Water hyacinth (
Composting is a bio-oxidative process in which microorganisms transform biodegradable organic matter into carbon dioxide (CO2), water vapour (H2O), ammonia, nitrate and methane, releasing heat during the process and, with time, forming a more stable organic matter called humus (humification process), which is structurally very similar to that present in soil. Compost stability indicates the extent or specific stage to which the readily biodegradable material has decomposed, and depends on the types of organic compounds remaining, and the prevailing biological activity in the composting biomass [7]. Bernai et al. [8] and Hue and Liu [9] related stability to the activity of the microbial biomass, and of the potential for unpleasant odour generation. Stability prevents nutrients from becoming tied up in rapid microbial growth, allowing them to be available for plant needs. On the other hand, maturity is the degree or level of completeness of composting [7], and implies improved qualities resulting from ‘ageing’ or ‘curing’ of the product. The UK Composting Association [10] defined mature compost as ‘compost that does not have a negative effect on seed germination or plant growth.’
The degree of stability attained within a certain time can be used for process performance monitoring and comparative evaluation of different composting systems [11, 12]. Application of unstable compost can also affect plant growth [13], as it shows phytotoxic behaviour due to the occurrence of toxic substances, resulting from insufficient biodegradation of organic compounds [14]. Das and Kalamdhad [15] have investigated the stability of water hyacinth compost obtained after 30 days agitated pile composting, through respiration tests i.e., CO2 production rate [16] and oxygen uptake rate (OUR) [17].
An efficient and promising technique of decentralized composting is the rotary drum composter that provides consistent agitation, aeration and mixing of the compost, to produce a uniform end product. Rotary drums of medium size can be placed at waste generation sites. Different types of waste viz. cattle manure, swine manure, municipal bio-solids, brewery sludge, chicken litter, animal mortalities, food residuals, etc. can be decomposed effectively in a rotary drum composter, within a remarkably reduced composting period of 2 to 3 wk [18-22]. No work has ever been done on the composting of water hyacinth in a rotary drum composter. Therefore, the present study performed a comparative investigation of the composting process of water hyacinth mixed with cattle manure, rice husk and sawdust in four different proportions in a pilot scale rotary drum, and evaluated the stability of the compost.
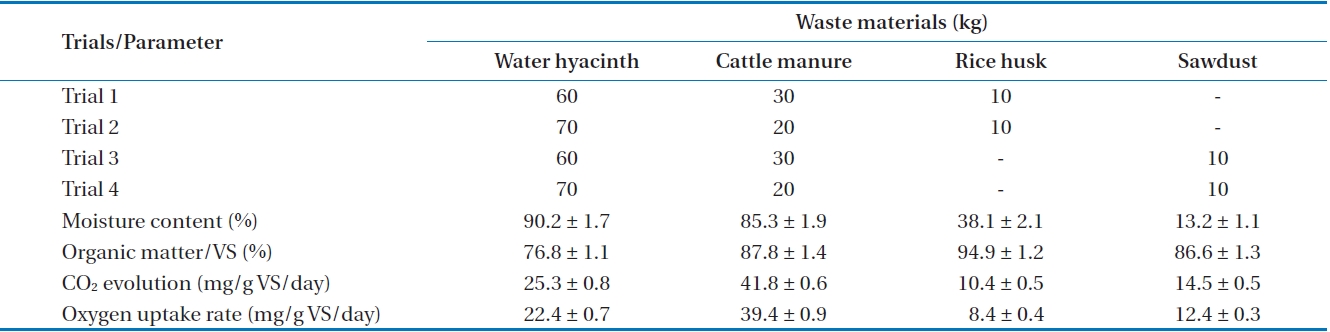
Water hyacinth, cattle manure, rice husk and sawdust were used for preparation of different waste mixtures. Water hyacinth was collected from the nearby ponds of the Indian Institute of Technology Guwahati campus. Fresh cattle manure was obtained from nearby Amingaon village. Rice husk and sawdust were purchased from the nearby rice mill and sawmill, respectively. The compost material was prepared by mixing different proportions of the collected waste, as described in Table 1.
2.2. Pilot Scale Rotary Drum Composter ? Batch Operation
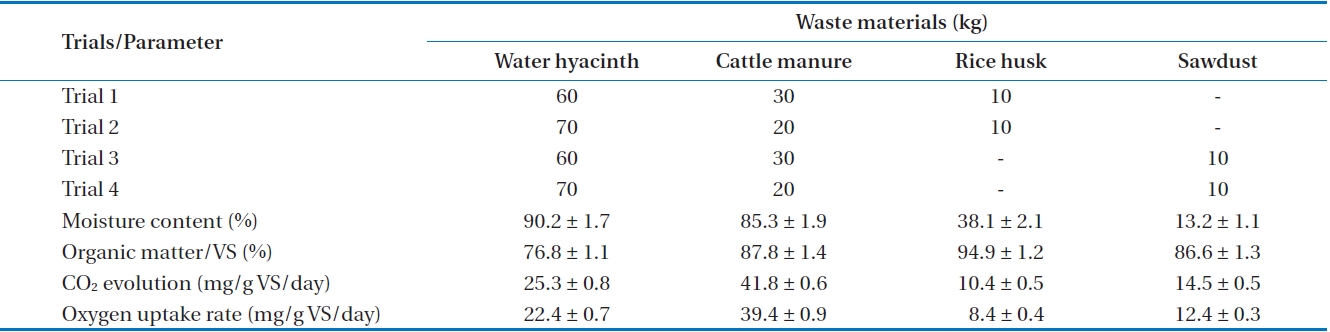
Fig. 1 shows a schematic diagram of the pilot-scale rotary drum composter of 250 L capacity. The main unit of the composter, i.e., the drum, is of 0.92 m in length and 0.9 m in diameter, made up of a 4-mm thick metal sheet. The inner side of the drum is covered with anti-corrosive coating. The drum is mounted on four rubber rollers attached to a metal stand, and is rotated manually by its handle. In order to ensure appropriate mixing, agitation and aeration of the wastes during rotation, 40 × 40 mm angles are welded longitudinally inside the drum. In addition, two adjacent holes are made on top of the drum to drain excess water. The capacity of the pilot scale rotary drum batch reactor was decided by keeping in view the capability of a single person, who can handle 60 to 100 kg of wastes by manual rotation. The composting period of 20 days was decided for both active degradation and stabilization periods, based on the performance of earlier studies of in-vessel composting reactors. Manual turning was done after every 24 hr, through two to three rotations of the rotary drum at a time, to ensure that the material on the top portion moved to the central portion, where it was subjected to higher temperature. Thereafter, aerobic condition was maintained by opening the top half side doors of the two circular faces.
2.3. Sampling and Parameter Analysis
About 100 g of samples were collected through grab sampling from six different locations, mainly from the mid span and end terminals of the pilot-scale rotary drum composter, using a compost sampler to minimize disturbance of the adjacent materials. All the grab samples were thoroughly mixed together to make a homogenized sample. Triplicate homogenized samples were
[Table 1.] Waste composition and characteristics of waste materials
Waste composition and characteristics of waste materials
collected on day 0 and after 2, 4, 6, 8, 10, 12, 14, 16, 18, and 20 days of the composting process, and stored at 4℃, for biological analysis of the wet samples within 2 days. The sub-samples were air dried immediately, ground to pass through 0.2 mm sieve, and stored for physico-chemical analysis.
Throughout the composting period the temperature was monitored every 6 hr, using a digital thermometer at three different locations within the pilot-scale rotary drum composter, i.e., at its centre and at its two ends, and the mean of the readings was reported. Moisture content (MC) was determined from weight loss of the compost sample (105℃ at 24 hr). Each air dried and ground sample was analyzed for the following parameters: pH and electrical conductivity (EC) (1:10 w/v waste: water extract with pH and EC meters); volatile solids (VS)/organic matter (loss on ignition at 550℃ for 2 hr), total nitrogen using the Kjeldahl method, ammonical nitrogen (NH4-N) and nitrogen (NO3-N) using KCl extraction [23], available and total phosphorus (acid digest) using the stannous chloride method [24], and potassium, sodium and calcium (acid digest) using flame photometry.
A liquid suspension of compost (8 g of compost in 500 mL of distilled water added with CaCl2, MgSO4, FeCl3, and phosphate buffer at pH 7.2) incubated at room temperature (24 ± 2℃) was placed in the sample bottle. A dissolved oxygen (DO) sensor was placed in the sample bottle at a depth of 5-7 cm below the water surface. The suspension was continuously stirred by means of a magnetic stirrer. The O2 concentration was measured continuously, and this value quoted as the OUR in mg O2/g VS/day.
The CO2 evolution rate was measured using a static measurement method [25]. Approximately, 10 g of sample was sealed in a 0.5 L vessel along with a beaker containing a known weight of oven-dried soda lime (105℃, 1.5-2.0 mesh) to trap CO2. The samples were incubated at room temperature (24 ± 2℃). Blank mea-
surement, necessary for initial CO2 in the vessel, was determined without sample in a similar vessel. The soda lime traps were removed after 24 hr, oven dried and reweighed to determine the CO2 absorbed.
All the results reported are the means of three replicates. Analysis of variance (ANOVA) was made using statistical software, to determine any significant differences among the parameters analyzed for different trials.
3.1. Physico-Chemical Analysis
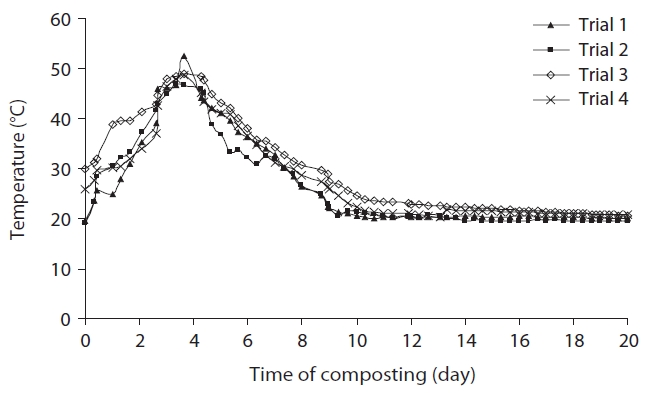
The temperature indicates the biological processes taking place in the composter, and plays a selective role on evolution and succession of microbiological communities. The variation in mean temperature of composting material in the composter with time is illustrated in Fig. 2. After 3 days, trial 1 with low initial temperature (19.6℃) recorded the highest maximum temperature (52.7℃) among the temperature profiles of all 4 trials, and entered the thermophilic phase, indicating quick establishment of microbial activities. The rapid rise in temperature at the beginning of composting is attributed to a higher content of easily biodegradable cattle manure, coupled with the presence of rice husk. trials 2, 3, and 4 with initial temperatures 18.9℃, 30℃, and 25.9℃ reached maximum temperatures 46.7℃, 49.5℃, and 48.9℃, respectively. Afterwards, a cooling period was observed until the end of the composting process. The comparatively low maximum temperatures in trials 2 and 4 containing a higher proportion of water hyacinth indicates that water hyacinth without appropriate amendments is less favourable for growth and biological activity of microorganisms. All the composting trials attained ambient temperature at 20 days, indicating a good degree of stability.
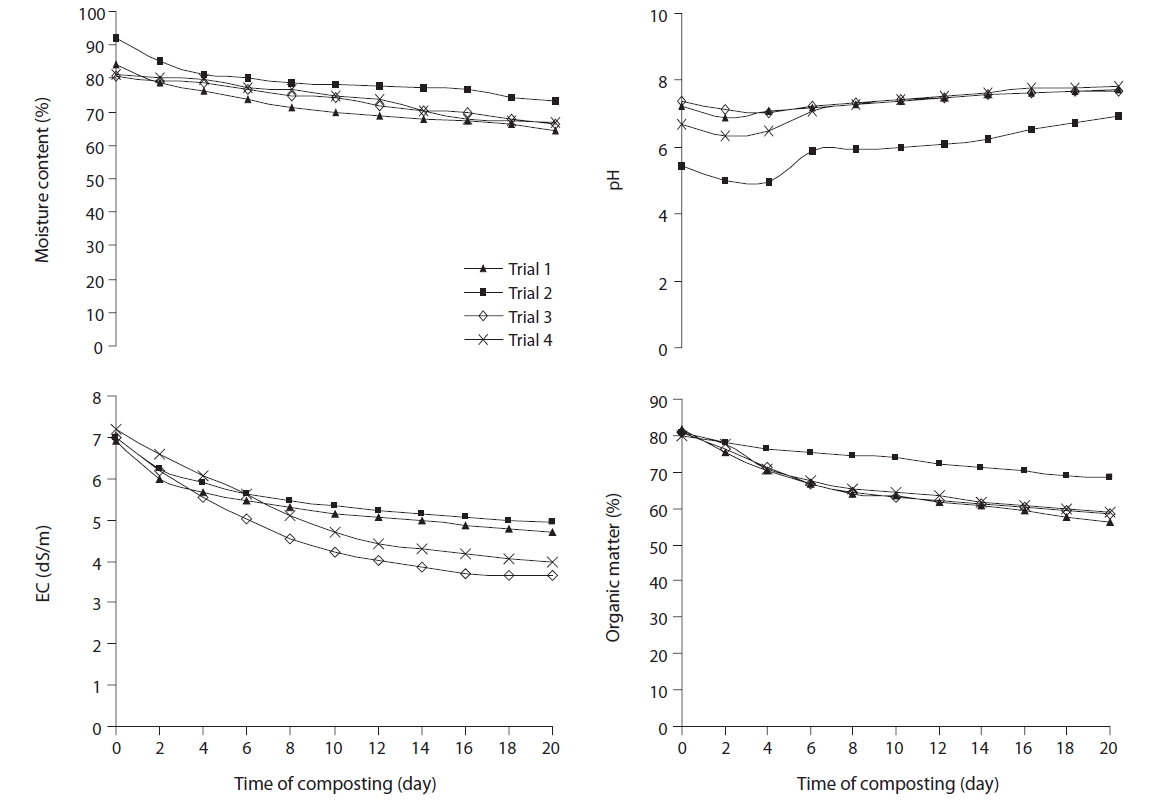
Moisture loss during the composting process can be viewed as an index of decomposition rate, since the heat generated during decomposition leads to vapourisation. An optimum MC is required for organisms to survive. Initial MC were 84.2%, 91.9%, 80.8%, and 81.3%, and reduced to 64.6%, 73.4%, 66.3%, and 66.8% within 20 days of the composting period in trials 1, 2, 3, and 4, respectively (Fig. 3). The highest moisture loss occurred in trial 1 (19.6%), and it correlated with the higher temperature profile of trial 1. The higher initial MC in trials 2 and 4 compared to trials 1 and 3 were due to the higher proportion of water hya-cinth, which has high moisture content.
During the nitrification process, the nitrifying bacteria lower the pH of the medium, due to the liberation of hydrogen ions. Once nitrification began after the thermophilic stage, the pH of the composting materials started to decrease, and pH values were directly related to nitrification [26]. As composting proceeds, the organic acids become neutralized, and compost material tends toward a neutral pH [27]. Similar observations were found in all trials as pH reduced during the initial 4 days, after which it gradually increased up to 8 (Fig. 3). Aeration tends to decrease CO2 level in the compost, which in turn will tend to increase pH [28]. Lower initial pH was observed in trials 2 and 4 containing a higher proportion of water hyacinth, and this indicated the acidic nature of water hyacinth.
The EC value reflected the degree of salinity in the composting material, indicating its possible phytotoxicity effects on the growth of plant. High initial EC values in composting biomass are due to release of ammonium ions. The volatilisation of ammonia and the precipitation of mineral salts are possible reasons for the decreased EC at the later phase of composting [29]. The EC value reflected the degree of salinity in the compost, indicating its possible phytotoxic/phyto-inhibitory effects on the growth of plant, if applied to soil [30]. For the improvement of agricultural soils, the acceptable level of EC required in compost should be lower than 4 dS/m [31]. After 20 days, only composts of trial 3 and 4 approached the desired EC values (Fig. 3). The high EC values indicated that all the composting trials needed further maturation, in order to be safe for plant use.
During the composting process, carbon dioxide is emitted from the composting mass, as one of the metabolic end products. Thus, the total carbon content of the composting mass decreases as composting proceeds. In matured composts, most of the organic matter (OM) is in the form of stable humic substances. Fig. 3 shows the trend of OM degradation during the drum composting process, of the four different combinations of water hyacinth, cattle manure and rice husk/sawdust. There was significant decrease shown by ANOVA analysis in the OM content, as the decomposition progressed in the four trials (
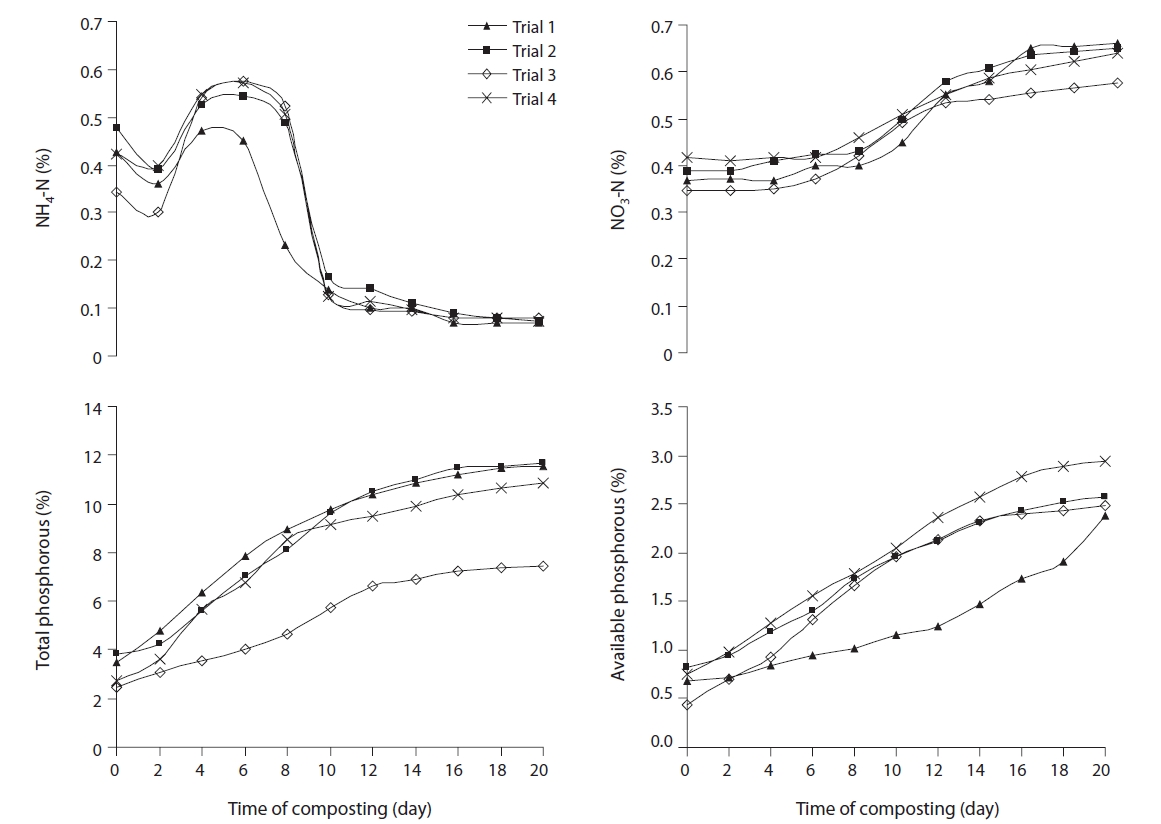
The changes in concentration of NH4-N and NO3-N in all trials followed the general trend during composting. During the first 2 days of composting, NH4-N concentration decreased in all trials. An increase was observed after 2 days for around 4-6 days, after which a sudden drop was noticed in all trials, which started stabilising by the end of 20 days trials at around 0.07%, 0.072%, 0.078%, and 0.074% in trials 1, 2, 3, and 4, respectively (Fig. 4). The initial increase in NH4-N concentration could be due to the conversion of organic nitrogen to NH4-N through volatilization and immobilization by microorganisms [30]. It has been noted that either the absence or decrease in NH4-N is an indicator of high-quality compost [29]. In this context trial 1 gave better compost, as compared to the rest of the trials. However, it far exceeded the 0.4% value recommended as the maximum content in matured compost [13].
The rapid decrease in NH4-N concentration during composting did not coincide with a similar increase in the NO3-N concentration. Nitrate is almost absent in cattle manure and sawdust, but a higher concentration is prevalent in water
hyacinth. So, all the composting trials showed initial nitrate concentrations. Otherwise, the high temperature and excessive amount of ammonia inhibited the activity and growth of nitrifying bacteria in the thermophilic phase [30]. Organic nitrogen mineralisation may be the limiting step in nitrification, since such mineralization is extremely low during the last phase of composting, when the supply of ammonium available to the nitrifying bacteria would have been reduced [26]. NO3-N increased significantly after 12th day composting in trials 1, 2, and 4, although no significant difference was observed among the trials (Fig. 4).
Phosphorous in organic material is released by a mineralization process involving micro-organisms, and gradually increased during the composting process. The water solubility of phosphorous decreased with the humification, thereby showing that phosphorous solubilised during the decomposition was subjected to further immobilization by the compost accelerator microorganisms. The higher initial available phosphorus in trials 2 and 4, compared to corresponding trials 1 and 3, was due to the higher proportion of water hyacinth in the composting material (Fig. 4). The gradual increase in the total and available phosphorus throughout the composting period was due to the net loss of dry mass resulting from loss of organic carbon, hydrogen, nitrogen and oxygen as CO2, H2S, and H2O during the composting process.
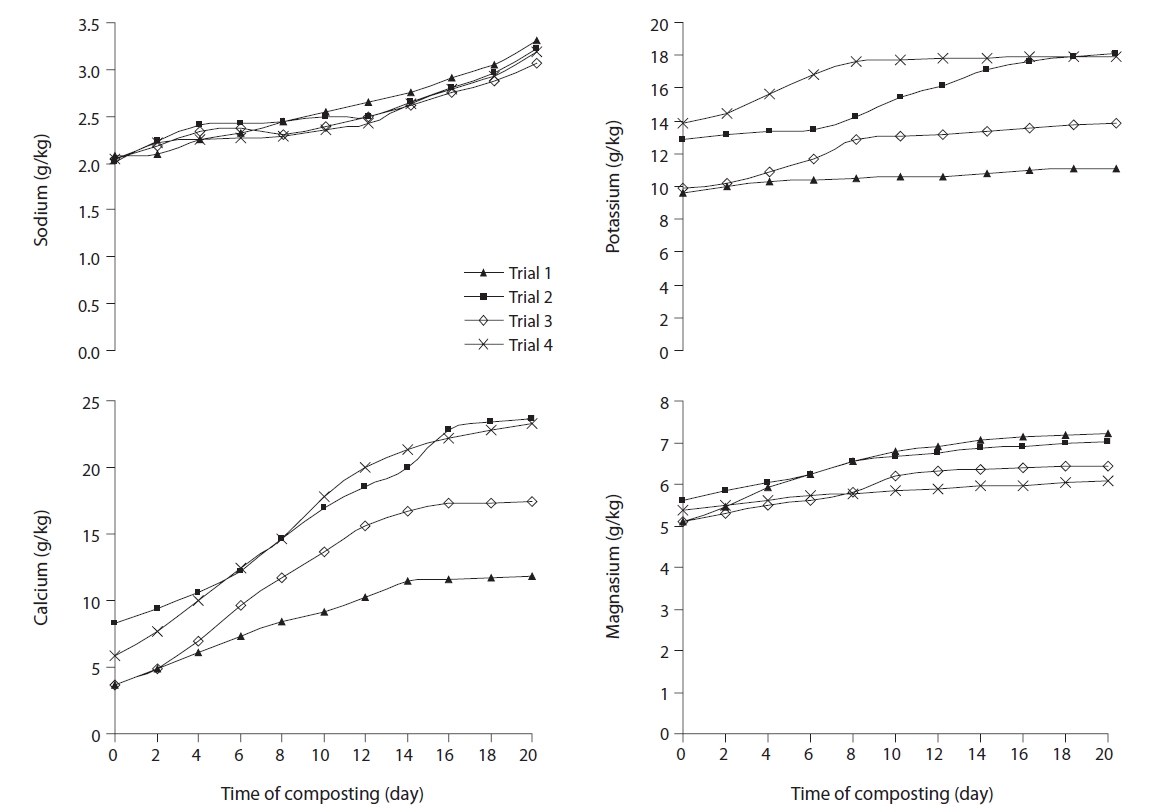
Fig. 5 illustrates the concentration profile of macronutrients, such as total K, Na, Ca, and Mg, in the four trials, during the rotary drum composting process. Organisms need nutrients for their metabolism. Nutrient content in compost is an indication of good quality fertilizers for plants. All macronutrients, i.e., K, Na, Ca, and Mg, showed steady increase in all the trials. A higher final concentration of nutrients was observed in trials 2 and 4, containing higher proportions of water hyacinth. Water hyacinths contain high concentrations of nutrients, particularly Ca and K.
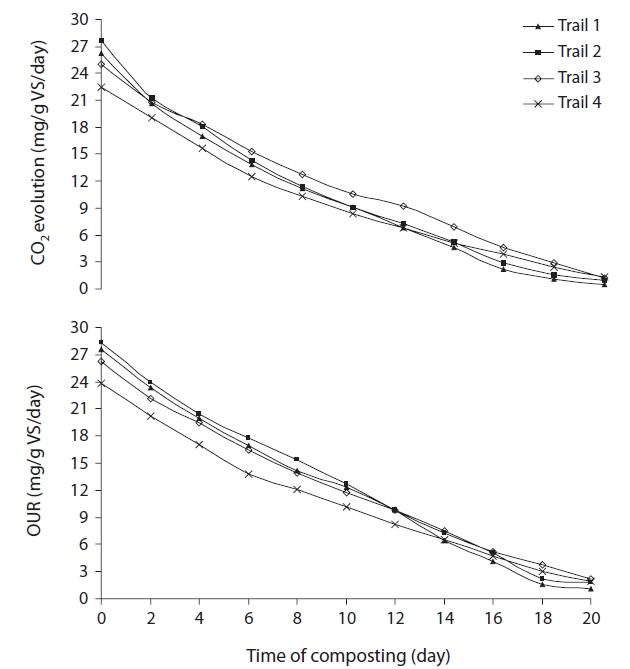
CO2 evolution is the most direct technique of compost stability, because it measures carbon derived directly from the compost being tested. Thus CO2 evolution directly correlates with aerobic respiration. The high CO2 concentrations indicate elevated microbial respiration of the readily available carbon in the composting mixture. The decrease in the rate of CO2 evolution with composting time is a result of a reduction in metabolic activity, due to the decrease of readily available carbon. The CO2 evolution rates decreased by 98.01%, 96.27%, 95.12%, and 93.80% after 20 days in trials 1, 2, 3, and 4, respectively (Fig. 6). The results, showing more decrease of CO2 evolution values in trial 1 than the rest of the trials, indicate more stabilization in trial 1.
OUR is the most accepted method for the determination of biological activity in composts. It evaluates the amount of readily biodegradable organic matter still present in the sample through its carbonaceous oxygen demand. Kalamdhad and Kazmi [31] found OURs to be high in the active stage of composting, as microbes grow rapidly from digesting readily biodegradable substrate. As composting begins, large organic molecules are broken down to smaller, soluble ones, and more substrate may temporarily become available.
Higher respiration rates were observed in the beginning of
the composting in all cases, especially in trial 1, due to availability of readily degradable cattle manure with the water hyacinth and rice husk. This was in agreement with Iannotti el al. [17], where OUR was found high in raw material when microbes grew rapidly. The sharp decreased observed in all trials after 1 wk correlated with the considerable drop in temperature and moisture content. The OUR of trials 1, 2, 3, and 4 decreased by 94.84%, 92.72%, 89.59%, and 89.82%, respectively (Fig. 6). The greater decrease of OUR value in trial 1 than trials 2, 3, and 4 again enhances our finding indicating more stability of trial 1. The OUR dropped steadily after the initial sharp decrease in all cases, while after 1 wk of composting the drop is moderate, indicating the compost is approaching stability.
The correlations of the CO2 evolution rate and OUR, both adopted as markers of compost stability, with other parameters such as MC, pH and OM, were investigated using a multi-linear stepwise regression analysis method. Analysis established strong correlations of the parameters with OUR and CO2 evolution. The simpler regression models generated was expressed by the following equations:
Trial 1: CO2 = -51.2633 - 7.501866 (pH) + 1.2820 (MC) + 0.4696 (OM) OUR = -42.4285 - 8.1609 (pH) + 0.9055 (MC) + 0.9098 (OM)
Trial 2: CO2 = -66.3939 - 1.4768 (pH) + 2.4104 (MC) - 1.4285 (OM) OUR = -165.971 + 0.2804 (pH) + 1.5932 (MC) + 0.8126 (OM)
Trial 3: CO2 = -76.8698 - 4.58082 (pH) - 0.1685 (MC) + 2.5524 (OM) OUR = -65.9169 - 5.6078 (pH) - 0.0255 (MC) + 2.6399 (OM)
Trial 4: CO2 = -129.8194 - 1.0811 (pH) + 0.9323 (MC) + 1.5602 (OM) OUR = -142.1565 + 0.0814 (pH) + 0.6444 (MC) + 2.0817 (OM)
Using this equation, CO2 evolution rate and OUR were pre-dicted for each compost type. A highly significant coefficient of multiple determinations (R2) during trials 1, 2, 3, and 4 for these models indicated that CO2 evolution rate and OUR could be estimated by these parameters. The observed CO2 evolution rate and OUR, and the predicted CO2 evolution rate and OURs, were evaluated using the above equation in all trials, to provide the minimum data required for correlation. Strong correlations between the observed CO2 evolution rate and the predicted CO2 evolution rate, and between observed OUR and predicted OUR, were obtained.
Higher temperature profiles were recorded during rotary drum composting of trials 1 and 3, which established favourable conditions for microbial activity of the composting materials. Higher initial MCs in trials 2 and 4 were due to higher proportions of water hyacinth, compared to the other two trials. Higher moisture loss in trial 1 correlated with its temperature profile. Lower initial pH observed in trials 2 and 4 was due to the high amount of water hyacinth. Higher loss of OM in trial 1 correlated with the higher temperature profile of trial 1. Higher initial OUR and CO2, and lower final OUR and CO2, observed in trial 1 compared to other trials, indicated the better composting mix for water hyacinth rotary drum composting. There were significant correlations of MC, pH, and OM loss with both CO2 evolution rate and OUR for each trial, and they can be predicted from the parameters.

![Pilot scale rotary drum composter [22].](http://oak.go.kr/repository/journal/11611/E1HGBK_2012_v17n2_69_f001.jpg)