This study was performed to analyze the singledose toxicity of Aconitum kusnezoffii Reichb. pharmacopuncture (AKRP).
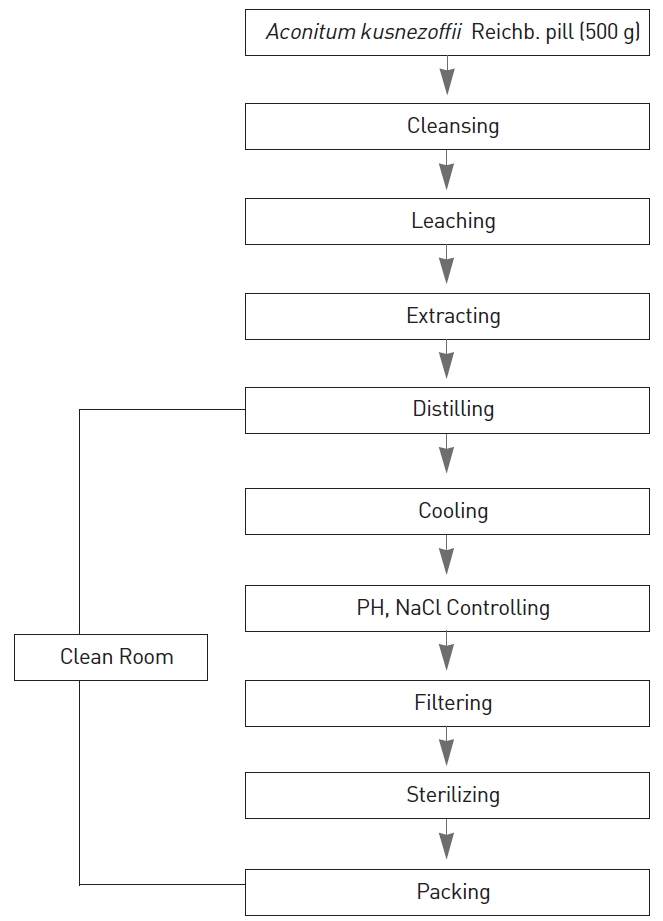
All experiments were conducted at the Korea Testing & Research Institute (KTRI), an institute authorized to perform non-clinical studies, under the regulations of Good Laboratory Practice (GLP). Twenty (20) Sprague-Dawley rats were chosen for the pilot study. The animals were divided into four groups of five animals per group: group 1 (G1) being the control group with each animal receiving an injection of 0.3 ml of saline and groups 2, 3, and 4 (G2, G3, and G4) being the experimental groups with each animal receiving an injection of 0.1, 0.2 or 0.3 ml of AKRP, respectively. This study was conducted with the approval of the Institutional Animal Ethics Committee.
No deaths occurred in any of the 4 groups, and the LD50 of AKRP administered via IV was higher than 1.77 ml/kg. Some changes in the weights of the male rates were observed between the control group and the experimental groups, but no significant differences were noted in the weights of the female rats. To check for abnormalities in organs and tissues, we stained representative sections of each specified organ with Hematoxylin & Eosin for light microscopic examination. The results showed no significant differences in any of the organs or tissues.
The above findings suggest that Aconitum kusnezoffii Reichb. pharmacopuncture is a relatively safe treatment. Further studies on the subject should be conducted to yield more concrete evidence.
A traditional Korean herbal medicine, Aconitum
The current research trend for single-dose toxicity testing of a pharmacopuncture is to study the acute and sub-acute toxicity through Good Laboratory Practice (GLP). All the experiments for this research were conducted at the Korea Testing & Research Institute (KTRI), an institute authorized to perform non-clinical studies, under the GLP.
The Animals used in this study were 6-week-old Sprague- Dawley rats. The mean weight of the male rats was 194.6 ± 5.8 g, and that of the female rats was 144.9 ± 7.3 g. For all animals, a visual inspection and weighing were done using a CP3202S system (Sartorius, Germany). After seven days of amllimatization, the rats’ general symptoms and changes of weight were recorded. No abnormalities were found. The temperature of the laboratory where the animals were kept was 21.6~23.7℃, and the humidity was 38.7~59.3%. The animals had amless to sufficient food (Teklad Certified Irradiated Global 18% Protein Rodent Diet 2918C) and water.
After the seven days of amllimatization, animals were selected and grouped by using the criteria of their weights being close to the mean weight. A total of 20 male rats and 20 female rats were selected, and the animals were distributed into 4 groups with 5 mice per group, as shown in Table 1.
The dose for the AKR pharmacopuncture (AKRP) which was 0.1~0.3 ml/ head, was determined from "The Study on Acute and Sub-acute Toxicity and Anti-cancer Effects of Cultivated Wild Ginseng Herbal Acupuncture" [7]. In the control group, 0.3-ml of normal saline solution was injected through IV at one point of the tail vein. This study was conducted under the approval of the Institutional Animal Ethics Committee.
On the day of dosing (day 0), the general symptoms (types of toxic symptoms, recovering time, etc.), and the mortality was examined at after 30 mins, and 1, 2, 4, and 6 h. From the 1st day to the 14th day of treatment, the general symptoms were examined once a day. The weights were measured immediately before treatment and at 1, 3, 7 and 14 days after treatment. After the termination of observation, all surviving animals were anesthetized with CO2 gas and phlebotomized at the abdominal aorta. The rats were then subjected to terminal necropsy. Principle organs (brain, liver, lungs, kidneys and spinal cord) and tissues were sampled at terminal necropsy and were fixed in 10% NBF (neutral buffered formalin). After fixation, paraffin embedding was conducted, and 3- to 4-㎛ sections were prepared by using routine histological methods. Representative sections of each specified organ were stained with Hematoxylin & Eosin for examination under a light microscope.
The weight results from the experiment were analyzed by using SAS (version 9.1.3, SAS Institute Inc., U.S.A.), and the
In this study, no deaths occurred by injecting AKRP, and the LD50 of AKRP administered via IV was found to be over 0.3 ml /head. In addition, no deaths or abnormal changes in clinical signs occurred in any of the groups (Tables 2 and 3); neither did any changes in weights, which were evaluated on the 3rd, 7th and 14th days after the injections (Table 4). No meaningful changes were noted on necropsy, and histopathological examination of all Group (0.3-ml saline/animal) showed no significant changes related to injections in the brain, lungs, liver, kidneys and spinal cord (
If the toxicity of general material is to be tested, the acute and the chronic harmful effects, as well as the relations to the capacity reaction, need to be studied more. To this end, animal testing is the most fundamental and basic way to assess safety. For the study of toxicity, we used the testing protocol guidelines from the Korea Food & Drug Administration [14], and all the experiments were conducted following the Good Laboratory Practice (GLP) regulations. All experiments were conducted at KTRI, an institute authorized to perform non-clinical studies.
No deaths occurred in any of the four groups, and the LD50 of AKRP administered via IV was higher than 1.77 ml/kg. Some changes in the weights of male rats were noted between the control group and the injection groups but no significant differences in weights were noted for the female rats. To check for abnormalities in organs and tissues, we stained representative sections of each specific organ with Hematoxylin & Eosin for examination under a light microscope. The results showed no significant differences in any organs or tissues.
The above findings suggest that
The object of this study was to analyze the single-dose toxicity of