Transdermal drug delivery (TDD) introduces therapeutic agents into the body through the skin. Drug transport can be either passive, relying on drug diffusion across the skin, or active, driven by application of an electric field (iontophoresis) [1]. Transdermal drug delivery systems have been studied for use in various areas such as hormonal therapy, pain relief, and smoking cessation aids; such delivery systems allow the controlled release of drugs over several hours or even days [2-4].
Hydrogel is a three-dimensional, crosslinked polymeric material that can retain a large amount of water. It can swell greatly by absorbing water and shrink after de-swelling. The swelling capability depends on the nature of polymeric chains and their crosslinking density. For more than two decades, considerable attention has been focused on environmentally sensitive hydrogels or so-called smart hydrogels. These hydrogels can undergo a reversible and yet discontinuous volume change in response to environmental stimuli such as pH [5], temperature [6-9], ionic strength [10], electric field [11], and so on. These smart hydrogels have potential applications in the biomedical and pharmaceutical fields.
One of the most commonly investigated temperature-sensitive polymers is poly(Nisopropylacrylamide) (PNIPAAm), which exhibits a phase transition temperature and a lower critical solution temperature (LCST, 32℃) in water [12]. PNIPAAm hydrogels thus can swell in water below the LCST and shrink above it [13,14]. PVA is a watersoluble hydrophilic polymer that has been extensively explored as a biomaterial, being used in drug delivery systems, sensors, and surgical repairs because of its excellent mechanical properties, biocompatibility, and nontoxicity [15]. Poly(acrylic acid) (PAAc) is considered as a pH and electrically sensitive material due to the ionic repulsion between its carboxylate anionic groups. As the pH decreases below the pKa (4.7) of PAAc or increases above the pKa of PAAc, the PAAc will shrink or swell, respectively, due to the variation in the content of carboxylate anion [16].
Carbon nanotubes (CNTs) possess unique and remarkable mechanical and geometrical properties, such as light weight, high aspect ratio, and high stiffness. In comparison with metal or metal oxide materials, CNTs have higher thermal conductivity. The thermal conductivities of single-walled CNTs (SWCNTs), double-walled CNTs (DWCNT), and multiwalled CNTs (MWCNTs) are 6000 W/mK, 3986 W/mK, and 3000 W/mK, respectively [17]. These thermal characteristics of CNTs are beneficial to the improvement in the thermal response of nanocomposites.
The toxicity of CNTs is also a concern for their application in drug release. Recently, Yun et al. [18] reported that effective TDD was developed by PVA/PAAc/MWCNTs nanofibers; this study proved the nontoxic nature of the ingredients using cytotoxicity tests. Therefore, in the present study, as a nontoxic component to improve the thermal conductivity, MWCNTs were incorporated in nanocomposites for drug release.
In this study, drug-loaded PVA/PAAc/PNIPAAm/MWCNT nanocomposite hydrogels were prepared for use as a carrier matrix of a model drug, coomassie brilliant blue. From the viewpoint of recent biomedical applications, it would be favorable if the hydrogel could respond to both temperature and pH stimuli either mutually or independently. Controlled drug release having both temperature and pH responses, together with the improved mechanical strength, would be preferable for the new drug delivery system. The dual-responsive release of a model drug was investigated for MWCNT-reinforced nanocomposite hydrogels consisting of both a temperature- sensitive PNIPAAm component and a pH-sensitive PAAc component.
The PVA, AAc, glutaraldehyde (GA), ethylene glycol dimethacrylate (EGDMA), and potassium persulfate (KPS) used in this study were obtained from Sigma Chemical Company. MWCNTs grown by chemical vapor deposition were supplied by Aldrich. MWCNTs have average diameters of 30-50 nm and lengths of 1-2 μm. NIPAAm was purchased from TCI Co. N,N'- methylenebisacrylamide (MBAAm) was purchased from Fluka Co. Coomassie brilliant blue R-250 (CBB), the model drug, was obtained from Bio-rad Laboratories.
2.2. Synthesis of PVA/PAAc/PNIPAAm/MWCNT nanocomposite hydrogels
PVA/PAAc/PNIPAAm/MWCNT nanocomposite hydrogels were synthesized by free radical polymerization. The polymerization was carried out in de-ionized water using GA, EGDMA, and MBAAm as the crosslinkers of PVA, AAc, and NIPAAm, respectively. KPS was used as an initiator. PVA solution (10 wt% w/v) was first prepared by dissolving PVA in deionized water for 6 h at 90℃ and subsequent cooling to room temperature. Various MWCNT dispersions (0.5, 1, and 3 wt%) were prepared in 3 mL ethanol by sonicating for 60 min. MWCNT dispersions were added to the PVA/AAc/NIPAAm solution with constant stirring. The reaction mixture was stirred for 30 min at room temperature followed by bubbling of nitrogen through the mixture for 20 min to remove the oxygen dissolved in the reaction mixture. The mixture was reacted at 70℃ by stirring for 2 h to produce PVA/ PAAc/PNIPAAm/MWCNT nanocomposite hydrogels.
Field emission scanning electron microscopy (FE-SEM) was used to investigate the surface morphology. FE-SEM was carried out at 10 kV using a Hitachi S-5500 microscope. Compression moduli were measured with a universal testing machine (UTM, Instron 5583) using a sample size of 20 mm × 20 mm × 10 mm with a rate of 1 mm/min.
2.4. Equilibrium swelling ratio of nanocomposite hydrogels
The equilibrium swelling ratio (ESR) of the nanocomposite hydrogels was determined by the gravimetric method under various temperatures and pHs. The dried samples were immersed in the buffer solution for a certain period until the swelling equilibrium was reached; then, these hydrogels were taken out, followed by removal of the excess buffer solution on the surface with tissue paper. Samples were weighed immediately. The ESR (%) of the hydrogels was calculated as follows:
ESR (%) = (Ws - Wd) ? Wd × 100
where Wd and Ws are the weights of dry and swollen samples, respectively.
PVA/PAAc/PNIPAAm/MWCNT nanocomposite hydrogels were dried in a vacuum overnight until their weights became stable. The dried hydrogels were immersed in 0.5 wt% CBB aqueous solution at 25℃ for at least 2 days to reach an equilibrated state. During this swelling period, the drug was loaded into the hydrogel networks. Drug loaded PVA/PAAc/PNIPAAm/MWCNT nanocomposite hydrogels were separated from the CBB solution, followed by drying in vacuum for future releasing experiments. The drug release experiments were conducted with vertical Franz-type diffusion cells (Disa, Milan, Italy). A nylon net was placed on the top of the Franz diffusion cell containing the buffer solution. The nanocomposite hydrogels were placed on the nylon net. The surface area of diffusion was 3.14 cm2. The drug release tests were conducted under various conditions such as 25℃, 34℃, pH 2, and pH 10. A buffer solution was used for the receptor phase and the temperature was maintained using a circulating water bath. Mixing of the receptor medium was provided by magnetic stirring. Two mL buffer solution was withdrawn from the receiver compartment and an equivalent volume of buffer solution was replaced immediately. The samples were assayed using a UV spectrometer at 290 nm to determine the amount of released drug [19].
3.1. Surface morphologies of nanocomposite hydrogels
FE-SEM images of PVA/PAAc/PNIPAAm/MWCNT nanocomposite hydrogels are shown in Fig. 1. MWCNTs were found in the hydrogel matrices without any noticeable crevices between the hydrogel and the MWCNTs. An increased numbers of uniformly dispersed MWCNTs were observed in the nanocomposites as the content of MWCNTs loaded increased up to 1 wt%. However, a large agglomeration of MWCNTs was observed for the nanocomposite hydrogels containing 3 wt% MWCNTs, as shown in Fig. 1d. The hydrophobicity of the MWCNTs was responsible for the poor dispersion in the hydrophilic polymer matrix at the higher content of MWCNTs.
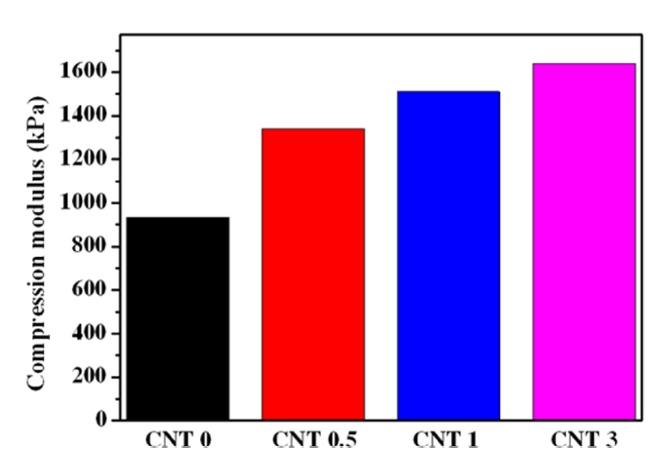
3.2. Compression moduli of nanocomposite hydrogels
Fig. 2 shows the compression moduli of the PVA/PAAc/ PNIPAAm/MWCNTs nanocomposite hydrogels. The swelling of the hydrogels is closely related to their structures and the swelling also influences the mechanical properties of the hydrogels. Therefore, it is essentially important to control both the swelling and the mechanical properties of the hydrogels. The compressive mechanical strength of the PVA/PAAc/PNIPAAm hydrogel increased greatly with the incorporation of MWCNTs into the hydrogel matrix. MWCNTs worked effectively to improve the compression properties of the PVA/PAAc/PNIPAAm/ MWCNTs nanocomposite hydrogels, even though the partial aggregation of hydrophobic MWCNTs occurred in the hydrophilic hydrogel matrix.
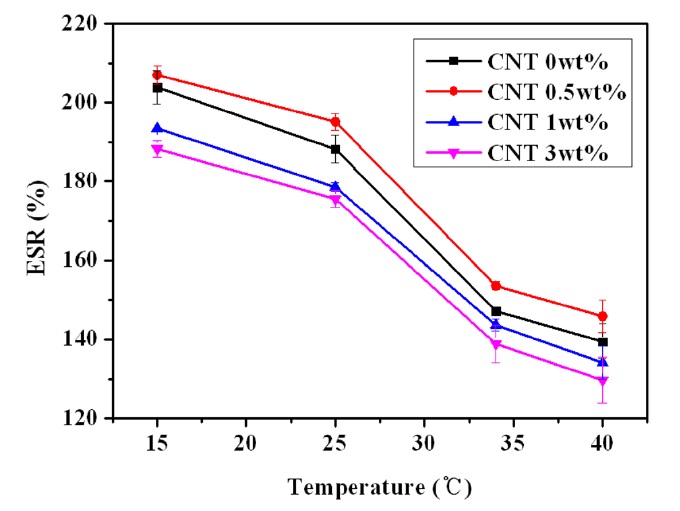
3.3. Temperature-sensitive swelling behavior of nanocomposite hydrogels
The temperature-sensitive swelling of the PVA/PAAc/ PNIPAAm/MWCNT nanocomposite hydrogels is shown in Fig. 3. The temperature sensitivity of the nanocomposite hydrogels was attributed to the unique LCST behavior of the PNIPAAm component. The swelling ratio of the PVA/PAAc/PNIPAAm/ MWCNT nanocomposite hydrogels decreased quickly as the temperature increased over the LCST of PNIPAAm and showed similar behavior regardless of the content of MWCNTs. As the temperature increased, decreased hydrogen bonding formation induced the deswelling of the hydrogel.
The swelling of the PVA/PAAc/PNIPAAm matrix is expected to decrease as the content of reinforcing ingredient increased due to obstacles to expansion. However, the highest swelling of the PVA/PAAc/PNIPAAm/MWCNT nanocomposite was observed over the whole temperature region at a content of 0.5 wt% MWCNTs, followed by a noticeable decrease in swelling as the content of MWCNTs increased further. The improved thermal
conductivity of the nanocomposite is believed to play an important role in increasing the temperature-sensitive swelling of the polymer matrix until the content of MWCNTs exerted a major effect of obstruction on swelling.
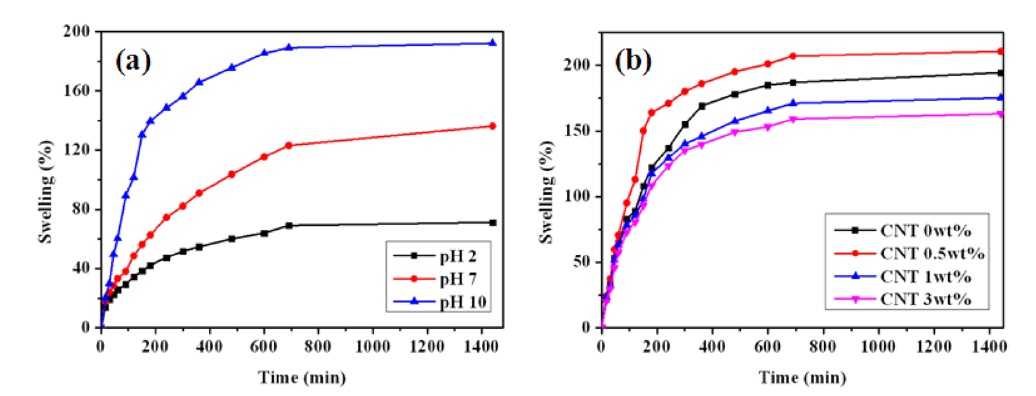
3.4. pH-sensitive swelling behavior of nanocomposite hydrogels
Fig. 4a shows the swelling behavior of nanocomposite hydrogels depending on pH. The swelling ratio increased dramatically as the pH increased to the basic condition. This pH-sensitive swelling was attributed to the ionization of the pendant carboxylic acid groups of the PAAc component in the nanocomposite hydrogels. The carboxylic acid groups are ionized into carboxylate anion above the pKa of 4.7. The amount of carboxylate anion increased as the pH increased. The carboxylate anions caused more electrostatic repulsion and hydrophilicity in the polymer chains in the hydrogels. Therefore, the swelling behavior of the hydrogels is very dependent on the ionization of the carboxylic acid groups [20]. The nanocomposite hydrogel containing 0.5 wt% MWCNT showed the highest swelling ratio, possibly due to the micro channel effect, which is caused by the uniformly dispersed MWCNTs. However, the swelling ratios of the nanocomposite hydrogels were generally lower than that of the PVA/ PAAc/PNIPAAm hydrogel as the content of MWCNTs increased to more than 0.5 wt% due to the partial aggregation of MWCNTs, as shown in Fig. 4b.
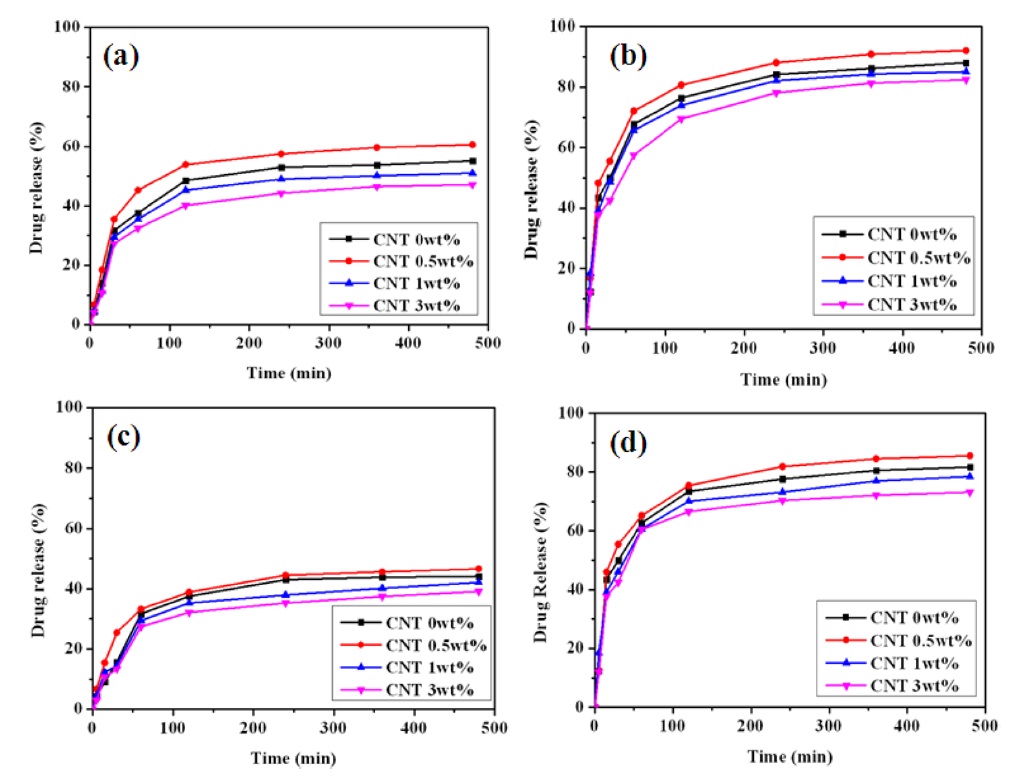
3.5. Drug release behavior of nanocomposite hydrogels
The dual sensitive release behavior of nanocomposite hydrogels was evaluated in detail based on both pH and temperature variations, as shown in Fig. 5. The drug release behavior of the various nanocomposite hydrogels was investigated at both 25℃ (below LCST) and 34℃ (above LCST), depending on the release time, as shown in Figs. 5a and b. Both the amount of released drug and the release rate increased as the temperature increased from 25℃ to 34℃ due to the higher shrinkage of the hydrogels. For the pH-sensitive characteristics of the nanocomposite hydrogels, shown in Figs. 5c and d, the amounts of drug release increased drastically as the pH increased from 2 to 10 due to the faster swelling of the hydrogels. The content of MWCNTs in the hydrogels also played some role in controlling the release behavior of the nanocomposite hydrogels, similar to the case of the swelling behavior. Therefore, the drug release behavior of the PVA/PAAc/PNIPAAm/MWCNT nanocomposite hydrogels can be controlled by varying the pH, temperature, and content of MWCNTs.
PVA/PAAc/PNIPAAm/MWCNT nanocomposite hydrogels were prepared by radical polymerization for use in dual sensitive drug delivery systems. MWCNTs were uniformly dispersed in the hydrogel matrix at a limited content. The increased contents of MWCNTs improved the mechanical properties of the PVA/ PAAc/PNIPAAm/MWCNT nanocomposite hydrogels, making them applicable as thin film devices in drug delivery systems. Both swelling and releasing behavior of the PVA/PAAc/ PNIPAAm/MWCNT nanocomposite hydrogels were dependent on the temperature, pH, and content of MWCNTs. PVA/PAAc/ PNIPAAm/MWCNT nanocomposite hydrogels can be applied as proper materials for stimuli-sensitive transdermal drug delivery systems due to the improved mechanical properties and dual sensitivities.