With space exploration, there is increasing concern about cosmic radiation exposure while astronauts are carrying out various missions, and many efforts have been made to secure the safety of the astronauts (Vetter et al. 2002, Lindborg et al. 2004). According to the data from National Aeronautics and Space Administration (NASA), it is known that the exposure is about 4.3 mSv when a single mission is completed using a space shuttle, and for the Skylab 4 mission, the exposure is reported to be maximum 178 mSv. This is about 4~180 times more irradiate dose than the permissible annual dose of the general public (1 mSv) recommended by the International Commission on Radiological Protection (Alexakhin et al. 2007). This is also a considerably high dose when compared to the permissible annual dose of radiation workers in Korea (50 mSv).
Cosmic radiation is broadly divided into galactic cosmic ray, solar cosmic ray, and the radiation captured in the Earth’s magnetic field, and consists of mostly protons (90%) and second mostly electrons (Sakaguchi et al. 1999, Keith et al. 1992). Therefore, the radiation exposure of astronauts is typically due to the protons or the neutrons and heavily charged particles which are the secondary radiation generated by protons. Unlike the ground, the proportion of charged particle radiation having high linear energy transfer (LET) is large, and thus the biological effects from radiation exposure are also large. In addition, the radiation environment changes significantly due to the solar flare explosion or solar wind, and the radiation shield effect from the atmosphere is insignificant. For evaluating the biological effects from radiation exposure, the equivalent dose needs to be applied which is the product of absorbed dose and radiation weighting factor. As the radiation weighting factor is determined by the LET of radiation (Alexakhin et al. 2007), the evaluation of LET is critical for monitoring the radiation in the space. Various kinds of detectors such as Bonner sphere, Sievert counter, thermo luminescence dosimetry (TLD), and solid state nuclear track detector (SSNTD) are used to evaluate an equivalent dose in the space. Among them, the tissue equivalent proportional counter (TEPC) developed by Rossi & Rosenzweig (1955) and diverse types of detectors which modified this counter are commonly used (Taddei et al. 2006, Kunz et al. 1990). The TEPC can measure an absorbed dose directly as the TEPC is equivalent to human tissue, evaluate an equivalent dose by measuring the LET spectrum, and measure a micro volume dose at the level of a cell or a blast cell by adjusting the site diameter using the gas pressure inside the detector.
In this study, to monitor the equivalent dose in the International Space Station, a spherical TEPC was developed which can measure the LET of cosmic radiation, and the characteristics of the detector were evaluated.
2. DESIGN OF TISSUE EQUIVALENT PROPORTIONAL COUNTER
2.1 Tissue Equivalent Material and the Structure of Detector
To directly evaluate the equivalent dose of a human body, a detector should be used which has the same scattering and absorption characteristics as those of a human body regarding radiation, and for this purpose, the material for the detector needs to be the tissue equivalent material.
The tissue equivalent material, A-150 was used for the wall of the TEPC developed in this paper, and the tissue equivalent gas with a mass ratio of C3H8 (55%) + CO2 (39.6%) + N2 (5.5%) was used for the internal detection gas. The A-150 tissue equivalent material has a mass ratio of hydrogen 10.2%, carbon 77.6%, oxygen 5.2%, etc.
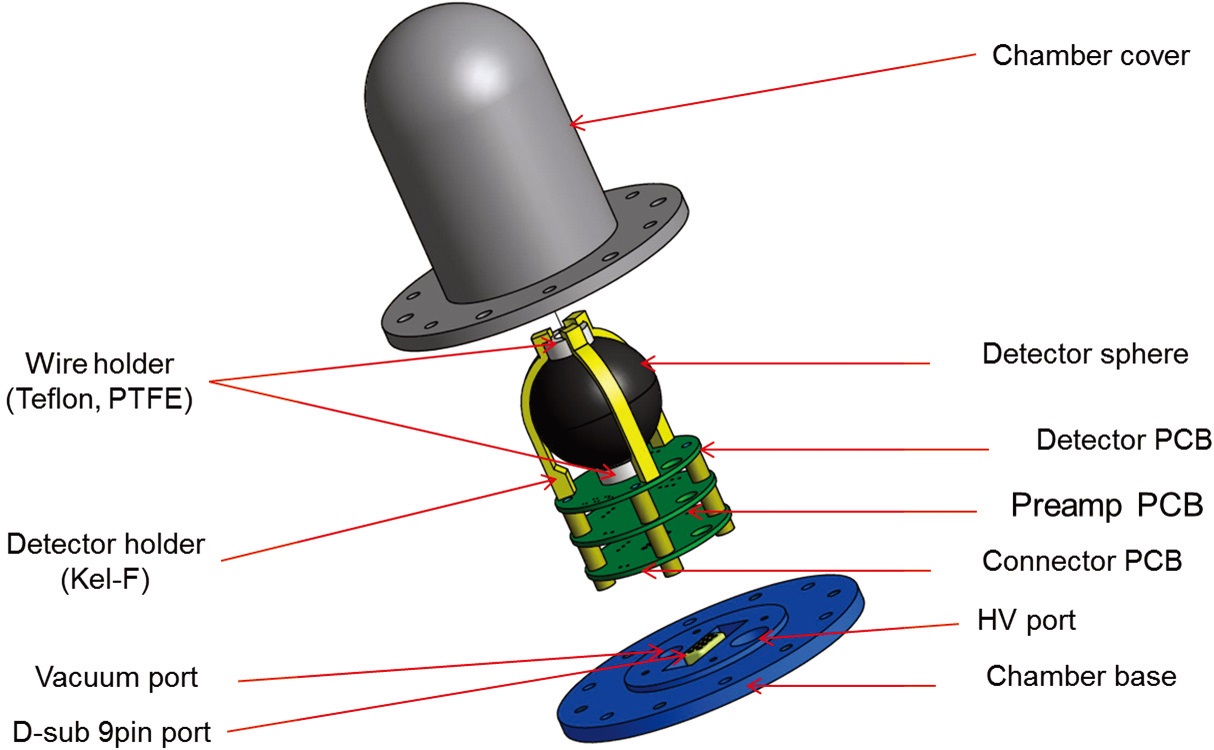
Fig. 1 shows the drawing of proportional counter and housing which were designed using the tissue equivalent material. The outer diameter of the detector is 40 mm, and the inner diameter is 30 mm. The detector was designed to have a spherical shape to minimize the dependence on the incident direction of radiation. The wall of the detector is the A-150 tissue equivalent material with a thickness of about 5 mm, and the center electrode is a gold-coated tungsten wire with a diameter of 10 μm. The spherical detector is fixed at 3 holders (KeI-F), and the detector circuit unit for signal detection consists of 3 printed circuit boards (PCB) which constitute the high-voltage unit and pre-amplifier unit for detector and the terminal unit for external interface. The outer housing was designed using SUS304 material with a diameter of 54 mm and a thickness of 1.5 mm, which protects the tissue equivalent proportional detector and maintains a detection gas. For the outer housing, if the
housing is too thick, the measurement value decreases as the incident radiation is shielded. If the housing is too thin, the physical strength is weak, and the measurement value also decreases as the secondary charged particles decrease.
2.2 Simulation of Site Diameter
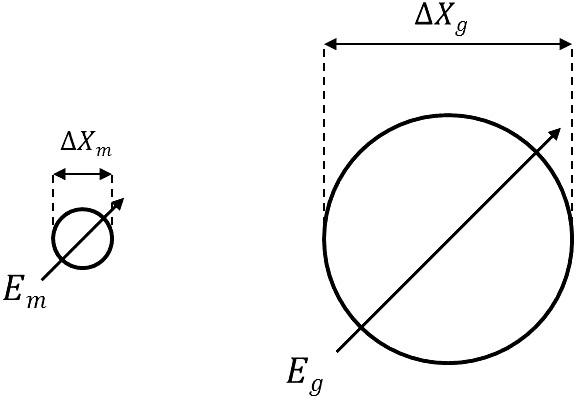
When a human body is exposed to radiation, chromosome mutation occurs within a cell through complicated processes. As it is important to analyze the effect of radiation on chromosome structure, the radiation energy, that is transferred to several μm distance which corresponds to the size of a cell, needs to be defined, and the relevant dose measurement is required. This is called microdosimetry, and can be defined as the linear energy transfer for micro volume in the structure of a cell or a germ cell. To evaluate the dose for a human body, the volume and gas pressure of measuring instrument, which correspond to the micro volume, need to be determined (Burmeister et al. 1999). Fig. 2 shows the conceptual diagram of site diameter simulation. In this study, for the purpose of microdosimetry, the site diameter (ΔXg) was chosen to be 2 μm. To achieve this condition, the gas pressure inside the measuring instrument was determined to be 27.76 torr based on the calculation result for the tissue equivalent gas inside the detector using Eq. (1).
where the inner diameter of TEPC (ΔXm) was 3.0 cm, the density of tissue was 1.0 g/cm3, and the density of tissue equivalent gas was 1.17×10-5 g/cm3 at 295 K. M is the molecular weight of measurement gas, and R is the gas
constant (8.32 J/mol-K).
3.1 Manufacture of Tissue Equivalent Proportional Counter
Fig. 3 is the photograph of the manufactured TEPC and stainless detector housing. Three PCBs are shown including the high-voltage distribution circuit, pre-amplifier unit, and signal connector which are installed at the lower part of the spherical detector.
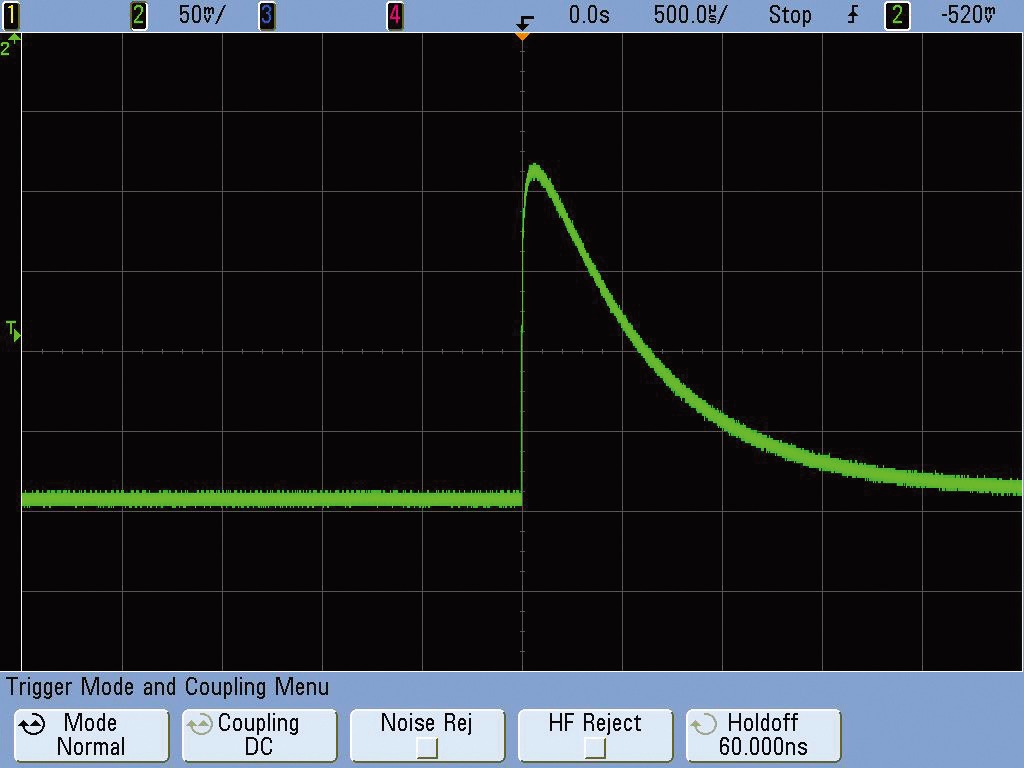
As the TEPC developed in this paper will be used for monitoring radiation in the International Space Station in the future, all kinds of electronic circuits, power supply, and data communication regulations should accord with the NASA standard (Mulville 1998). Therefore, to minimize the noise of detector signal, the voltage divider circuit and pre-amplifier for the high-voltage supply of the detector were installed inside the detector. For the pre-amplifier, considering the noise of detector, the A250 (charge sensitive pre-amplifier) of Amptek Company was used which utilizes the field effect transistor on the outside. Fig. 4 shows the signal outputted from the pre-amplifier which was measured with an oscilloscope.
To investigate the basic characteristics of TEPC, for the output of pre-amplifier, the signal was amplified using the 571 shaping amplifier of ORTEC Company, and the linear energy spectrum was obtained using the MCA8000A Multi- Channel Analyzer (MCA) of Amptek Company.
3.2 Characteristics of Tissue Equivalent Proportional Counter
To determine the operating voltage condition and linear energy-channel calibration factor of the manufactured
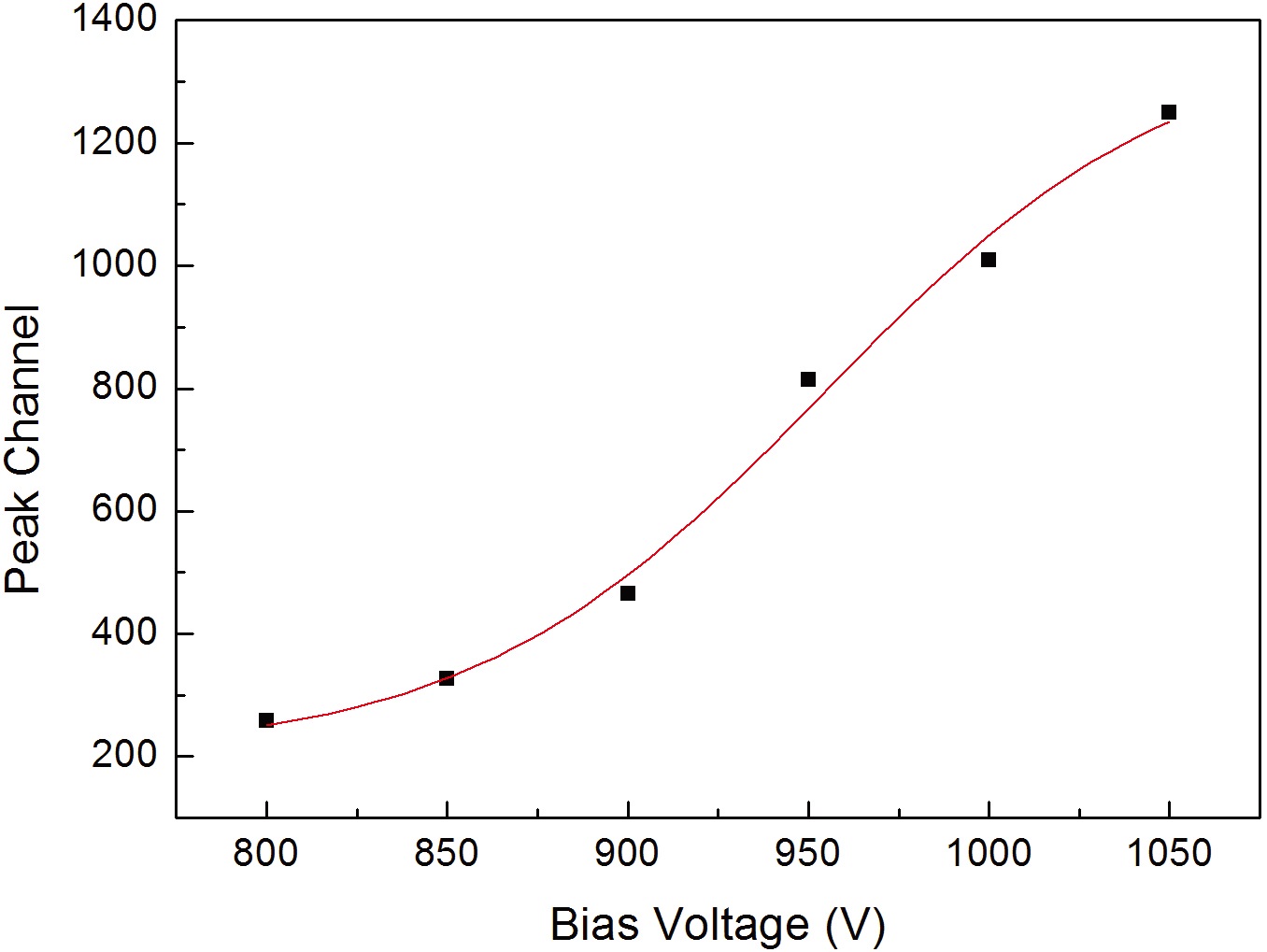
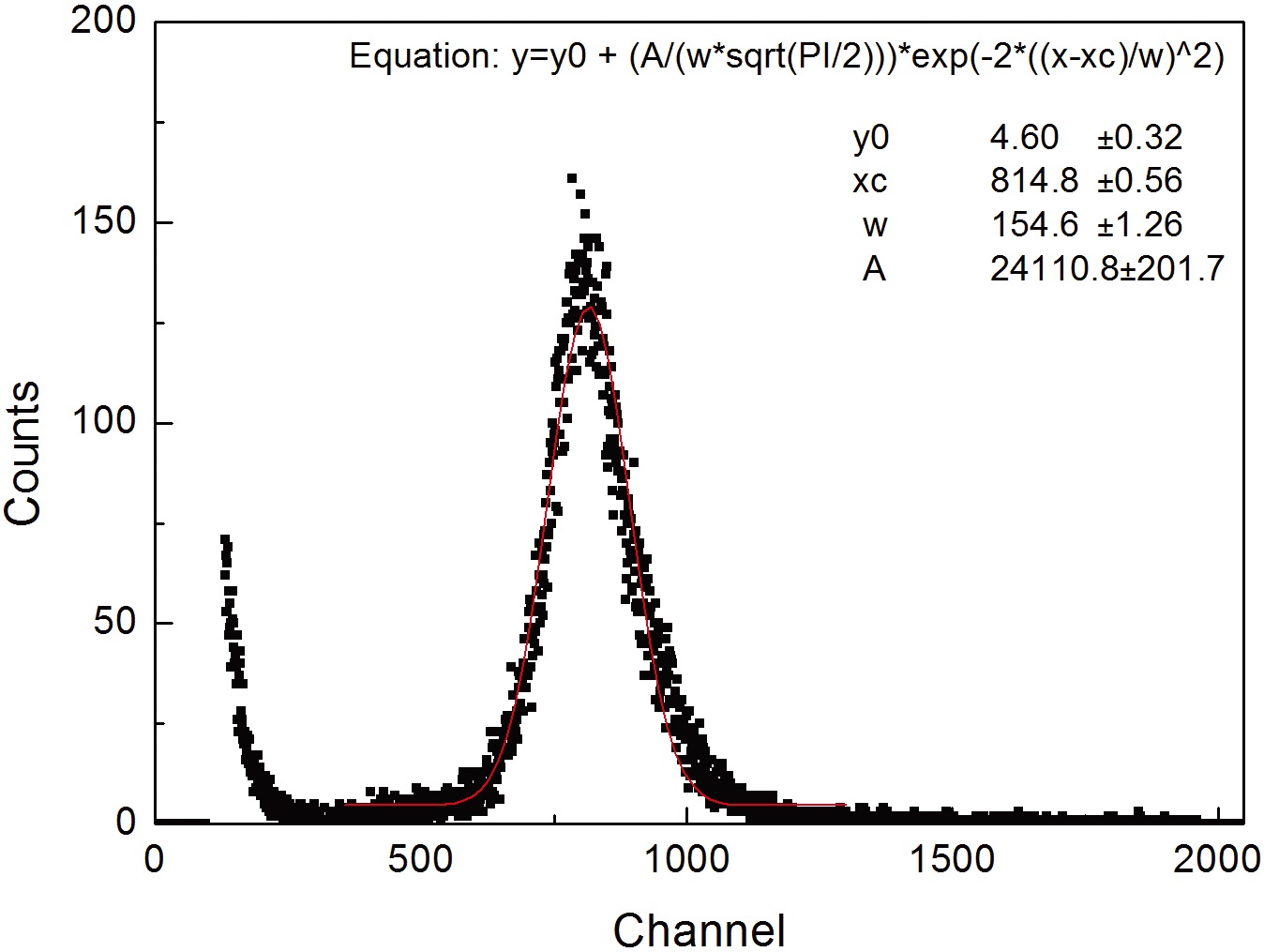
TEPC, the 241Am α-ray source was inserted inside the detector, and the linear energy spectrum for 5.5 MeV α-ray was measured. Fig. 5 shows the peak channel for the 241Am 5.5 MeV α-ray which was measured while changing the applied voltage. As shown in Fig. 5, the peak channel increases as the applied voltage becomes higher. As the measurement range of the measuring instrument is 0.2~300 keV/μm, the appropriate applied voltage was determined to be 950 V considering the linear energy of α-ray. Fig. 6 shows the linear energy spectrum of the 241Am 5.5 MeV α-ray which was measured at the applied voltage at 950 V. In Fig. 6, the dot represents the factor for each channel of MCA and the solid line represents the result of the Gaussian fitting for the measured value, and the linear energy resolving power for 5.5 MeV was about 23.5%. For the developed TEPC, the internal calibration method using the α-ray source for calibrating the channel to linear energy and the external
calibration method using 252Cf neutron were carried out.
3.2.1 Linear energy-channel calibration using the 241Am α-ray source
Fig. 7 shows the stopping power which was calculated through the Geant 4 simulation to examine the response of the TEPC developed in this paper for the energy of 241Am α-ray. Based on the simulation result, the linear energy that the 241Am average energy 5.5 MeV α-ray assigns to the TEPC geometry was 173 keV/μm. According to microdosimetry, the linear energy (y, keV/μm) is the energy assigned to micro space divided by the mean chord length (
channel (I) of MCA is determined by the following equation.
Eα: α-ray energy allocation for the tissue equivalent, 173 keV/μm
Iα: Linear energy peak channel of α-ray shown on the MCA
When the applied voltage was fixed at 950 V, the linear energy per channel determined by Eq. (2) was 0.15 keV/μmch. If the MCA channel is extended to 2,048, it is possible to measure up to 326 keV/μm. Thus, it was demonstrated that the linear energy can be measured from 0.2 to 300 keV/μm which is the target measurement range.
3.2.2 Determination of dose calibration factor using the 252Cf neutron standard source
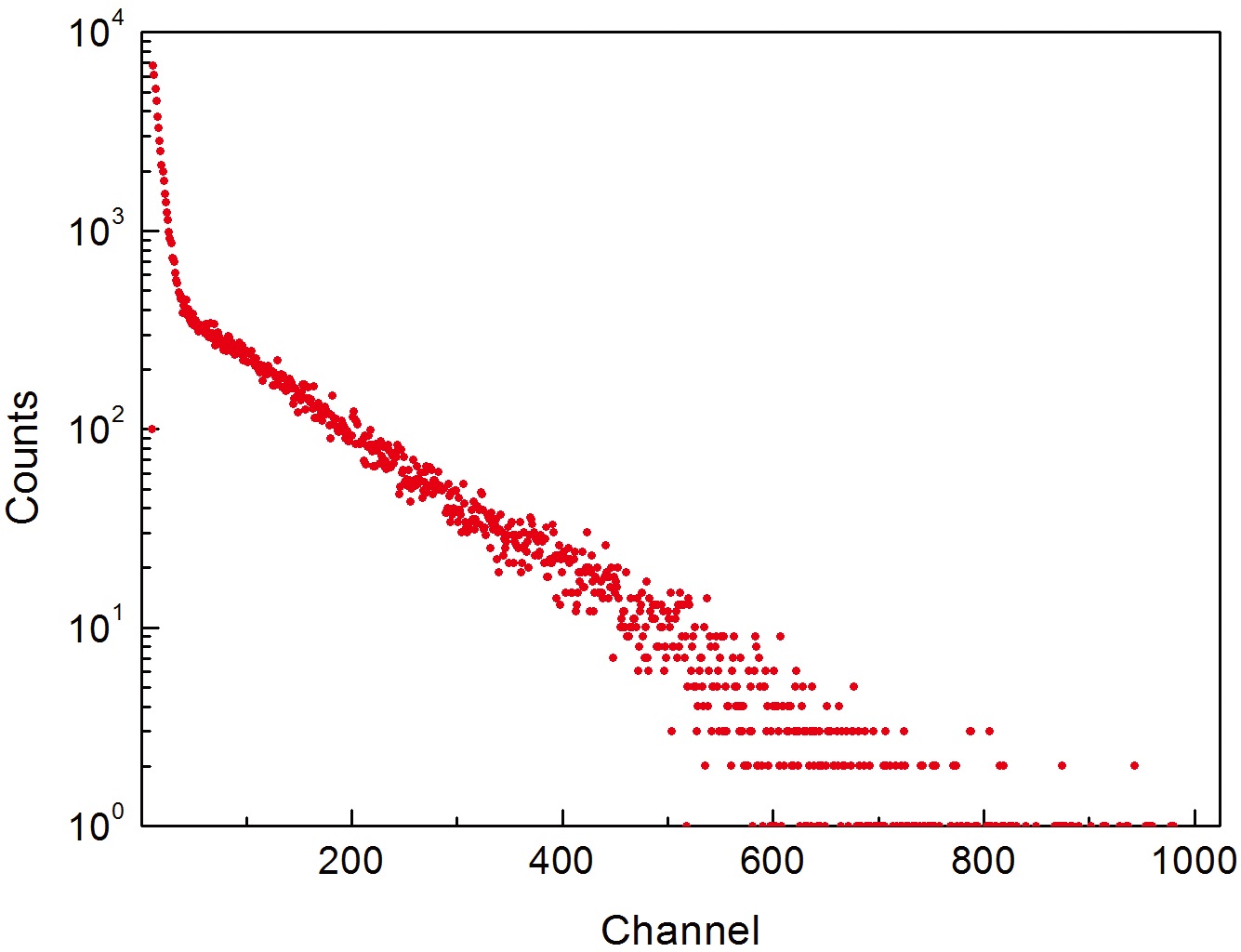
For the developed TEPC, the dose calibration factor was determined using the 252Cf neutron standard source of the Korea Research Institute of Standards and Science. Fig. 8 shows the LET spectrum of 252Cf neutron source which was measured with the developed TEPC. The 252Cf neutron source of the Korea Research Institute of Standards and Science is the standard neutron source with a neutron flux of 2.36×108 cm-2s-1 (calibrated on November 9, 2012), and the dose rate at the measurement point was 53.9 mSv/ hr. For the measurement, the applied voltage was 950 V, the gain was 100, the peak shaping time was 3 μs, and the measurement time was 20 minutes. As shown in Fig. 8, the detection gas is tissue equivalent material which has a small absorption cross section for neutron and has a low gas pressure. Therefore, the 2 MeV neutron peak of 252Cf was not
observed, but the channel could be calibrated by identifying the proton end point near the 600 channel.
Eqs. (3) and (4) were used for the dose calibration factor, and the equivalent dose for unknown source can be determined through the calibration.
where, H is the equivalent dose, D is the absorbed dose, Q is the quality factor, R is the charge distribution of pulse, h is the channel, n(h) is the counting rate for each channel, and q(h) is the quality factor for each channel. kf is the calibration factor of TEPC and can be determined by the relation of Eqs. (2) and (3) (Dietze et al. 1984). The determined calibration factor was 3.59×10-7 mSv/R.
A prototype TEPC for monitoring the equivalent dose in the International Space Station was developed and tested. For the TEPC detector, the site diameter for microdosimetry was designed to be 2 μm. The energy channel was calibrated using the α-ray source of the designed detector, and based on this result, it was demonstrated that the energy can be measured from 0.2 to 300 keV/μm with a single MCA channel, which is the target measurement range. Also, the calibration factor was determined using the 252Cf standard source of the Korea Research Institute of Standards and Science. These experimental results will be reflected in the detailed design of TEPC for monitoring the equivalent dose in the International Space Station, and especially the basic results for the low power design and high density integration of electronic circuits were obtained. In the future, the detailed design standard of electronic unit will be developed following the design standard of NASA.
The TEPC developed in this paper can directly measure the equivalent dose of a human body, and is capable of realtime monitoring in a high LET radiation field. Therefore, the TEPC is expected to be used for the dose rate evaluation in the places where various kinds of radiation exists such as nuclear power plant as well as in the space radiation environment of the International Space Station.