The thermostability of antioxidant activity of dieckol, a phlorotannin isolated from brown seaweed Ecklonia cava was investigated. The thermostable antioxidant properties of dieckol were evaluated at 30, 60, and 90℃ for 7 days using 1,1-diphenyl-2-picrylhydrazyl (DPPH) and hydroxyl radical scavenging activities, and comparing its performance to that of ascorbic acid. The intracellular reactive oxygen species (ROS) scavenging activity and apoptotic body formation were investigated using DCF-DA assay and nuclear staining with Hoechst 33342, propidium iodide and flow cytometry. Dieckol treated at different temperatures during 7 days showed stable scavenging activities on towards DPPH and hydroxyl radicals. In addition, dieckol showed a stable protective effect against H2O2-induced apoptotic body formation in Vero cells. On the other hand, the radical scavenging activities and intracellular ROS scavenging activities of ascorbic acid, used as a positive control, were significantly decreased at 60℃ and 90℃ from on the 4th day and 3rd days, respectively. In conclusion, the results indicated that food grade antioxidant extracts containing dieckol derived from E. cava remain a stable during the temperatures encountered during the processing of food and cosmetics.
According to economic development and the increasing diversity in lifestyle choices, the customer awareness of food and food additives had been significantly increased, and the use of functional and safe and natural food additives is required for an ever increasing majority of products. Many food processing technologies have been developed, but during the processing and storage the functional components, color or flavor may be changed (Fritsch 1981, Yang et al. 2000, Ukeda et al. 2002). The lipid oxidation caused by several unfavorable environmental conditions such as oxygen, temperature, and light is a serious problem in the food industry (Park et al. 2005). Lipid oxidation produces unpleasant flavors in foods and can also produce reactive oxygen species (ROS) that may lead to harmful effects such as cancer, diabetes, mellitus, liver injury, skin damage, aging, and inflammation. Therefore, many attempts have been made to cope with lipid oxidation of food (Frolich and Riederer 1995).
Over the past few decades, considerable numbers of antioxidant studies have been conducted by many researchers and they have tried to find more effective lipid oxidation inhibitors which can be used as antioxidants for food or medicine. Currently, synthetic antioxidant such as butylatedhydroxyanisol and butylatedhydroxytoluene have been used as ingredients or additives to suppress oxidation in food. Synthetic antioxidants are suspected as being carcinogenic and responsible for a variety of unidentified health disorders (Sherwin 1990). Therefore, the development of natural antioxidants which can prevent oxidation of food or medicine in a more effective yet safe manner is increasingly important. Recently, many studies have been conducted on marine organisms, including algae and animals, in order to determine the biological activities of metabolites with potential functional values. Among the biological activities of seaweeds, antioxidant activity has attracted more attention (Ye et al. 2009). Seaweeds are rich in vitamins, polysaccharides, and various functional polyphenols (Lordan et al. 2011). Therefore, and not surprisingly, they have been identified as a rich source of bioactive compounds (Dykens et al. 1992, Sukenik et al. 1993, Matsukawa et al. 1997).
The marine brown seaweed,
>
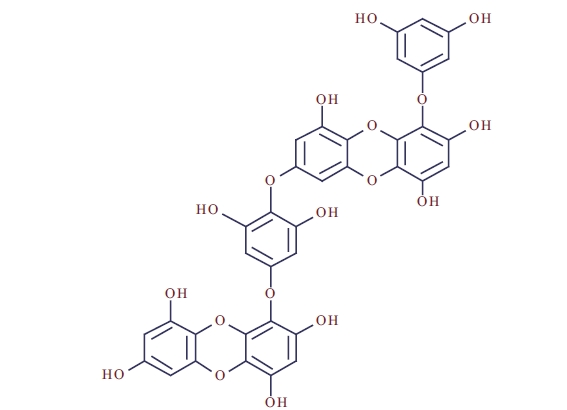
Isolation of dieckol from Ecklonia cava
The powdered
The purity of dieckol (Fig. 1) was >95%, based on the peak area of all components absorbed at each specific wavelength in HPLC analysis. Dieckol was dissolved in DMSO and was used for experiments adjusting the final concentration of DMSO in the culture medium to <0.01%.
To evaluate thermal stability, both dieckol and ascorbic acid were kept in the dark at 30, 60, and 90℃ for 7 days. The antioxidant activities were determined via DPPH and hydroxyl radical scavenging activities. The intracellular ROS scavenging activity was investigated using a DCF-DA assay and the apoptotic bodies were investigated via nuclear staining with Hoechst 33342 and cell cycle.
>
DPPH radical scavenging assay
The DPPH radical scavenging activity was measured using the method described by Nanjo et al. (1996). An ethanol solution of 60 μL each sample at the concentration of 62.5 μM and 125 μM (or ethanol itself as a control) was added to 60 μL of DPPH (60 μmol L-1) in ethanol solution. After mixing vigorously for 10 s, the solutions were then transferred to a 100 μL Teflon capillary tube which was fitted to the cavity of the electron spin resonance spectrometer (JEOL, Tokyo, Japan). The spin adduct was measured on an electron spin resonance (ESR) spectrometer exactly 2 min later. Measurement conditions: central field 3475 G, modulation frequency 100 kHz, modulation amplitude 2 G, microwave power 5 mW, gain 6.3 × 105, temperature 298 K.
>
Hydroxyl radical scavenging assay
Hydroxyl radicals were generated by Fenton reaction, and reacted rapidly with nitrone spin trap DMPO. The resultant DMPO-OH adducts were detectable with an ESR spectrometer. The ESR spectrum was recorded 2.5 min after mixing in a phosphate buffer solution (pH 7.4) with 0.3 M DMPO 0.2 mL, 10 mM FeSO4 0.2 mL, and 10 mM H2O2 0.2 mL using a JES-FA electron spin resonance spectrometer (JEOL) set at the following conditions: central field 3475 G, modulation frequency 100 kHz, modulation amplitude 2 G, microwave power 1 mW, gain 6.3 × 105, temperature 298 K.
The cells of an African green monkey kidney (Vero) were maintained at 37℃ in an incubator, under a humidified atmosphere containing 5% CO2. The cells were cultured in DMEM containing 10% heat-inactivated FBS, streptomycin (100 μg mL-1), penicillin (100 unit mL-1), and sodium pyruvate (110 mg L-1).
>
H2O2 scavenging assay by DCF-DA
For the detection of intracellular H2O2, the Vero cells were seeded in 96-well plates at a concentration of 1.0 × 105 cells mL-1. After 16 h, the cells were treated with dieckol samples (10 μL) and incubated at 37℃ under a humidified atmosphere. After 30 min, H2O2 was added at a concentration of 1 mM, and then the cells were incubated for an additional 30 min at 37℃. Finally, DCF-DA (5 μg mL-1) was introduced to the cells, and 2′,7′-dichlorodihydrofluorescein fluorescence was detected at an excitation wavelength of 485 nm and an emission wavelength of 535 nm, using a Perkin-Elmer LS-5B spectrofluorometer (Perkin-Elmer, Waltham, MA, USA).
>
Nuclear staining with Hoechst 33342 and PI
The nuclear morphology of the cells was evaluated using the cell-permeable DNA dye, Hoechst 33342 and PI. Cells with homogeneously stained nuclei were considered viable, whereas the presence of chromatin condensation and / or fragmentation was indicative of apoptosis (Gschwind and Huber 1995, Lizard et al.1995). The cells were placed in 24-well plates at a concentration of 1 × 105 cells mL-1 (950 μL). Sixteen hours after plating, the cells were treated with various concentrations of the compounds (50 μL), and were further incubated for 1 h prior to exposure to H2O2 (1 mM). After 24 h, 3 μL of Hoechst 33342 (stock 10 mg mL-1) and PI were added to each well, a DNA-specific fluorescent dye, were added to each well, followed by 10 min of incubation at 37℃. The stained cells were then observed under a fluorescence microscope equipped with a CoolSNAP-Pro color digital camera (Carsen Group, Markham, ON, Canada), in order to examine the degree of nuclear condensation.
To check whether dieckol reduces the proportion of apoptotic sub-G1 hypodiploid cells increased by ethanol treatment, PI staining assay was used according to a method suggested by Nicoletti et al. (1991). The Vero cells were placed in a 6-well plate at a concentration of 4.0 × 105 cells mL-1. The cells were treated with dieckol isolated from
>
Thermostability of dieckol on radical scavenging activities
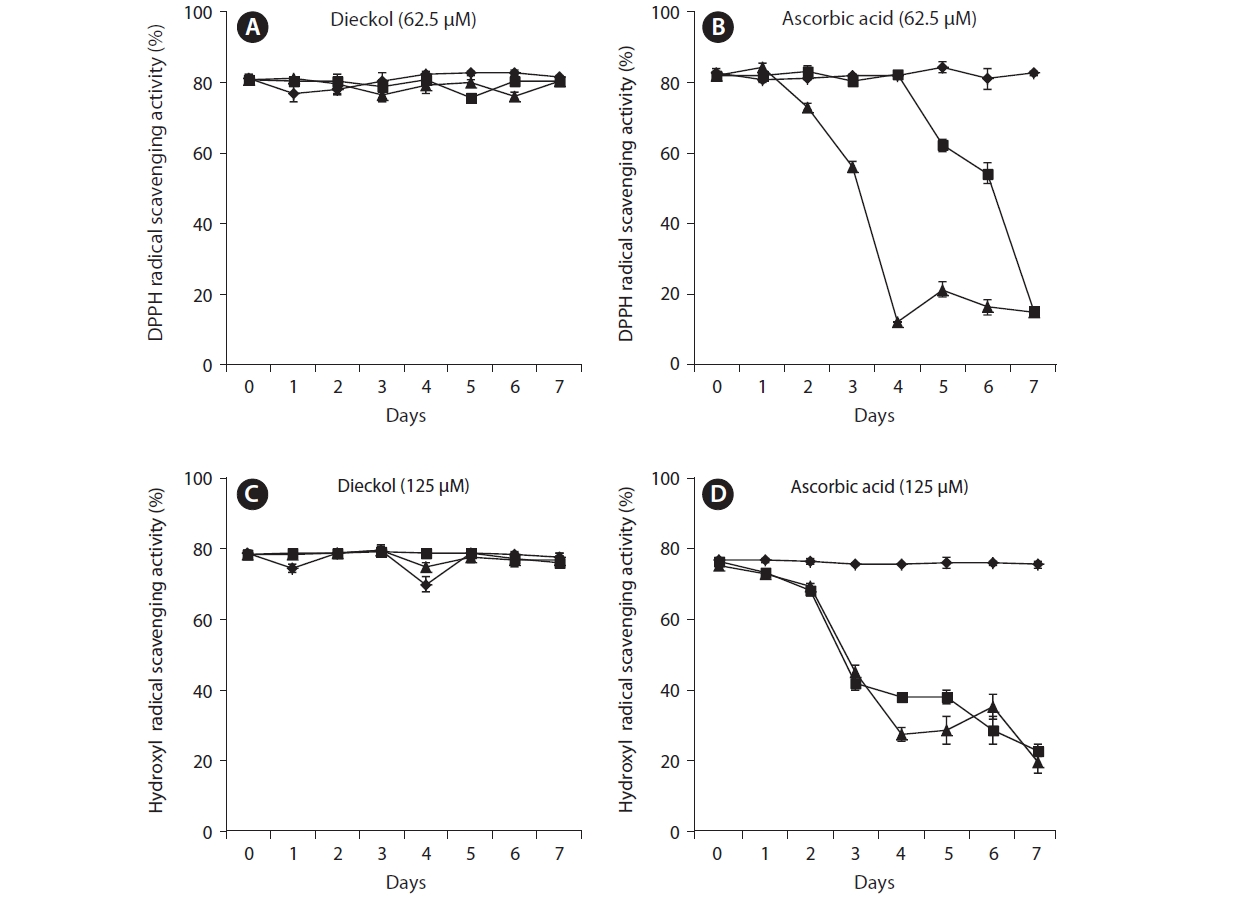
In this study, the thermostability of dieckol derived from
decreased over the seven day period at 60℃ and 90℃. Especially, on day 4 after heating at 90℃, the activity was decreased to 70.26%. On day 7, the activity was decreased to 66.81% and 67.61% at temperatures of 60℃ and 90℃, respectively (Fig. 2B). This result demonstrated that dieckol is more stable under conditions of heating than the positive control, ascorbic acid.
The thermostable patterns of dieckol and ascorbic acid (125 μM) during hydroxyl radical scavenging were similar to those during DPPH scavenging (Fig. 2C & D).
>
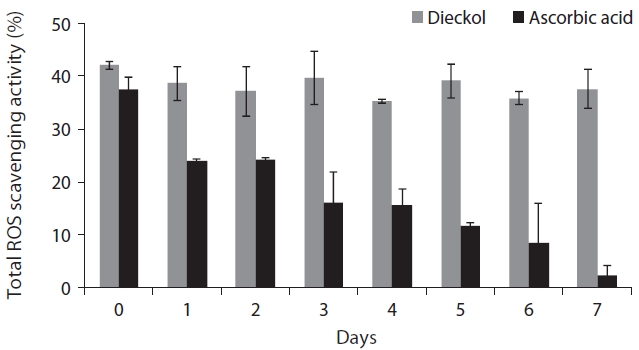
Thermostability of dieckol in intracellular ROS measurement
The changes of intracellular ROS scavenging activity of dieckol and ascorbic acid during 7 days at 60℃ were investigated and the results are exhibited in Fig. 3. In the case of dieckol, a slight change in ROS scavenging activity was observed, but there was no significant difference throughout the seven day experimental period. At day 0 the intracellular ROS scavenging activity of dieckol (12.5 μM) was 42.09% and at day 7 it was 37.5% However, the ROS scavenging activity of ascorbic acid at the same temperature dramatically decreased over time.
>
Protective effect of dieckol on H2O2 induced cell apoptosis
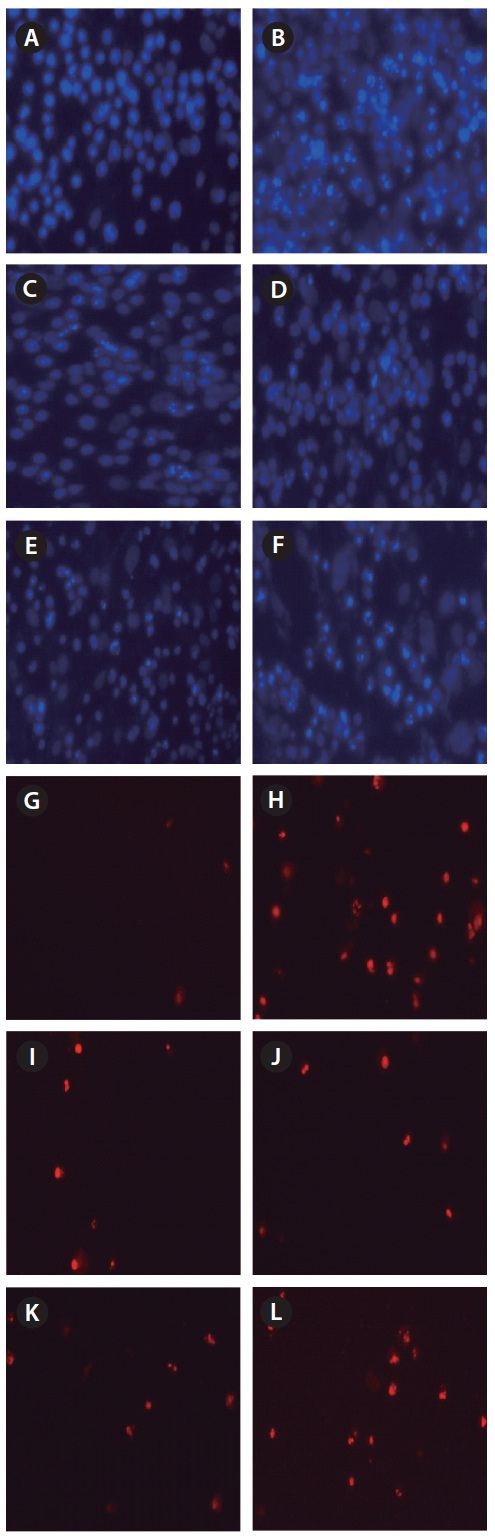
On the day 7 the changes of protective effects of dieckol and ascorbic acid against H2O2-induced cell apoptosis and necrosis were investigated via nuclear staining with Hoechst 33342 and PI. The microscopic photograph in Fig. 4A showed the uninjured nuclei and no necrosis. When the cells were treated with H2O2, a significant nuclear fragmentation and destruction which characteristics of
apoptosis (bright blue) and necrosis (red) were observed (Fig. 4B). However, cell damage, which was exhibited by both nuclear cleavage and necrosis, was significantly reduced for the cells treated with both dieckol and ascorbic acid at dat 0. On the 7th day, dieckol also showed a protective effect on the reduction of apoptosis and necrosis compared with H2O2-only treated cells. However, ascorbic acid showed no such effect after 7 days.
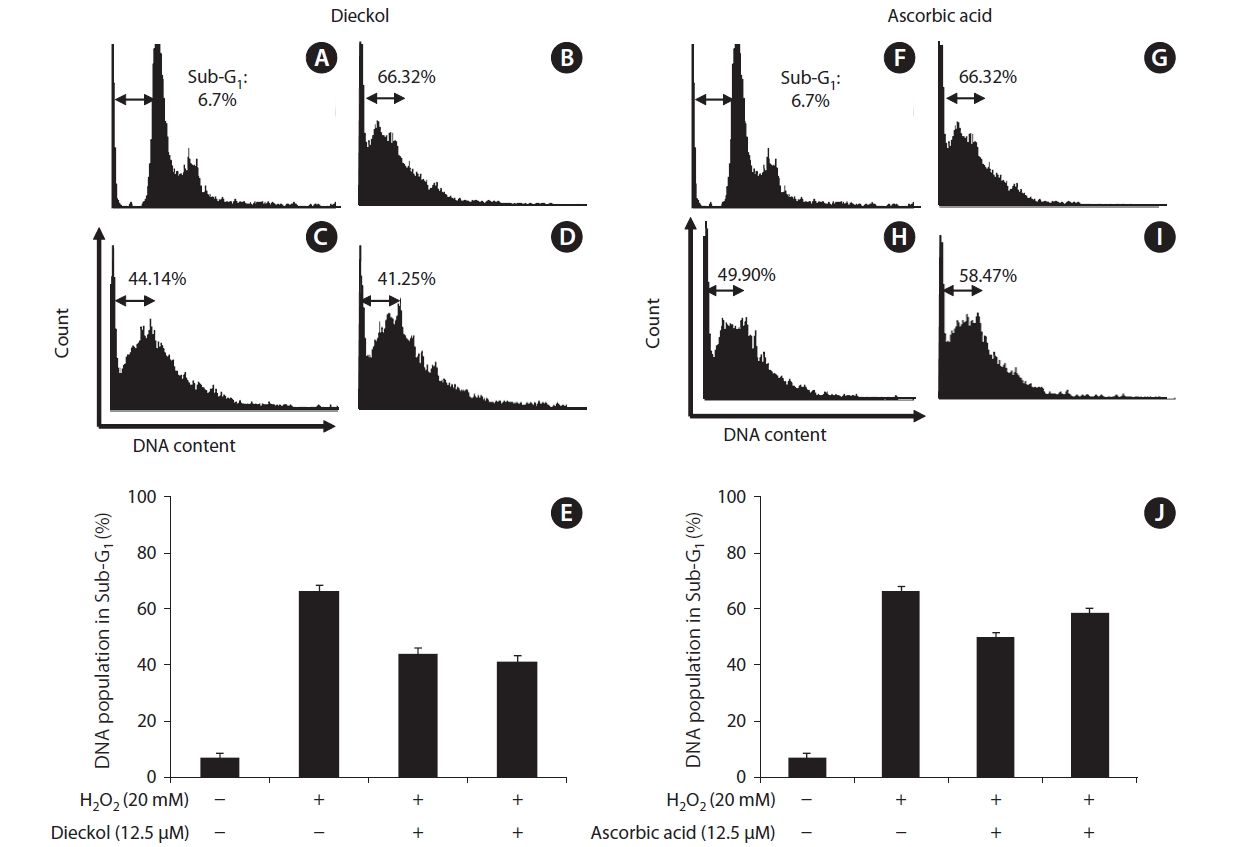
The changes of protective effects of dieckol and ascorbic acid on the influence of the cell population for 1 day were also measured by flow cytometry. In the control, the sub-G1 DNA content was 6.7% (Fig. 5A). When the cells were exposed to H2O2, the sub-G1 DNA content was increased to 66.32% (Fig. 5B). At 0 day, dieckol and ascorbic acid at the concentration of 12.5 μM reduced the sub-G1 DNA content to 44.1% and 49.9% (Fig. 5C & H). This indicated that both dieckol and ascorbic acid have the protective effect via the influence of the cell population distribution. At day 7, dieckol can reduce sub-G1 content to 41.25% in H2O2-treated cells (Fig. 5D). While cells treated with ascorbic acid for 7-days showed a slight reduction in the sub-G1 content (Fig. 5I).
Oxidation of food which occurs during storage of raw materials, food processing heat treatments, and the storage of the final products is one of the most common causes of food spoilage (Lehtinen et al. 2003). Oxidation by ROS such as superoxide anions, hydroxyl radicals, and hydrogen peroxide causes the decay of food, destruction of fat and vitamins and development of toxicants and off-flavors. In particular, the heating of food caused lipid oxidation (Laguerre et al. 2007). Ascorbic acid is widely used in the food industry as an antioxidant, and many researchers have reported the profound antioxidant effect of ascorbic acid over past decades (Cordenunsi et al. 2002). Ascorbic acid is an important water-soluble antioxidant (Frei et al. 1989). However, the antioxidant properties of ascorbic acid will be affected by thermal processing. In addition, ascorbic acid is also sensitive to light and oxygen (Gupta et al. 2008). Therefore, thermostable antioxidants are useful in various applications such as food processing industry. In addition, the use of thermostable natural antioxidants may have beneficial health implications (Amarowicz et al. 2000).
A wide variety of bioactive compounds and antioxidant properties have been reported for the brown seaweed
Dieckol exhibited a more stable scavenging effect when compared to ascorbic acid. From the results, it is clear that dieckol has a higher degree of thermostability at the tested temperatures. In addition, the phlorotannin compound effectively scavenged the intracellular ROS generation in Vero cells in response to H2O2 induced stress at 60℃ up to 7 days. Furthermore, the results of the current study confirmed that the dieckol isolated from
In conclusion, dieckol derived from