Kelps (Order Laminariales) form the basis of essential habitats in temperate coastal ecosystems because of their high productivity and function as spawning and nursery grounds for many invertebrate and fish species (Steneck et al. 2002). However, intense grazing can severely com-promise kelp abundance and function (Scheibling et al. 1999). Mesograzers of the nearshore marine environment such as gastropods (Iken 1999, Granado and Caballero 2001) are capable of affecting the growth and reproduc-tive abilities of individual macroalgal species (Dean et al.1984, Dethier et al. 2005) and even the composition and density of macroalgal communities (Carney et al. 2005). Mesograzer habitat and food choice is often attributed to algal morphological characteristics like shape, size and tissue toughness (Littler and Littler 1980, Steneck and Watling 1982, Hay et al. 1994), and to chemical properties related to nutritive value (Cruz-Rivera and Hay 2003) and anti-herbivore defense (Paul et al. 2001, Amsler 2008).
Kelps usually contain phlorotannins, phloroglucinol-based polymers present in most brown algae (Ragan and Glombitza 1986, Amsler and Fairhead 2006), which have primary metabolic roles in wound healing (Luder and Clayton 2004) and cell wall construction (Schoenwaelder and Clayton 1999). Phlorotannins also have been assigned secondary functions such as protection against UV radia-tion (Pavia et al. 1997), bacterial and fungal growth and fouling (Ragan and Glombitza 1986, Wikstrom and Pavia 2004, Iken et al. 2009), and grazing damage (for reviews see Targett and Arnold 1998, Amsler and Fairhead 2006). The function and effectiveness of phlorotannins as feed-ing deterrents may differ by algal and herbivore species, with some studies demonstrating grazing reduction or in-hibition (Geiselman and McConnell 1981, Pavia and Toth 2000a) and others showing negligible responses or even increased herbivory (Steinberg and van Altena 1992, Deal et al. 2003, Kubanek et al. 2004).
Phlorotannin content in brown algae can be regulat-ed by a variety of abiotic and biotic variables, with large species-specific variability in responses to these factors (reviewed by Amsler and Fairhead 2006), which may act synergistically to affect distribution of phlorotannins within species and within thalli (e.g., Hay 1996, Targett and Arnold 1998). Phlorotannin content may be influ-enced by temporal variability in physical factors such as irradiance (Pavia and Toth 2000b, Cruces et al. 2012), sa-linity (reviewed by Ragan and Glombitza 1986), nutrients (Yates and Peckol 1993, Cronin and Hay 1996, Svensson et al. 2007), and water movement (Dayton 1985). Addition-ally, the risk of attack by grazers may affect patterns of phlorotannin content (Rhoades 1979, Amsler 2001). For example, the presence of grazers or mechanical wound-ing may affect patterns of phlorotannin production if the macroalgal species in question demonstrates induc-ible defenses (Hammerstrom et al. 1998, Pavia and Toth 2000a, Jormalainen et al. 2003, Hemmi et al. 2004).
In the present study, we investigated seasonal phloro-tannin content of the annual, canopy-forming kelp spe-cies Nereocystis luetkeana (Mertens) Postels et Ruprecht, and the perennial, understory species Agarum clathra-tum Dumortier, Saccharina latissima (Linnaeus) C. E. Lane, C. Mayes, Druehl & G. W. Saunders and S. groen-landica (Rosenvinge) C. E. Lane, C. Mayes, Druehl & G. W. Saunders in the Northeast Pacific (Alaska). We analyzed phlorotannin content in relation to seasonal environ-mental variables (light and nutrients). We further deter-mined seasonal field densities and laboratory grazing by the gastropod Lacuna vincta on these kelp species. These kelp and grazer species were chosen due to their ubiquity within the study area (Dubois 2006, Chenelot and Konar 2007, Konar et al. 2009). The grazer is known to be able to decimate algal biomass and to be a structuring force for macroalgal communities (Fralick et al. 1974, Johnson and Mann 1986, Chenelot and Konar 2007). Our objectives were to assess relationships between phlorotannin levels, environmental conditions and grazing rates, and to de-termine if these relationships differed between seasons in species with different life history strategies.
Field studies occurred off the western end of Hesketh Island (59°30.3′ N, 151°31.8′ W) within Kachemak Bay, south-central Alaska. The study site was located on ex-posed shores approximately 5 m below mean lower low water. Common kelp species in the study area include the canopy-forming annual kelp Nereocystis luetkeana and the perennial understory kelps Agarum clathratum, Saccharina latissima and S. groenlandica. The perennial kelp species may persist through the winter as complete individuals or as stipes (O’Clair and Lindstrom 2000, Chenelot 2003). The gastropod grazer L. vincta is season-ally abundant and has been reported to have significant grazing impact on canopy (Duggins et al. 2001, Chenel-ot 2003, Carney et al. 2005) and understory kelp thalli (Fralick et al. 1974, Johnson and Mann 1986, Krumhansl and Scheibling 2011).
Collection of kelp thalli occurred at Hesketh Island ev-ery two weeks in summer (June to September of 2004 and 2005) and monthly in winter (December 2004 to March 2005). L. vincta densities were measured on the same sampling dates during 2005. Gastropod density was de-termined within three 100 cm2 quadrats per blade (one blade haphazardly chosen for N. luetkeana) of three ran-domly selected individuals per kelp species.
The same kelp individuals observed for gastropod den-sities were then removed from the substrate below the holdfast and kept in cold seawater in the dark for trans-port (n = 3 thalli per species). Thallus portions (<1.5 g each) were sampled for phlorotannin analysis from the stipe and blades. Excised tissue was weighed and kept frozen at -20℃ until phlorotannin analysis. In addition, larger amounts of blade material (~500 g) were collected for each species and kept frozen for phlorotannin stan-dard preparation.
Preparation of phlorotannin standards and analysis of kelp tissue phlorotannin content followed the 2, 4-dime-thoxybenzaldehyde (DMBA) method described by Stern et al. (1996). Briefly, phlorotannin standards were made by grinding (Power Gen 500 homogenizer; Fisher Scien-tific, Hampton, NH, USA) and extracting frozen kelp tis-sue three times in 80% methanol for 12 h. The combined extracts were filtered (8 μm cellulose filters, Whatman; Fisher Scientific, Hampton, NH, USA), evaporated under reduced pressure at 35℃ and further purified using sub-sequent elutions with toluene and acetone (Stern et al. 1996). Each standard was freeze dried for 48 h and then ground to a fine powder. Two phlorotannin standards were made for each species. Replicate standard curves for both standards for each species were determined using a 0-500 μg μL-1 range of phlorotannin mass to methanol volume. The use of a mean slope from multiple replicates accounted for some procedural variability in standard curves, though differences among replicates were very low.
For phlorotannin determination in kelps, thallus sam-ples (stipe and blade for seasonal collections, only blade for feeding assay collections ? see below) were homoge-nized separately and 80% methanol-extracted overnight. Phlorotannin measurements were conducted on a solu-tion of the DMBA assay working reagent (Stern et al. 1996), 200-400 μL of extract (depending upon kelp species) and N, N-dimethylformamide. Solutions were incubated and absorbance determined spectrophotometrically at 510 nm (Thermospectronic Genesys 5; Cole-Parmer, Vernon Hills, IL, USA) against blanks of the same volume in all as-says (Stern et al. 1996). Calculation of phlorotannin con-tent as percent dry mass (% DM) was based on the ratio of wet : dry mass for stipes and blades of each kelp species (n = 6) after drying at 60℃ for 48 h.
Laboratory feeding assays with L. vincta were conduct-ed during the summer (August) of 2004 and 2005 to test for the palatability of kelp blades of the four species in non-choice experiments. Grazers were collected from ar-eas adjacent to the study site and allowed to clear guts for 24 h in running seawater tanks. Prior to the start of each experiment, ten individuals per kelp species were col-lected and three 1.5 ± 0.1 g blade pieces excised adjacent to each other from the meristematic region of each kelp individual. One of these pieces was used in the feeding treatment, the second in the control treatment and the third was frozen for corresponding phlorotannin analysis (for methods see above). Fifty L. vincta of average size (~6 mm) were placed in a treatment container (n = 10) with a piece of kelp blade. The quantity of L. vincta was based on the maximum density observed in the field on a blade area similar to that used in feeding assays. A paired con-tainer per kelp species and replicate held only a match-ing blade segment (no grazers) as a control to account for autogenic weight changes due to growth or decay (Peter-son and Renaud 1989). Containers were floated in flow-through seawater tanks to maintain ambient temperature regimes, and water within containers was replaced every 24 h. Relative mass change after 72 h was determined by the difference in wet mass exposed to grazers minus the mass change of ungrazed control blade segments.
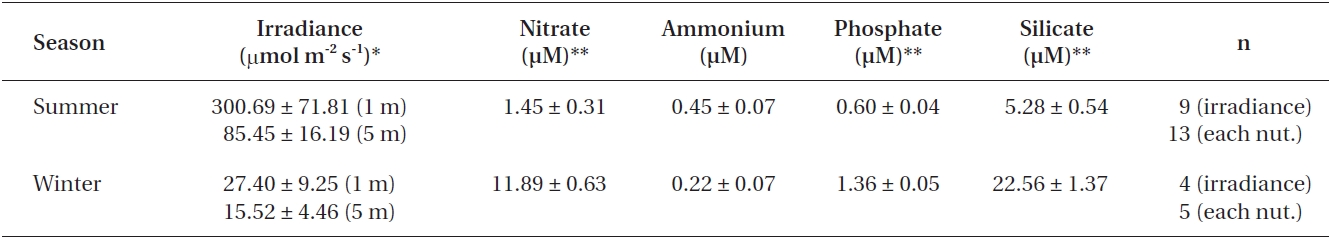
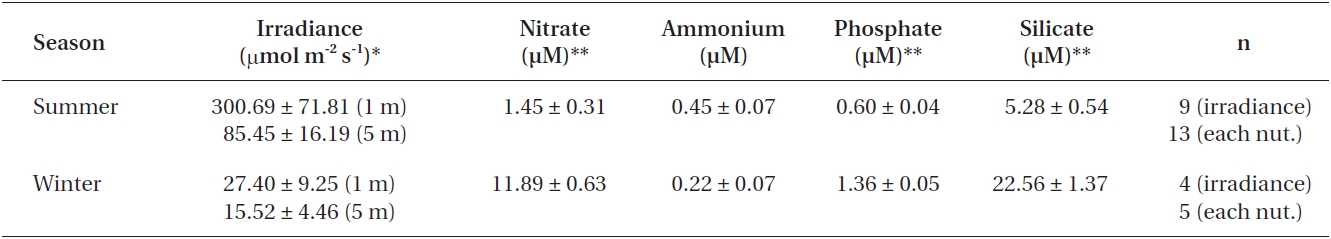
Nutrient concentrations and light attenuation were measured at each field sampling. Water samples obtained ~1 m above the substrate were used for analysis of nitrate, ammonium, phosphate and silicate concentrations us-ing an Alpkem model RFA-300 continuous nutrient ana-lyzer (Alpkem Co., Clackamas, OR, USA). Light profiles were taken with a LiCor 193SA spherical sensor (Li-Cor, Lincoln, NE, USA) to determine irradiance (μmol m-2 s-1) just above and below the water surface and at 5 m depth. Since N. luetkeana blades are close to or at the water sur-face for a large proportion of their life cycle, percent light attenuation at 1 m depth was used for this species, while the 5 m depth measurements were taken to approximate attenuation within the understory kelp canopy. Tempera-tures from a HOBO data logger (Onset Computers) at the study site were used to establish “summer” (10.3 ± 0.10℃, June-September) and “winter” (4.88 ± 0.07℃, December-March) seasons.
Prior to statistical assessments, parametric assump-tions for analyses of variance (ANOVA) were evaluated using residual plots and Shapiro-Wilk tests (Zar 1999). L. vincta grazer density data were left untransformed and phlorotannin content data were arcsine-square root transformed to improve error variance. Grazer density on blades of each of the four kelp species was analyzed for between-season differences using single-factor ANOVA (season as the orthogonal factor). Single-factor ANOVA were also used to separately assess between-season dif-ferences in phlorotannin content in stipe and blades of each of the four kelp species. Untransformed tissue mass and phlorotannin data from palatability assays were ana-lyzed by single-factor ANOVAs with kelp species as the fixed factor. All post-hoc assessments of significant ef-fects and interactions were made using the Tukey-Kramer method. Between-season comparisons of environmental data were conducted using single-factor ANOVA (season
as the orthogonal factor) on untransformed irradiance and log-transformed nitrate, ammonium, phosphate and silicate. Pearson’s product-moment correlations were used to separately determine the relationship between blade phlorotannin content with grazer density and en-vironmental variables (light and nutrients) for each kelp species. Phosphate and silicate were excluded from this analysis due to high correlation (p < 0.01) with nitrate. All statistical tests were conducted using SAS, v9.1 (SAS Insti-tute Inc., Cary, NC, USA) at α = 0.05.
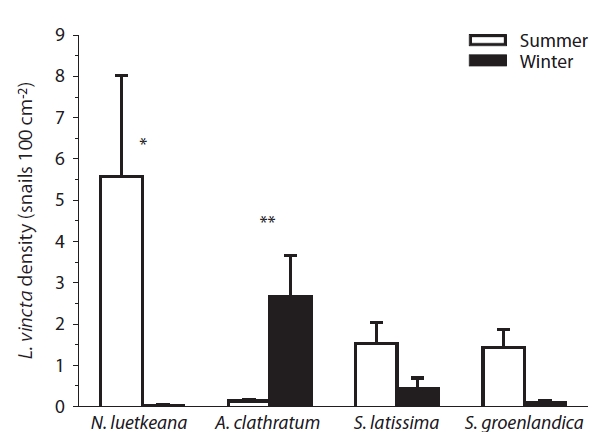
Seasonal patterns of L. vincta were not consistent among kelp species. During summer, snail density was significantly higher than during winter on N. luetkeana (p = 0.03), and higher but not significantly on S. latissima and S. groenlandica. Conversely, L. vincta density was sig-nificantly higher during winter than summer on A. clath-ratum blades (p < 0.01, Fig. 1).
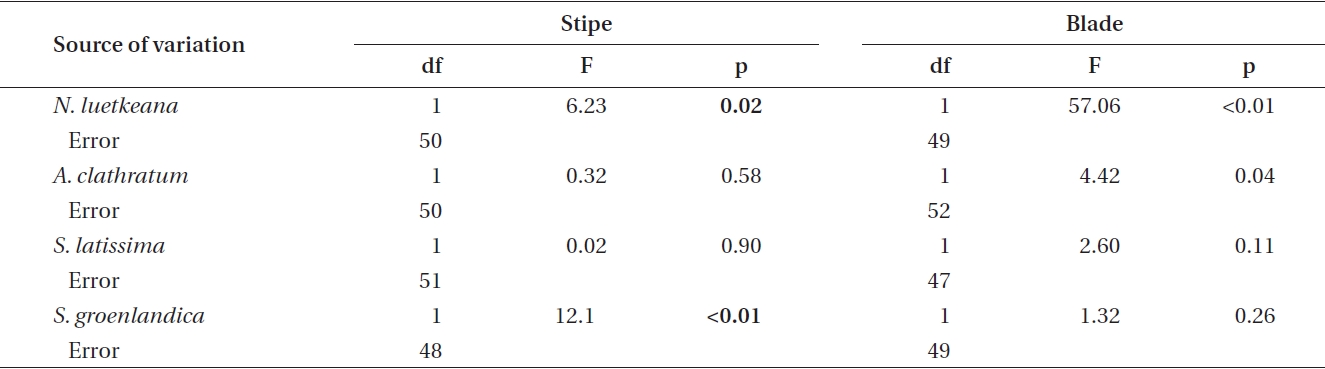
Phlorotannin content in stipes and blades of the four kelp species was not consistently higher in either summer or winter (Fig. 2). N. luetkeana was the only kelp species that demonstrated a significant difference in phlorotannin content between seasons in both thallus parts, with higher content in summer in stipe and blade (Table 1, Fig. 2).
A. clathratum had similar phlorotannin concentrations between seasons in stipes and significantly higher concentrations in summer in blades. Conversely, S. latissima and S. groenlandica phlorotannin content was typically higher during winter in both thallus parts, but with the only significant difference in S. groenlandica stipes (Table 1, Fig. 2).
As much as 30% of the total wet mass of kelp tissue was consumed by L. vincta within 72 h (Fig. 3A). Lacuna vincta consumed significantly more blade tissue of N. lu-etkeana than of any understory species (p < 0.05). Grazing was lowest on the two Saccharina species. Phlorotannin content of tissues used in feeding experiments also var-ied significantly between kelp species (p < 0.01). Blade phlorotannins were generally low (<2.0% DM) in all kelp species, with concentrations in N. luetkeana and S. latis-
sima less than 1% DM (Fig. 3B). The greatest consump-tion occurred in the species containing the lowest phloro-tannin content (N. luetkeana), but no overall relationship between snail grazing and tissue phlorotannin content existed (p > 0.05).
Irradiance was higher during summer and decreased in winter by ~80-90% at 1 and 5 m depths (Table 2). With the exception of ammonium, all other nutrient concentrations were significantly different between seasons (Table 2), with higher concentrations in winter than summer (p < 0.01).
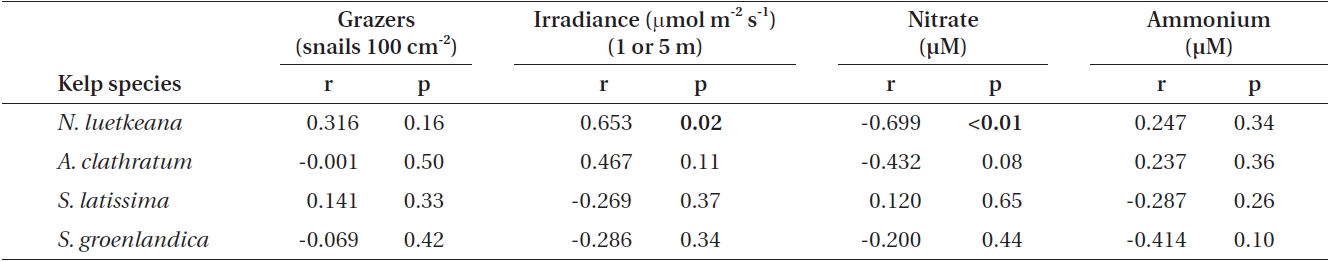
Pearson’s product-moment correlations between blade phlorotannin content and L. vincta density were not significant for any kelp species (Table 3). Correlation analyses of blade phlorotannin content of the four kelp species with irradiance, nitrate, and ammonium demonstrated significant relationships only for N. luetkeana (Table 3). N. luetkeana phlorotannins were positively cor-related with irradiance at 1 m (r = 0.653, p = 0.02) and neg-atively correlated with nitrate at 5 m (r = -0.699, p < 0.01). No significant relationship existed between N. luetkeana phlorotannins and ammonium. None of the correlations between A. clathratum, S. latissima and S. groenlandica blade phlorotannins and environmental variables at 5 m were significant and varied in terms of the direction of the relationship between factors (Table 3).
Northeastern Pacific kelps from this study exhibited detectable phlorotannin concentrations in stipe and blade tissues throughout the duration of field surveys. Mean annual phlorotannin values of all kelp species ap-proximate those measured previously in northern Pa-cific or Atlantic brown algae (Ragan and Glombitza 1986, Targett et al. 1992, Van Alstyne et al. 1999), though the remarkably high content in S. latissima stipes is unprec-edented. The ecological and physiological significance of these high phlorotannin values is as of yet unexplained. However, it is unlikely that methodological problems with stipe phlorotannin assays caused inflated values since the same laboratory procedures were used to pro-duce phlorotannin values for all kelp species and thallus parts, all of which are within range of those reported in the scientific literature. We also repeated these measures several times with identical results. Higher phlorotan-nin concentrations in stipes, which is the perennial thal-lus part in S. latissima (O’Claire and Lindstrom 2000), or holdfasts than in blades has been observed before in sev-eral macroalgal species (e.g., Ragan and Glombitza 1986, Tugwell and Branch 1989, Iken et al. 2007). The large vari-
ability in phlorotannin content in the studied kelps is also similar to other observations of substantial differences within (e.g., Pavia et al. 2003, Fairhead et al. 2005, Iken et al. 2007) and between (e.g., Ragan and Glombitza 1986, Van Alstyne et al. 1999) species.
L. vincta was overall more abundant in the summer than winter, indicating seasonal rather than continuous recruitment in the study area (also see Maney and Eb-ersole 1990, Martel and Chia 1991). Gastropods during summer were on average more abundant on the low-phlorotannin containing thalli of N. luetkeana and less frequent on the thalli of those species containing higher phlorotannins (especially A. clathratum), despite N. luet-keana being, on average, the least abundant of the four kelp species within the study area (Dubois 2006). In win-ter, snail densities were highest on A. clathratum, possi-bly because this perennial species maintains much of its blade structure during the winter. In contrast, most of the annual N. luetkeana thalli disappear in winter and the two Saccharina species mostly overwinter with reduced blade portions. Hence, seasonal kelp abundance might influ-ence L. vincta’s preference for A. clathratum in the winter. In the northern Gulf of St. Lawrence, Canada, however, L. vincta was more abundant on A. cribrosum than several other macroalgae also in the summer (Begin et al. 2004).
In our palatability assays, L. vincta grazed significantly more on N. luetkeana blades with the lowest phlorotannin levels of the studied kelp species, similar to the high grazing rates on juvenile N. luetkeana previously reported for the study area (Chenelot and Konar 2007). While these relationships may indicate grazer avoidance of phlorotannin-rich kelps, consumption by L. vincta in feeding assays was not consistently proportional to phlorotannin content of each kelp species (Table 3). Therefore, other or additional factors, such as tissue toughness or nutri-tive value, may have affected kelp palatability (Chavanich and Harris 2002). Based on the lack of a consistent rela-tionship between L. vincta and phlorotannin content in the four kelp species in both field and laboratory obser-vations, it is plausible that this grazer is not significantly deterred by just the presence of phlorotannins in kelps. Phlorotannins can have concentration-dependent effects on gastropods (Johnson and Mann 1986, Pavia and Toth 2000a), and mean phenolic levels of 2% DM have previ-ously been suggested as the lower concentration limit that deters grazing (Hay et al. 1994). When offered phe-nolic-rich and -poor algal species, the gastropod Tegula funebralis preferentially grazed on individuals with phlo-rotannin content less than 1.65% DM (Steinberg 1985). L. vincta avoided Saccharina longicruris meristematic blade tissue (approximately 5.5% DM phlorotannins) but grazed on all other tissue types with phlorotannin levels less than 1% DM (Johnson and Mann 1986). Most kelp species tested in the present study had blade phlorotan-nin concentrations of 1-3% DM, which may have been too low to elicit a feeding deterrent effect. Unfortunately, experiments using various concentrations of isolated phlorotannins could not be performed in the present study because L. vincta did not respond to artificial food pellets. Therefore, a causal relationship between phloro-tannins as quantitative chemical deterrents and L. vincta grazing could not be established.
Some macroalgal species are able to induce chemi-cal defense levels such as phlorotannins in the presence of grazers, particularly small mesograzers (Amsler 2001, Toth and Pavia 2007). Hence, alternative to the explana-tion that L. vincta densities were not strongly driven by the presence and concentration of phlorotannins in the studied kelps, it also is possible that kelps did not respond appreciably to the presence of L. vincta by increasing their blade phlorotannin content. This explanation could indicate that the studied kelp species may not have the ability to induce phlorotannin production in response to grazers, or just not in blade tissue (Taylor et al. 2002). L. vincta also could not be an appropriate grazer to trigger induction in these kelp species as induction can be graz-er-specific (Amsler 2001). Inducible phlorotannin pro-duction has been identified for some brown algae (Van Alstyne 1988, Toth et al. 2005, Toth 2007, see Toth and Pavia 2007 for overview), including several kelp species (Hammerstrom et al. 1998, Molis et al. 2006, 2008), but not in other kelp species (Steinberg 1994, Toth and Pavia 2002, Macaya and Thiel 2008). Varying macroalgal re-sponses of phlorotannin induction may be due to differ-ences in the mechanism of wounding by particular graz-ers (reviewed by Amsler 2001). Alternatively, variation in ambient environmental variables such as irradiance and nitrogen levels may be important in triggering phlorotan-nin production (Pavia and Toth 2000b, 2008, Cronin 2001, Jormalainen et al. 2003).
The analysis of environmental factors demonstrated that N. luetkeana phlorotannins were significantly nega-tively correlated with nitrate and positively with light attenuation. In contrast, none of the phlorotannin con-centrations in the perennial understory species were significantly related to any of the irradiance or nutrient measurements. Hence, if environmental variables affect-ed phlorotannin production in our study species, then this was not based on general but on species-specific patterns. As an annual species, N. luetkeana has limited time to attain its mature thallus length and complete its life cycle, and the necessary fast growth rates are strongly dependent on the availability of sufficient light and nu-trients. Possibly, overall low phlorotannin content in this species is due to an r-selected life history where energy is mostly allocated towards growth and reproduction in-stead of defenses. However, excess photosynthesis (high light conditions) but slowed growth (nitrogen limitation) in the summer may result in the production of carbon-based compounds such as phlorotannins in an annual kelp such as N. luetkeana. This is in agreement with some plant resource allocation theories, such as the carbon-nutrient balance hypothesis, which explains phenotypic expression of defenses being driven by resource avail-ability (Bryant et al. 1983, Tuomi et al. 1991, Stamp 2003). However, the predictive and explanatory values of this particular hypothesis are debated (e.g., Hamilton et al. 2001, Koricheva 2002), and given the low overall phloro-tannin concentrations in N. luetkeana and the obvious lack of grazer deterrence, we conclude that phlorotannin patterns in this species are not likely to be driven by evo-lutionary mechanisms of defense allocation as predicted by the carbon-nutrient balance hypothesis. Likewise, the ecological meaning of seasonal variation of the overall low phlorotannin content (<1% DM) as grazer defense is probably low, although we cannot discount that phlo-rotannins play other roles in assisting with N. luetkeana fitness.
Conversely, none of the seasonal phlorotannin pat-terns in the three perennial species were related to en-vironmental variables, and overall phlorotannin con-centrations were higher than in the annual N. luetkeana. These perennial species may exhibit more of a K-selected life strategy, where competitive and other biological pres-sures can drive the production of mostly constitutive (consistently expressed) defenses, especially in the pe-rennial thallus parts (Herms and Mattson 1992, Astrom and Lundberg 1994, Pavia et al. 2002, Toth et al. 2005); such constitutive defenses are less variable in response to grazer abundance and environmental conditions than induced defenses. In support of this, the low feeding rates of L. vincta on the understory species seems to indicate that phlorotannins in these species indeed may have some grazer deterrent function.
The results from the current study do not indicate a strong correlation between kelp phlorotannin content and L. vincta density and grazing on the four studied kelp species within Kachemak Bay. Likewise, seasonal envi-ronmental variables seem to play a minor, if any, role in kelp phlorotannin concentrations. Since the perennial understory species had much higher concentrations and in some cases inverse seasonal pattern of phlorotannin content as compared to the annual N. luetkeana, it seems that life history strategy may be a stronger determinant of phlorotannin concentrations in these North Pacific kelps than grazers or environmental conditions.





![Phlorotannin content (% dry mass [DM]; mean ± 1 SE) of Nereocystis luetkeana, Agarum clathratum, Saccharina latissima and Saccharina groenlandica stipe (A) and blade tissue (B) (n = 5-36 per thallus part) during the summer and winter at Hesketh Island. Significant differences in phlorotannin content between seasons are denoted by *0.01 < p < 0.05 and **p ≤ 0.01. Note difference in y-axis scales between thallus parts.](http://oak.go.kr/repository/journal/11251/JORHBK_2012_v27n1_9_f002.jpg)
![Lacuna vincta grazing (% WM ± 1 SE) (A) and phlorotannin content (% dry mass [DM]; mean ± 1 SE) (B) of Nereocystis luetkeana, Agarum clathratum, Saccharina latissima and Saccharina groenlandica blade segments (n = 10 per species). Letters above bars indicate significant differences among species (p < 0.05).](http://oak.go.kr/repository/journal/11251/JORHBK_2012_v27n1_9_f003.jpg)
![Results of single-factor ANOVA of phlorotannin content (% dry mass [DM]) in Nereocystis luetkeana, Agarum clathratum, Saccharina latissima and Saccharina groenlandica stipe and blade during summer (June-September) and winter (December-March)](http://oak.go.kr/repository/journal/11251/JORHBK_2012_v27n1_9_t001.jpg)
![Results of Pearson’s product-moment correlations of blade phlorotannin content (% dry mass [DM]) in Nereocystis luetkeana, Agarum clathratum, Saccharina latissima and Saccharina groenlandica, with grazer density, irradiance (n = 13 per species), nitrate, and ammonium (at 5 m; each n = 17 per species)](http://oak.go.kr/repository/journal/11251/JORHBK_2012_v27n1_9_t003.jpg)