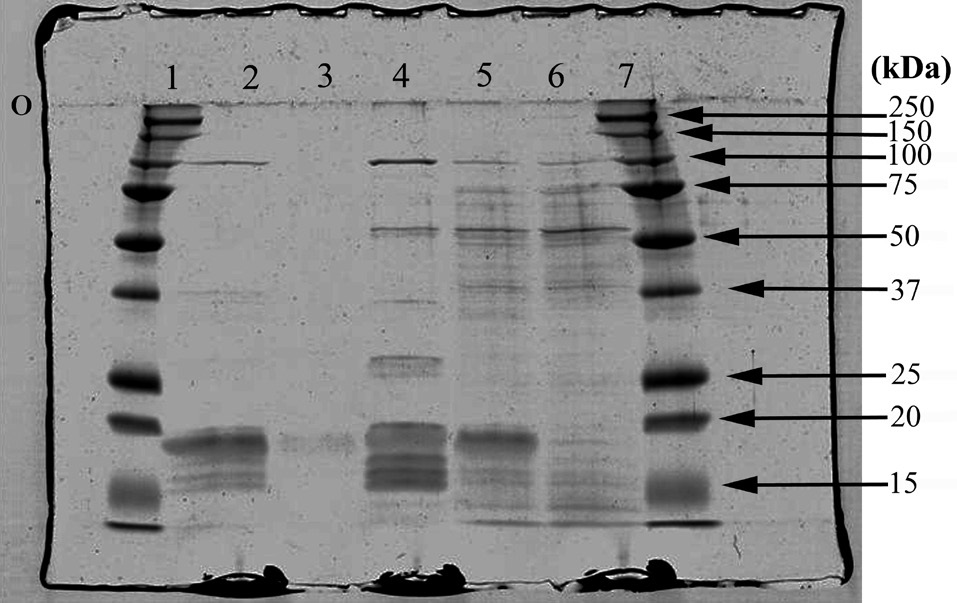
The apparent molecular masses of the dissociated constituents of the protein complexes of all samples are listed in Table 3. The results of the SDS-PAGE analysis further demonstrate some differences among the phycobiliprotein subunit complexes in the extracts collected (Fig. 6).
The use of colour mutants has proved a valuable tool in red algal biology (see review by van der Meer 1990). Such mutations have played an important role in uncovering the details of red algal life histories and development, in particular in the genera
The composition of red algal PBS typically consists of an APC core and several peripheral rods containing PC or PC/PE (Arteni et al. 2008, Su et al. 2010
The linker polypeptides are believed to mediate the assembly of PBP into highly ordered arrays within the PBS, and to facilitate the interactions between the PBS and the thylakoid membrane (Liu et al. 2005). With limitations on the amount and / or structure of PE, the green mutant would require an alternate means of energy capture and utilization. The purple PBS band from the wild type red
Unlike the two distinct pigment zones isolated by Luder et al. (2001) from
Not all the phycobiliprotein subunits and structural components such as linker polypeptides have been identified in the samples, but by making comparisons to data collected by others, some potential explanations can be proposed. Of particular interest is the polypeptide that seems to appear in the blue band only, with a molecular mass around 26 kDa. Liu et al. (2005) have identified a Rod-core linker peptide at 26.8-31.9 kDa. Furthermore, both the blue and the pink extracts appear to be missing clear evidence of an important R-PE linker protein, which suggests the PE in the pink band may be partially dissociated. This is consistent with Algarra et al. (1990) and Luder et al. (2001) who proposed an enhanced acclimation strategy in some red algae, facilitated by a pool of PE proteins present in an intermediate state of assembly.
SDS-PAGE analysis was performed on samples after washing and concentrating them numerous times to minimize potential contaminants, such as Triton-X and sucrose. Attempts to wash and concentrate the pink-pigmented band may have contributed to some PBP losses, since this band had been isolated in the densest layer of the sucrose gradient, relative to the others, and it demonstrated significant PBS dissociation. Consequently, the sample protein from the pink band analyzed for subunit complexes was quite dilute, although this should not negate the possibility that the pink zone consisted of a pool of PE in a state metabolic transition.
Kursar et al. (1983) found that colour mutants of
The specific phycobilisome composition of the