Polyacetylene (PA) is the most representative conducting polymer because of its simple linear conjugated molecular structure and intrinsic high electrical conductivity [1]. The polymerization procedure reported by Shirakawa and coworkers yields a randomly oriented fibril morphology [2]. However, the morphology of PA can be neither changed nor modified after the polymerization, because PA is infusible and insoluble in organic solvents. In order to solve this problem, liquid crystals (LC) have been adapted as an anisotropic reaction solvent for controlling the morphology of PA during acetylene polymerization. In fact, highly oriented PA films were obtained under a gravity flow [3] or magnetic field [4] of catalyst solution in which a nematic LC (N-LC) was used instead of common solvents such as toluene and cumene. Furthermore, higher-ordered helical PA (H-PA) was synthesized in a chiral nematic LC (N*-LC) reaction field [5].
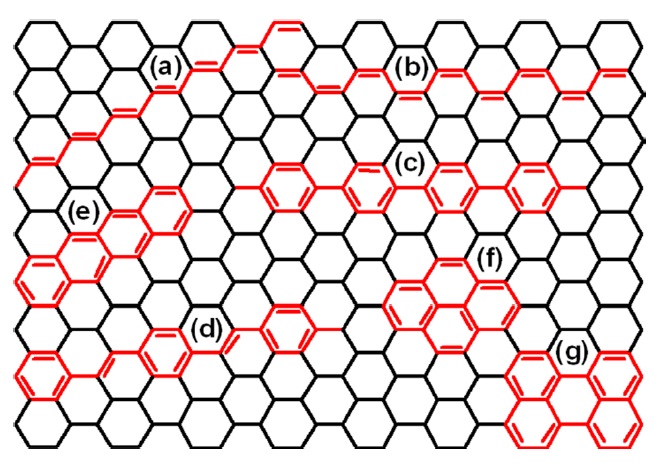
Carbon films are interesting materials because of their special physicochemical and mechanical properties. They have been used in various applications, such as electrochemical energy storage, cell electrodes, and gas absorbent materials. Freestanding carbon thin film, which has a peculiar nanofibril structure, is expected to afford novel electrical properties. However, it has been considered difficult to prepare a freestanding carbon thin film through the carbonization of an organic polymer film. This is because carbonization at high temperatures causes thermal decomposition and volatilization of hydrocarbon gases, destroying the morphology of the original film. Above all, the low carbon atom contents of organic polymers would lower the carbonization yield. On the other hand, because of their high carbon atom content and sp2-hybridized carbon, conjugated polymers have received a lot of attention as carbonization precursors. As shown in Scheme 1, many kinds of conjugated polymers or sp2-hybridized carbon materials can draw on a graphene sheet. Recently, car-
bon and graphitic films have been successfully prepared from iodine-doped PA film using morphology-retaining carbonization with high carbonization yield [6,7].
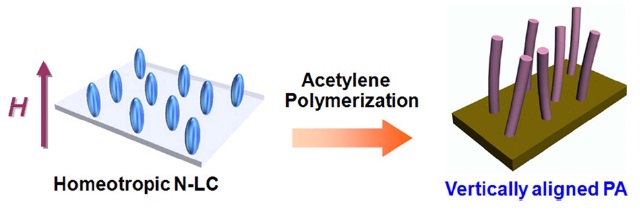
For practical use, vertically oriented carbon materials are required. This condition is well understood in the case of carbon nanotubes; for applications in areas such as field emission and nanoelectronics, it is necessary to grow vertically oriented carbon nanotubes [8]. If vertically aligned PA film was synthesized, we can make vertically aligned carbon materials by using a morphologyretaining carbonization method. Recently we synthesized vertically aligned PA in homeotropic LC solvent. Homeotropic N-LC was prepared by adding a so-called orientation dopant into the N-LC; in this process the orientation dopant is composed of two mesogenic cores linked with a hexamethylene spacer [9]. Although the orientation dopant technique gives us a very convenient and easy way to achieve the homeotropic alignment of LC, it prevents us from obtaining highly homogeneous vertically aligned fibrils. This is because the effect of the orientation dopant in fluid N-LC is sensitively affected by changes of temperature and pressure during acetylene polymerization. Therefore, we employed an alternative method for construction of the homeotropic LC phase. Namely, by using a magnetic field of 5 Tesla as an external perturbation, a PA film with vertically aligned fibril morphology was synthesized in homeotropic N-LC solvent. Furthermore, the synthesis of the vertically aligned carbon film was examined with iodine-doped PA film as a carbonization precursor by using morphology-retaining carbonization. This could be a step toward obtaining hierarchically morphology controlled carbon and graphite materials.
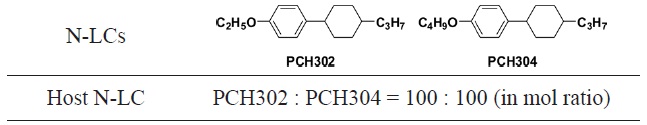
N-LCs of phenylcyclohexane derivatives, 4-(
[Table 1.] Molecular structures of N-LCs and mixing ratios of host N-LC
Molecular structures of N-LCs and mixing ratios of host N-LC
phase, the LC temperature region is very narrow, i.e., less than 1 to 2℃ . This is not suitable for acetylene polymerization, irrespective of N-LC reaction field, because the exothermal heat evoked during the acetylene polymerization would raise the temperature inside the Schlenk flask and easily change the LC phase into an isotropic one. Hence, we prepared an LC mixture by mixing the two equimolar N-LCs. In the N-LC mixture, the nematic to isotropic temperature, TN-I, and the crystalline to nematic temperature, TC-N, might be raised and lowered, respectively. In fact, the mixture exhibited an LC phase in the region from 20 to 35℃ .
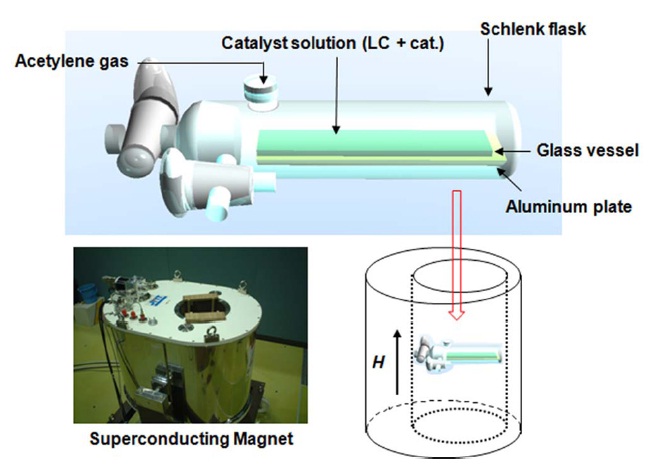
N-LC was used as a solvent for the Ziegler-Natta catalyst, which consists of Ti(O-n-Bu)4 and AlEt3. The concentration of [Ti] was 30 mM; the mole ratio of the co-catalyst to the catalyst [Al]/[Ti] was 4.0. The catalyst solution was aged for 30 min at room temperature; it was then completely degassed through a vacuum line and was transferred to a flat-bottomed container inside a Schlenk flask. The Schlenk flask was placed in a super conducting-magnet so that the solution in the container could be kept horizontal. A 5 Tesla magnetic field (JASTEC) was employed, and temperature was kept at 18 to 20℃ during the application of the magnetic field and the polymerization, in order to maintain the LC phase. A vertical magnetic field was applied to the N-LC for 30 min. Then, acetylene gas was introduced into the Schlenk flask. The initial acetylene pressure was about 40 torr and the polymerization time was about 5 min. The apparatus for acetylene polymerization under vertically applied magnetic field is shown in Fig. 1. After polymerization, the PA film was carefully stripped from the container, washed with purified toluene several times and, subsequently, washed with a methanol solution of hydrochloric acid (1N) and tetrahydrofuran. The films were dried through vacuum pumping on a Teflon sheet and stored in a freezer at -20℃ . Morphologies of the PA films were observed using an scanning electron microscope (SEM) of type DS-130 (TOPCON). Before measurements of SEM, the PA films were coated with Pt- Pd alloy using an ion coater of type IB-5 (EIKO). The coating thickness was evaluated and found to be ca. 10 nm. The SEM operation was performed under an acceleration voltage of 10 kV. Note that the helical pitch and the spiral morphology of the H-PA were maintained during the Pt-Pd alloy coating and the SEM observation, because of its infusibility.
Fig. 2 shows SEM photographs of the PA film synthesized in N-LC. Vertically aligned fibril morphology was observed. The fibrils (whose diameters are 70?200 nm) are grown inparallel to the direction of the magnetic field. The fibril length was in the range of 5?35 μm, depending on the polymerization time.
LC molecules with positive or negative anisotropy in diamagnetic susceptibility were aligned parallel or perpendicular to the direction of the magnetic field, respectively [10]. In this work, owing to the positive anisotropy in diamagnetic susceptibility of the phenyl moiety of the LCs (PCH302 and PCH304), the LC molecules tend to be aligned parallel to the direction of magnetic field. Moreover, it has been reported that the PCH derivatives can be aligned by a magnetic field of 10 000 gauss (1 Tesla) [4]. Therefore, the magnetic field of 5 Tesla is strong enough to change the multi-domain to the mono-domain structure N-LC one, namely aligned N-LC. It is thought that the disorder of the fibrils comes from the fluctuation of the LC solvent by the generation of exothermic heat during acetylene polymerization.
Fig. 3 provides a schematic representation of the synthesis of a vertically aligned PA film using a magnetic field. The N-LC containing the catalyst vertically aligns from a multi-domain to a mono-domain structure under a vertical magnetic field, giving rise to an anisotropic reaction field. The PA grows along with the longitudinal direction of the LC molecules. Therefore, interfacial acetylene polymerization in the anisotropic reaction field prepared with the aligned N-LC reaction field gives a vertically aligned PA film. The aligned PA film has aligned bundles of fibrils that are free from entanglement.
Iodine doping was performed by exposing iodine gas to the PA film at room temperature for 24 h in a glass vessel. The atomic ratio of doped iodine to carbon in the PA film (I/C) was 0.26?0.30%. The doped PA film was placed between graphite plates (80 × 80 × 2 mm) and was inserted into an electric furnace (KDF75, Denken). The doped PA film was then carbonized at 800℃ for 1 h under argon gas atmosphere. The hydrogen content of the carbon film after carbonization was less than 1.0 wt%. The carbonization yield of the carbon film is about 70?90% of the weight of the PA film. Fig. 4 provides SEM images of the vertically aligned carbon films prepared by carbonization of iodine-doped PA film. Interestingly, the vertically aligned morphology of the PA film was maintained after carbonization. The fibril length of the carbon film was controlled at 10?35 m by changing the acetylene polymerization time from 30 min to 6 h. The maximum value of electrical conductivity of the vertically aligned carbon film was 200 S/cm.
Vertically aligned PA film was synthesized in a homeotropic N-LC. A homeotropic N-LC of the mono-domain structure was constructed with a magnetic field of 5 Tesla. The fibrils of PA grow parallel to the magnetic field. We successfully demonstrated the synthesis of vertically aligned carbon film using a vertical PA film as a carbonization precursor, with a high carbonization yield of 70?90%. A more detailed carbonization mechanism using iodine doping was discussed in another report [6,7]. It is expected that, by virtue of their electrical conductivity, surface area, porosity, and gas permeability, these vertically aligned carbon materials might be useful for many applications in such areas as electrochemical energy storage, cell electrodes, and gas absorbent materials.