In this study, the treatability of two target contaminants during the Fenton reaction, n-hexadecane and 1-methylnaphthalene, was evaluated as a function of the amounts of FeCl2 and H2O2 injected into open and closed reaction systems. In the Fenton reaction of nhexadecane and 1-methylnaphthalene, the mass recovery of the target contaminants was above 95% in the closed system. However, when the Fenton reaction was performed with high amounts of H2O2 and FeCl2 injected in the open system, a reduction of approximately 40% of the initial mass of 1-methylnaphthalene was observed. This trend may be explained by the unique physical properties of 1-methylnaphthalene, which has higher volatility than n-hexadecane. Further, this trend was well correlated with the rise in high temperature at the initial reaction stage. Considering the mass recovery of the two target contaminants, the reaction temperature, and the residual concentration of H2O2 at different amounts of FeCl2 and H2O2 injected, it can be suggested that the Fenton reaction should be performed with controlled conditions that can provide a suitable reaction environment between oxidant and contaminants.
Petroleum products such as gasoline and diesel obtained from crude oil are the most important fuels in a modern industrialized society. Petroleum-derived pollutants generated in the process of manufacturing these products pollute the soil and groundwater [1,2]. Petroleum-derived pollutants are characterized by their broad range and frequency of occurrence, and are accordingly considered to be a serious issue.
Methods of treating polluted soil or waste water include biological restoration technology, physical treatment technologies such as soil steam extraction methods, soil cleansing technology, as well as other technologies, such as incineration, and thermal desorption technology. In soils composed of silt or clay with small particle sizes, there is a limitation in the application of physical treatment technology due to the small size of the crevices and large surface area. In addition, the thermal desorption and incineration technologies not only have application limitations in terms of economic value, but also have problems with generating secondary pollutants during the treatment process [3].
Recently, advanced oxidation processes, such as ozone or Fenton oxidation reaction, have been carried out actively in order to supplement the shortcomings of physical treatment technology and thermal desorption technology. Among them, Fenton oxidation reaction, which oxidizes organic matter using iron salt and hydrogen peroxide, has been used frequently. However, there is insufficient technological data for the application of this process in the remediation of actual pollution sites, since the disintegration mechanism of diesel during Fenton oxidation has not been definitively revealed [4]. Therefore, there is a need to identify how much oxidation or disintegration of diesel actually occurs during the Fenton reaction in specific conditions. The Fenton reaction has widely been applied in the oxidation of numerous water-soluble waste matters [5-11]. Barbeni et al. [5] examined and recorded that chlorophenyl is disintegrated into CO2, H2O, and chloride. Zazo et al. [12] suggested a removal mechanism for phenol oxidation by the Fenton reaction. Although phenol can be easily oxidized into other compounds due to high reactivity, aromatic hydrocarbons with functional groups composed of hetero-atoms do not disintegrate into H2O or CO2 [13]. Therefore, in order to oxidize hydrocarbons, catalytic agents and heating must be applied. In this reaction, hydrocarbons can be transformed into other compounds, rather than producing CO2 and H2O. As revealed in the literature, highly severe conditions are necessary to oxidize hydrocarbons and disintegrate them into CO2 and H2O. N-hexadecane and 1-methyl naphthalene are representative substances that are difficult to remove
with oxidation reaction. Very high pressure and/or temperature, in addition to high-priced catalytic agents, are necessary to oxidize these substances. Furthermore, even if oxidation occurs, complete conversion into CO2 and H2O may not occur.
The objective of this study was to evaluate the treatability of two target contaminants, n-hexadecane and 1-methylnaphthalene, during the Fenton reaction in open and closed reaction systems. In order to identify any generation of volatile organic products in addition to the vaporization of the two target contaminants due to high temperature during the Fenton reaction, reaction was performed in open and closed system. Any oxidation or disintegration of these two target contaminants was evaluated through quantitative analysis of the residual organic compounds using gas chromatography (GC) and qualitative information obtained by infrared spectrometry (IR) and GC-mass selective detector (MSD) before and after the Fenton reaction.
In field-contaminated soils, organic substances may exist in addition to the total petroleum hydrocarbon (TPH) substances. These organic substances can compete with TPH substances in the reaction with Fenton’s reagent. Thus, in order to eliminate this possibility, sea sand (Junsei Chemical, Tokyo, Japan) with a particle diameter of 20?30 mesh was used as an artificial soil. The results of the elemental analysis of the sea sand are summarized in Table 1. Fig. 1 shows a picture of the sea sand. N-hexadecane (Sigma-Aldrich, St. Louis, MO, USA) and 1-methylnaphthalene (Alfa Aesar, Ward Hill, MA, USA), important ingredients of diesel oil, were used as model pollutants [13,14]. The initial concentration of each pollutant in the sea sand was set at 50,000 ppm by adding 2.5 g of the pollutants into 50 g of sea sand.
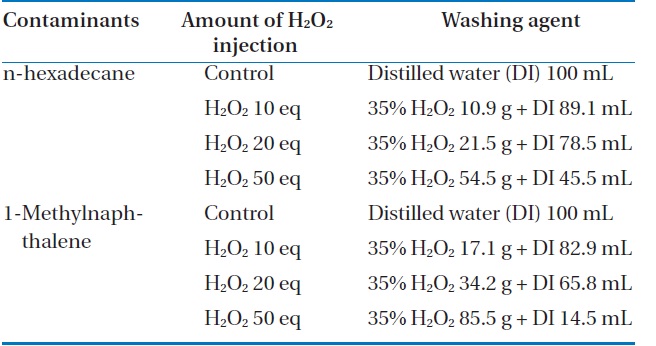
In the experiment, 50 g of artificially polluted soil was placed in individual 500-mL Erlenmeyer flasks, and 1,000, 3,000, 5,000, and 20,000 ppm of FeCl2 (0.05, 0.15, 0.25, and 1 g/50 g soil) were added into the respective flasks, and mixed thoroughly. Then, the pH was adjusted to 3.0 using 1 N H2SO4. Solutions with concentrations of hydrogen peroxide, including a control, 10, 20, and 50 equivalent concentrations in 100 mL were prepared. Each experiment was carried out for 1 hr in both an ordinary open beaker as an open system, and in a reflux condenser as a closed system. The details of the experimental conditions are summarized in the Table 2.
After Fenton reaction, the residual concentrations of hydrogen peroxide, n-hexadecane, and 1-methylnaphthalene were measured. N-hexadecane and 1-methylnaphthalene analysis was carried out with the method presented in the soil contamination process test [15]. An Agilent HP 7890 GC (Agilent, Santa Clara, CA, USA), an flame ionization detector (FID), and a 30-m DB-TPH capillary column were used for GC analysis. The initial oven temperature of 50℃ was maintained for 3 min and then increased at 10℃/min. The final oven temperature of 290℃ was maintained for 33 min. The temperature of the inlet section was set to 290℃, while that for the detector was set to 300℃. A constant flow of helium was maintained at 1.5 mL/min. An HP 6890 GC-mass spectrometry (MS), an Agilent HP 5975 quadrupole mass spectrometer, and a 60-m INNOWax capillary column were used for GC-MS analysis. The initial oven temperature of 50℃ was maintained for 3 min, and then increased at 10℃/min. The final oven temperature of 260℃ was maintained for 23 min. The temperature of both the inlet section and the detector were set to 260°C while maintaining a constant flow of helium at 1.5 mL/ min. The residual hydrogen peroxide was measured using an iodometric method appropriate for oxidation-reduction. After the extraction of n-hexadecane and 1-methylnaphthalene by dichloromethane (SK Chemicals, Seoul, Korea), Fourier transform infrared (FT-IR) spectroscopy (Nicolet 380; Thermo Scientific, Waltham, CA, USA) was employed for analyzing two compounds qualitatively and quantitatively.
[Table 1.] Element analysis of sea sand used in this study

Element analysis of sea sand used in this study
[Table 2.] Experimental conditions by contaminants and amount of H2O2 injection
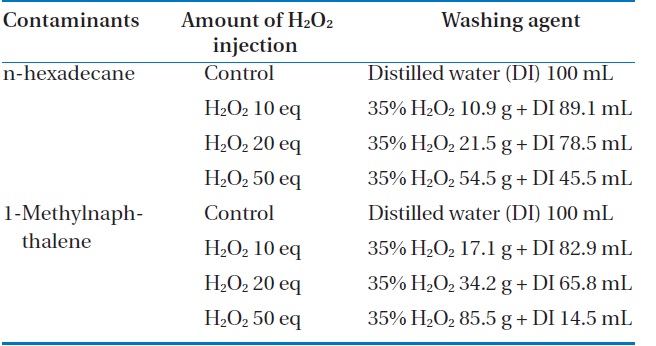
Experimental conditions by contaminants and amount of H2O2 injection
3.1. Removal of N-hexadecane in Closed and Open Systems
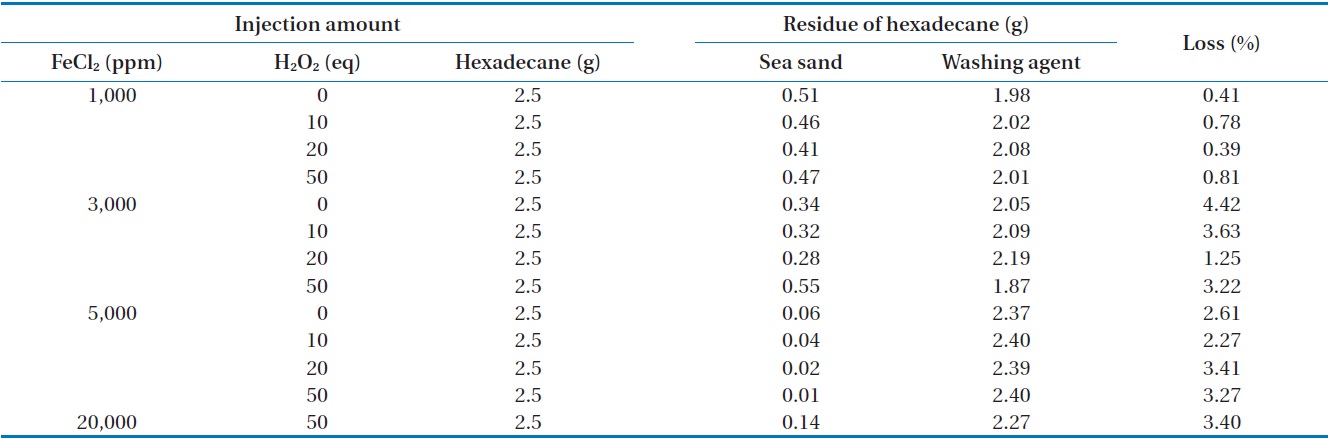
The removal of n-hexadecane as a function of the amounts of FeCl2 and H2O2 injected was investigated in closed and open systems. The measured concentration of n-hexadecane in sea sand and washing agent at different amounts of FeCl2 and H2O2 in a closed system is illustrated in Table 3. For all conditions, the mass recovery of n-hexadecane was above 96% of the initial concentration of n-hexadecane, suggesting that most of the n-hexa-
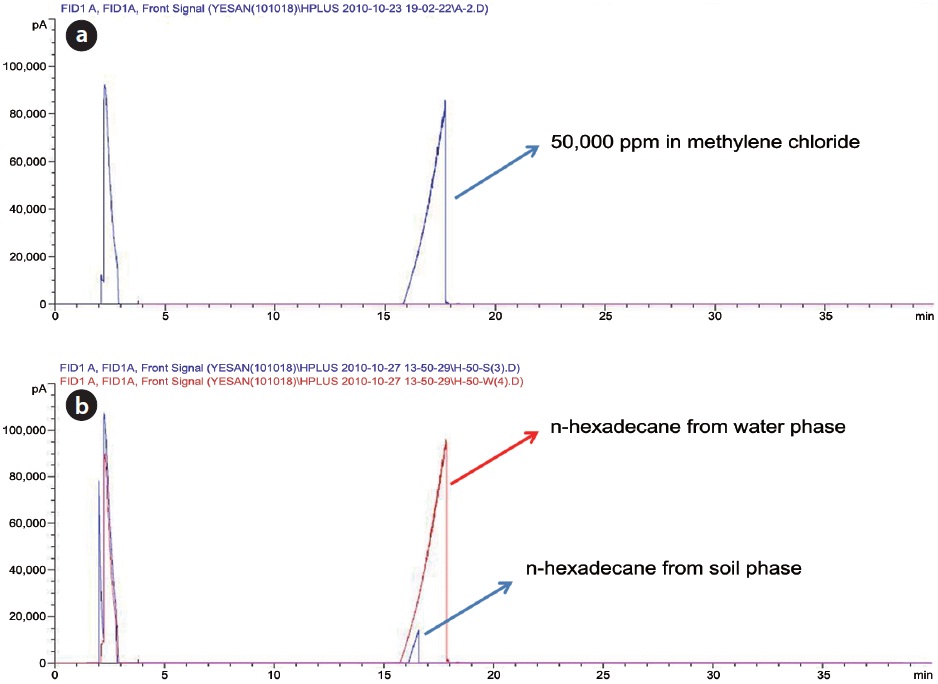
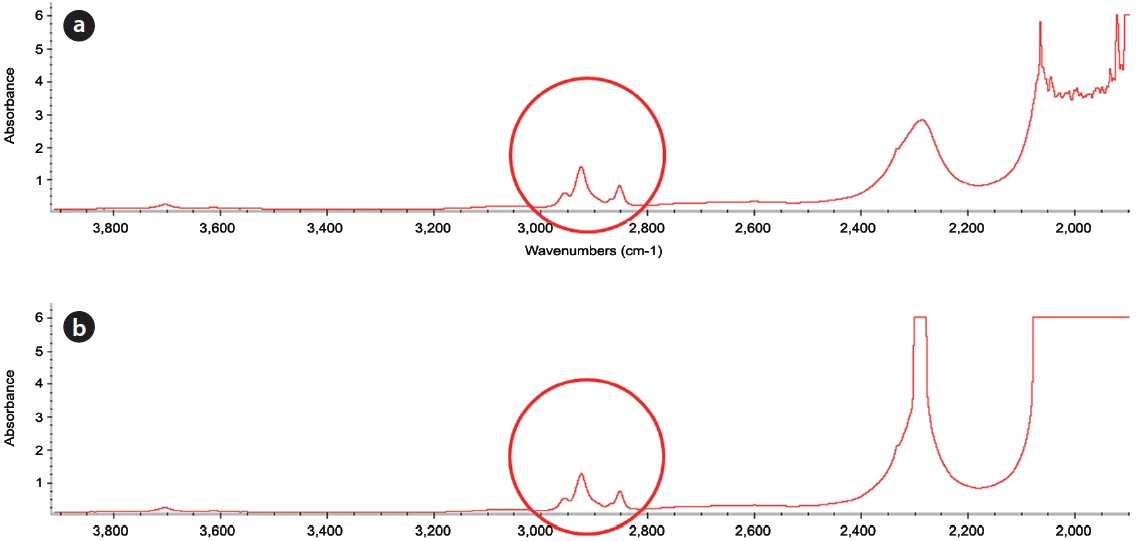
decane exists in its original form. In order to find any oxidation products of n-hexane during Fenton reaction, the reaction samples were analyzed by GC (Fig. 2) and FT-IR (Fig. 3). In this analysis, the major peaks observed in GC analysis before and after Fenton reaction were quite similar in terms of retention time and peak area. In addition, three peaks between 2,800/cm and 3,000/ cm in the FT-IR spectrum were also quite similar. There are several reasons to explain this result. First, the reaction conditions applied in this work were not sufficient to disintegrate the two model compounds. Second, no important oxidation products could be produced due to the limited contact between the original hydrophobic chemicals and hydrophilic oxidant. Lastly, general analytical methods cannot be used to detect any oxidation products. Chen et al. [16] reported that Fenton’s reagent reacts rapidly with n-hexadecane in soil, and complete mineralization of n-hexadecane was observed when enough hydrogen peroxide was added. However, they did not find any by-products by GC analysis. Jerkovic et al. [17] reported that oxidation products can
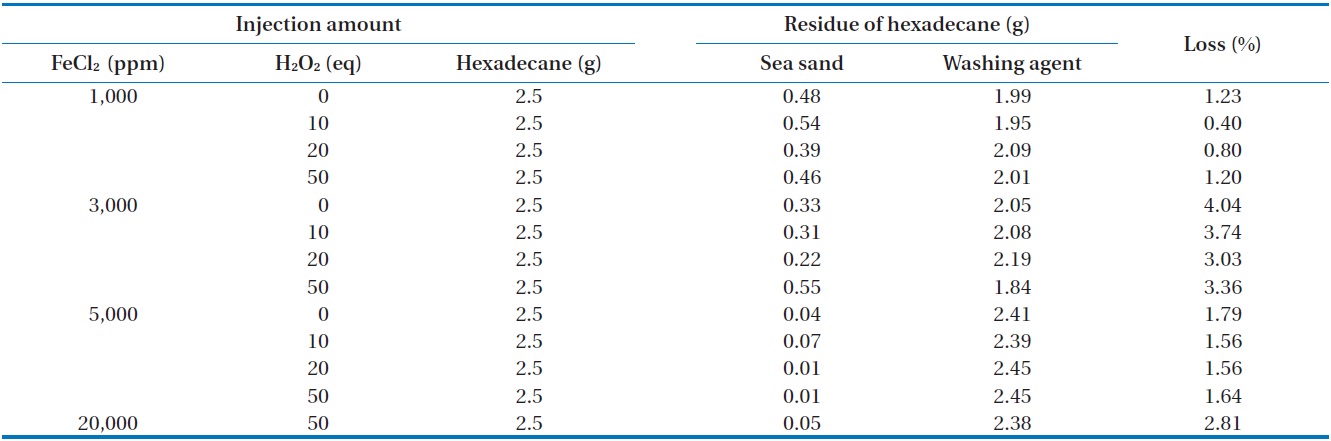
[Table 3.] Removal of n-hexadecane in closed system
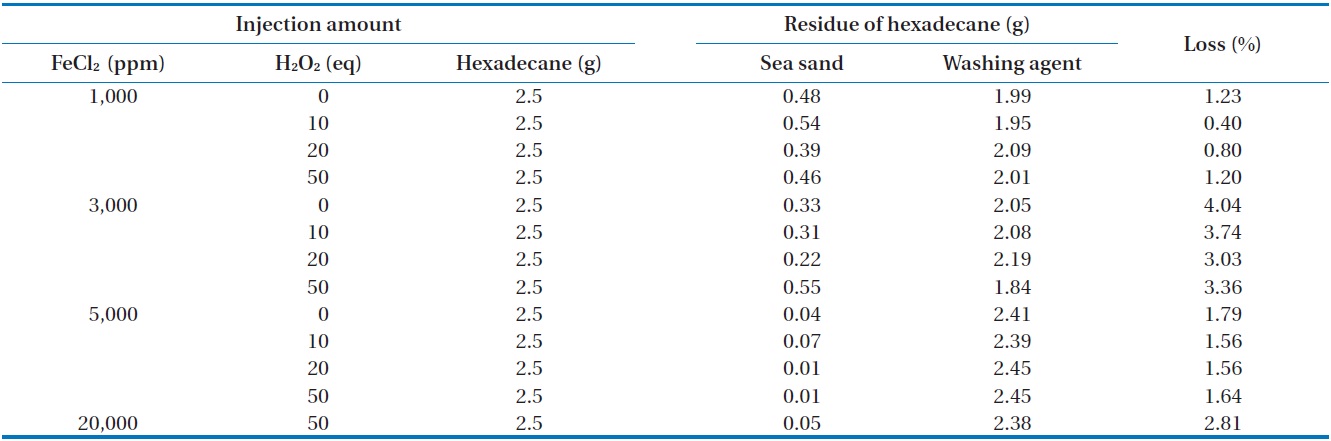
Removal of n-hexadecane in closed system
[Table 4.] Removal of n-hexadecane in open system
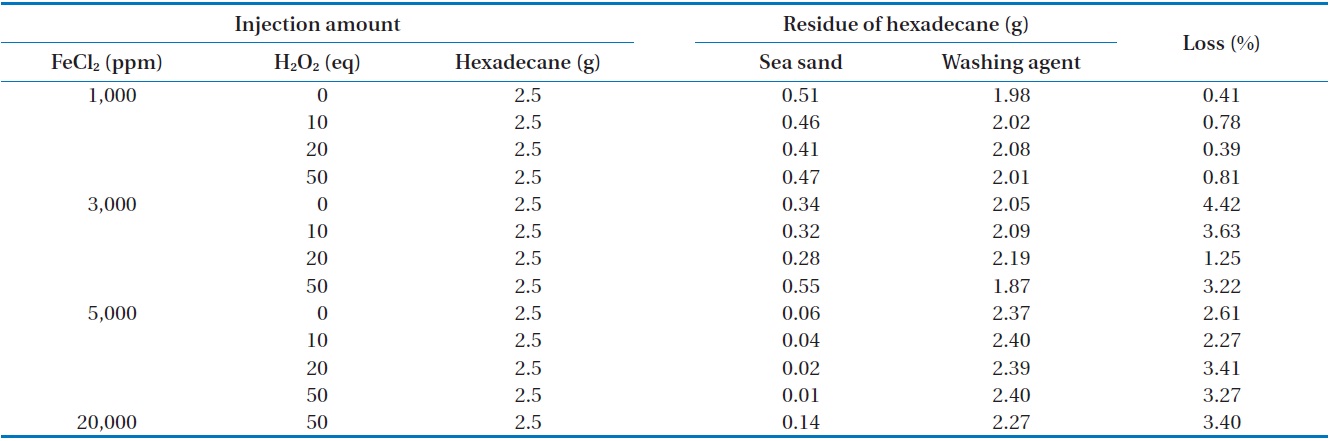
Removal of n-hexadecane in open system
[Table 5.] Removal of 1-methylnaphthalene in closed system
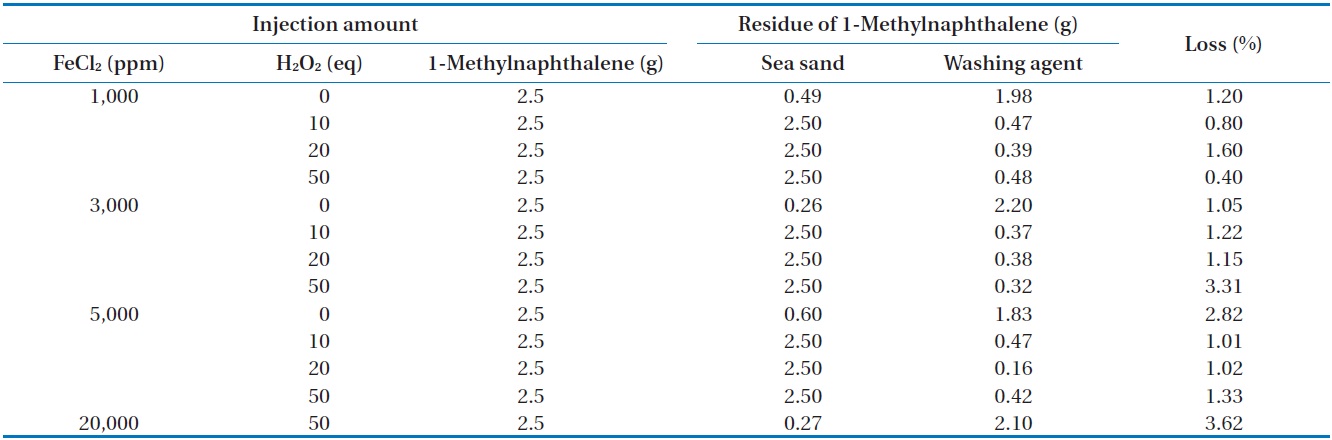
Removal of 1-methylnaphthalene in closed system
be identified through an acetylation method, because oxidation products cannot be detected by GC-MS.
The measured concentration of n-hexadecane in sea sand and washing agent at different amounts of FeCl2 and H2O2 injected in an open system is illustrated in Table 4. For all conditions, the mass recovery of n-hexadecane was above 95.5% of the initial concentration of n-hexadecane, which is quite similar to that observed in the closed system. These results indicate that no volatile reaction products were produced during the Fenton reaction. However, this result was quite different with removal of n-hexadecane by Fenton oxidation [16]. Chen et al. [16] reported complete mineralization of n-hexadecane in soil by Fenton oxidation when enough hydrogen peroxide was added.
3.2. Removal of 1-Methylnaphthalene in Closed and Open Systems
The Removal of 1-methylnaphthalene as a function of the
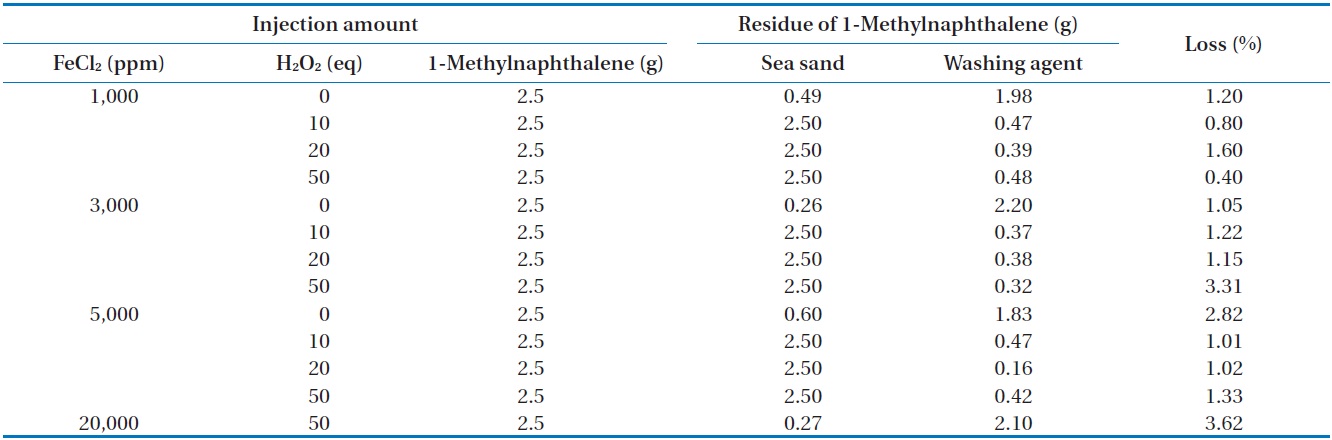
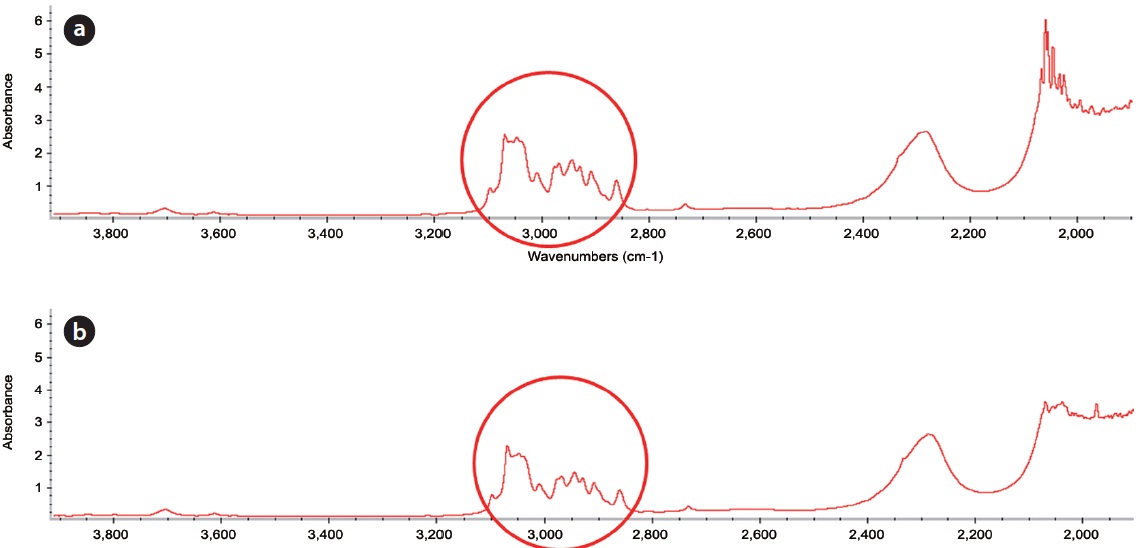
amounts of FeCl2 and H2O2 injected was also investigated in closed and open systems. The measured concentration of 1-methylnaphthalene in sea sand and washing agent at different amounts of FeCl2 and H2O2 injected in a closed system is illustrated in Table 5. For all conditions, the mass recovery of 1-methylnaphthalene was above 96.4% of the initial concentration of 1-methylnaphthalene, suggesting most of the 1-methylnaphthalene exists in its original form. In order to find any oxidation products of 1-methylnaphthalene during Fenton reaction, the reaction samples were analyzed by GC (Fig. 4) and FT-IR (Fig. 5). From this analysis, major peaks observed in GC analysis before and after Fenton reaction were quite similar in terms of retention time and peak area. In addition, several peaks between 2,800/cm and 3,200/cm in the FT-IR spectrum were also similar.
The measured concentration of 1-methylnaphthalene in sea sand and washing agent with different amounts of FeCl2 and H2O2
[Table 6.] Removal of 1-methylnaphthalene in open system
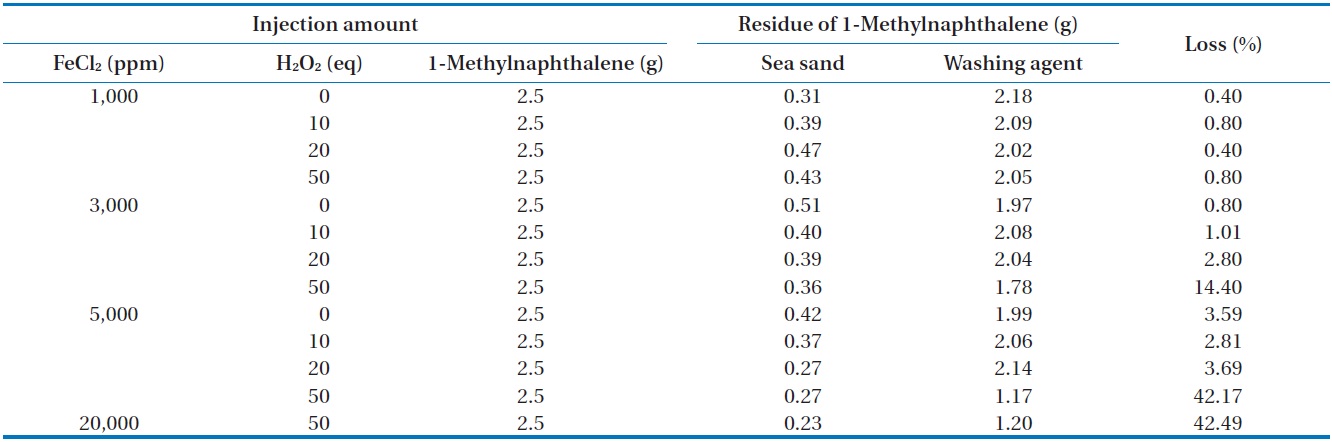
Removal of 1-methylnaphthalene in open system
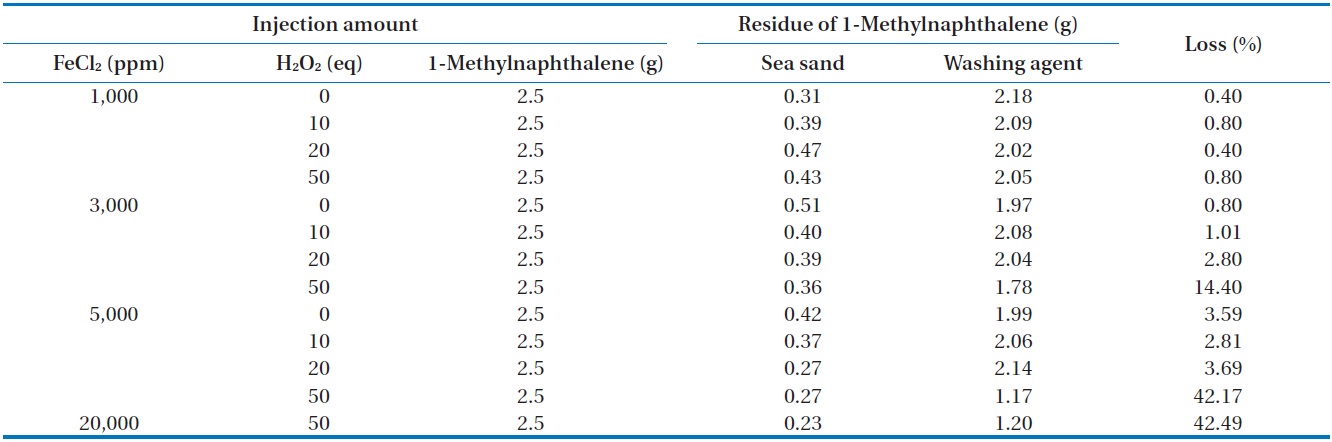
injected in an open system is illustrated in Table 6. For most conditions, except for the injection of 50 equivalents of H2O2 at 3,000, 5,000, and 20,000 ppm of FeCl2, the mass recovery of 1-methylnaphthalene was also above 95.3% of the initial concentration of 1-methylnaphthalene, which is quite similar to that observed
in the closed system. The reason for the relatively greater loss of 1-methylnaphthalene compared to n-hexadecane seems to be the unique physical properties of 1-methylnaphthalene, which has higher volatility than n-hexadecane [18]. When this work was compared with that reported by Chen et al. [16], quite different results were observed. They reported complete mineralization of 1-methylnaphthalene in soil by Fenton oxidation when enough hydrogen peroxide was added.
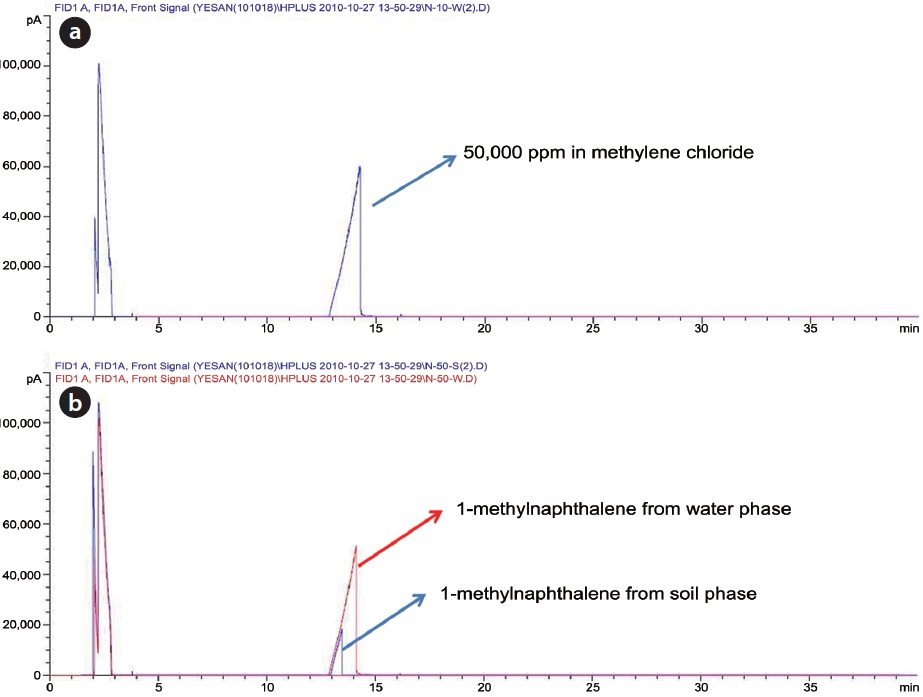
GS-MS with an INNOWax capillary column and polar characteristics was used to perform analysis of the oxidation products. As shown in Fig. 6, a small quantity of impurities was found in the standard substance of 1-methylnaphthalene. The GS-MS chromatograms for the samples before and after Fenton reaction were quite similar to that of the standard substance. As shown in Table 6, approximately 40% of the initial 1-methylnaphthalene content was reduced when the Fenton reaction performed with a high amount of FeCl2 and H2O2 injected in open conditions. But, there was a negligible reduction of 1-methylnaphthalene in the reaction using a reflux condenser. As mentioned previously, oxidation products can be identified through an acetylation method because oxidation products cannot be detected by GC-MS, suggesting the need for further study with acetylation technique [17].
3.3. Changes in the Temperature Profile According to the Quantity of H2O2 Injected
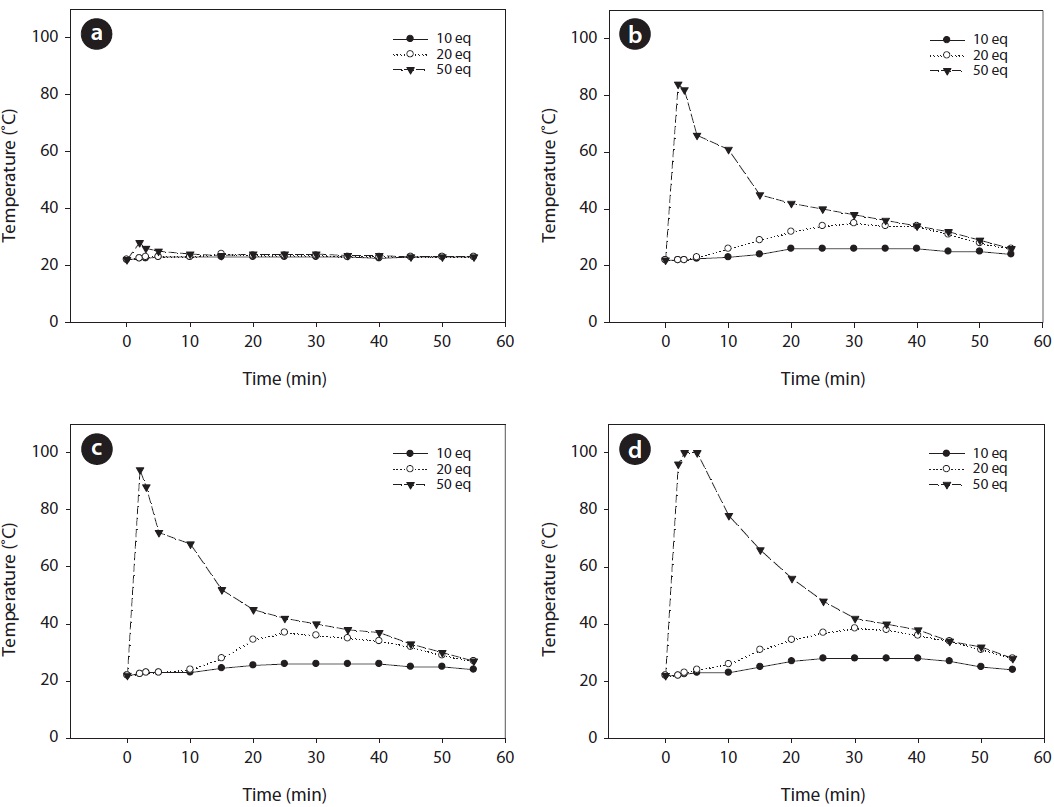
The variation of temperature during the Fenton reaction performed at different amounts of FeCl2 and H2O2 was monitored. The measured temperature in the reactor at different amounts of FeCl2 and H2O2 injected is illustrated in Fig. 7. When 1,000 ppm of FeCl2 was injected, no noticeable rise in temperature was observed for all concentrations of H2O2. This result indicates that very limited reaction occurs between iron salt and hydrogen peroxide these conditions. However, an important Fenton reaction occurred when the amount of hydrogen peroxide injected is 50 equivalent and the amount of iron salt injected is more than 3,000 ppm. As illustrated in Fig. 7, a rapid increase in temperature at the initial stage was observed as the concentrations of hydrogen peroxide and iron salt increased. In particular, the temperature increased rapidly to more than 80℃ when the hydrogen peroxide was 50 equivalents and the concentration of iron salt injected was more than 3,000 ppm, as mentioned previously. As shown in Table 6, the substantial loss that occurred in the reaction between 1-methylnaphthalene and higher concentration of Fenton’s chemicals in the beaker in the open system can be explained by the vaporization of a substantial quantity of 1-methylnaphthalene due to high temperature.
3.4. Changes in the Concentration of Hydrogen Peroxide According to the Quantity of H2O2 Injected
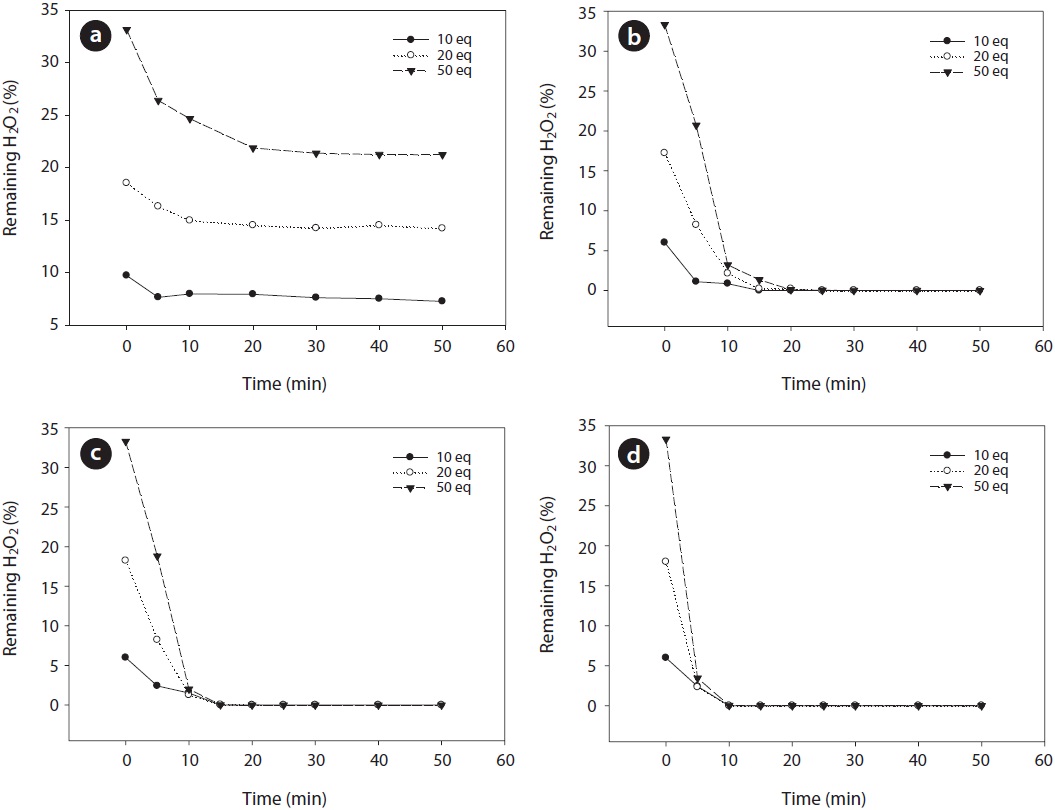
The variation of hydrogen peroxide during the Fenton reaction performed with different amounts of FeCl2 and H2O2 was investigated. The measured hydrogen peroxide in the reactor with different amounts of FeCl2 and H2O2 injected is illustrated in Fig. 8. When the concentration of FeCl2 was 1,000 ppm, the reaction between FeCl2 and H2O2 was not harmonious and a consider-
able fraction of hydrogen peroxide remained over the reaction period. When the concentration of FeCl2 was above 3,000 ppm, the residual concentration of the hydrogen peroxide dropped to less than 0.1% within 10 min. This phenomenon is well matched by a rapid increase in temperature at the initial stage, suggesting that decomposition of hydrogen peroxide by Fe2+ occurs actively at a higher concentration of FeCl2.
Considering the rise in temperature and the stability of H2O2 at different amounts of FeCl2 and H2O2 injection, it can be suggested that the Fenton reaction should be performed with controlled conditions that can provide a sufficient amount of oxidant in the removal of n-hexadecane and 1-mehtylnaphthalene.
When the oxidation of n-hexadecane by Fenton reaction was performed with a reflux condenser under closed system conditions, more than 96% of the initial n-hexadecane remained. More than 95.5% of n-hexadecane also remained when the Fenton reaction was performed with a beaker under open conditions. This illustrated that the oxidant was not sufficiently reacted with n-hexadecane during Fenton reaction under the reaction conditions in this study. In the Fenton reaction performed with high amounts of FeCl2 and H2O2 injection, approximately 40% of the initial 1-methylnaphthalene was reduced in a beaker under open conditions, but, there was negligible reduction of 1-methylnaphthalene in the reaction using a reflux condenser. This result suggests that 1-methylnaphthalene may be vaporized due to the heat generated by the Fenton reaction during the reaction time. These results were supported by GC chromatography and FT-IR spectroscopy. No important intermediates or reaction products of 1-methylnaphthalene were found. Considering the rise in temperature and stability of H2O2 with different amounts of FeCl2 and H2O2 injected, it can be suggested that the Fenton reaction should be performed with controlled conditions that can provide a suitable reaction environment between oxidant and contaminants.