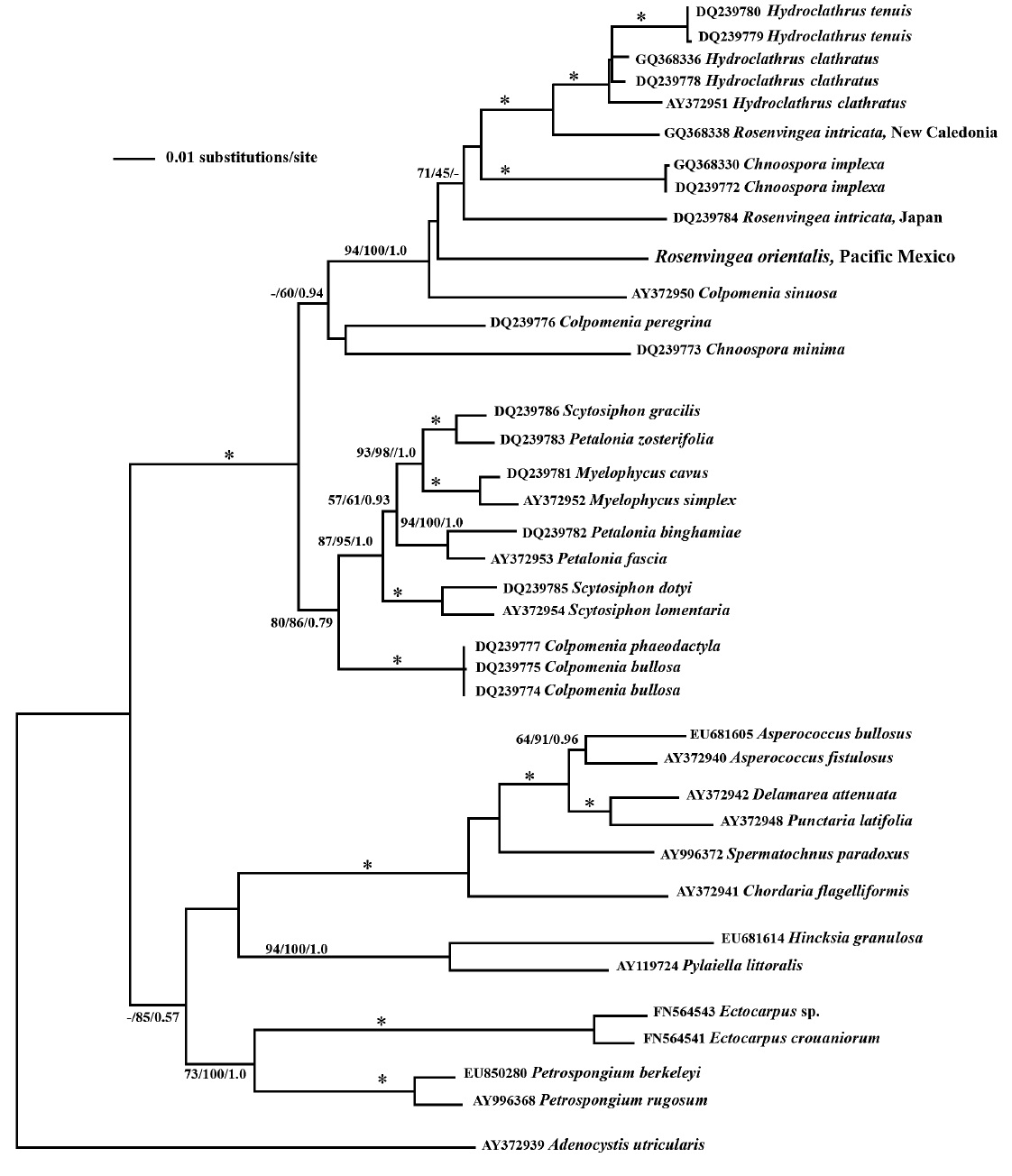
The genus Rosenvingea is well known in the tropics. Four species have been reported from Pacific Mexico: R. floridana,R. antillarum, R. intricata and R. sanctae-crucis. We collected a plant (Boca del Cielo, Chiapas) that we identified as Rosenvingea orientalis, a species not previously reported from Pacific Mexico. We were able to characterize the life cycle of this species for the first time in laboratory culture. It reproduced exclusively by plurilocular sporangia (plurangia). The mature plants were up to 6 cm long with cylindrical to compressed fronds (to 2 mm wide) with dichotomous branches in the upper half of the thallus. The medulla was hollow with 2-3 layers of large inflated colourless cells at the periphery.The cortex was comprised of 1 layer of small cells, each with a single chloroplast and pyrenoid. Linear plurangial sori with phaeophycean hairs formed along the mature fronds. Zoospore germlings developed into prostrate filamentous systems, each with a single phaeophycean hair that gave rise to a single erect shoot with multiple hairs arising near the tip. Molecular phylogeny using the psaA gene placed this isolate within the Scytosiphonaceae. It does not confirm the exact identification of R. orientalis, although its placement close to other Rosenvingea sequences was confirmed and morphological evidence supports its placement in R. orientalis. Our culture investigations indicated that it has an asexual life cycle. Further collections are needed to resolve the full generic and specific relationships of Rosenvingea and related taxa, and their reproductive patterns.
The brown alga
Seven species of
Molecular data presented by Cho et al. (2006), based on plastid-encoded
In Pacific Mexico
General culture methods are provided in West and Zuccarello (1999). The initial culture was established in a 50 × 70 mm crystallizing dish with 70 mL of 30-32‰ seawater, quarter-strength Modified Provasoli’s Medium (MPM) (West 2005) at 18-22°C, 2-3 ㎛ol photons m-2 s-1 cool white light emitting diodes, 12 : 12 light-dark diurnal cycle. For faster growth 30-35 ㎛ol photons m-2 s-1, a rotary shaker at about 70 rpm and full strength MPM were used. For slower growing low-light cultures the medium was changed every 3 wks. With faster growing cultures in 500 mL deep storage dishes the medium was changed weekly. To follow spore germination a 22 mm cover slip was placed in each 50 × 70 mm crystallizing dish with 70 mL of MPM and a 0.1 mL of spore suspension was added. Photos were taken every 3-4 days. Cell numbers of the basal system were counted and overall dimensions measured for 10-20 sporelings each time. Also the length and diameter of developing erect shoots were measured.
Most photography was done on live unfixed specimens using bright field optics on a Zeiss GFL microscope (Carl Zeiss, Thornwood, NY, USA) with a Canon G3 camera (Canon, Tokyo, Japan) and Photoshop CS (Adobe Systems Incorporated, San Jose, CA, USA) to capture the image. To more clearly see the arrangement of cells in mature shoot tips, a blade segment (2 mm long) was microwaved in distilled water for 20 s to shrink cells at the apex, mounted on a slide with a cover slip and then gently pressed to flatten and spread the tissue.
Total DNA was isolated from dried samples using a modified CTAB procedure (Zuccarello and Lokhorst 2005). The molecular marker used, following previous work on the family by Cho et al. (2006) was the plastid-encoded
PCR products were cleaned using ExoSAP-IT (USB Corp., Cleveland, OH, USA) and commercially sequenced (Macrogen, Inc., Seoul, Korea). Available sequence data of the Scytosiphonaceae for
Maximum-parsimony trees were constructed in PAUP* (Sinauer Associates, Inc., Sunderland, MA, USA), using the heuristic search option, 500 random sequence additions, tree-bisection-reconstruction branch swapping, unordered and unweighted characters, gaps treated as missing data and 1 million rearrangements per replicate. Support for individual internal branches was determined by bootstrap analysis (Felsenstein 1985), as implemented in PAUP*. For bootstrap analysis, 1,000 bootstrap data sets were generated from resampled data (10 random sequence additions).
Likelihood settings were calculated with Modeltest version 3.7 (Posada and Crandall 1998). The best fit model selected by Akaike information criterion. When
[Table 1.] Key to currently accepted species of Rosenvingea

Key to currently accepted species of Rosenvingea
the sequence evolution model had been determined, maximum-likelihood was performed in PAUP* using the estimated parameters (substitution model, gamma distribution, proportion of invariable sites, transition?transversion ratio and 5 random additions). Bootstrap values using maximum likelihood analyses were performed on PHYML, with 100 replicates and the same model parameters.
Bayesian analyses were performed using MrBayes version 3.0b4 (Huelsenbeck and Ronquist 2001) with a random starting tree and run for 3 × 106 generations in each of the two four-chain runs, keeping one tree every 100 generations. The first 0.5 × 106 generations (burn-in) were discarded and the remaining 25,000 trees (representing 2.5 × 106 generations) were used to calculate a posterior probability tree topology.
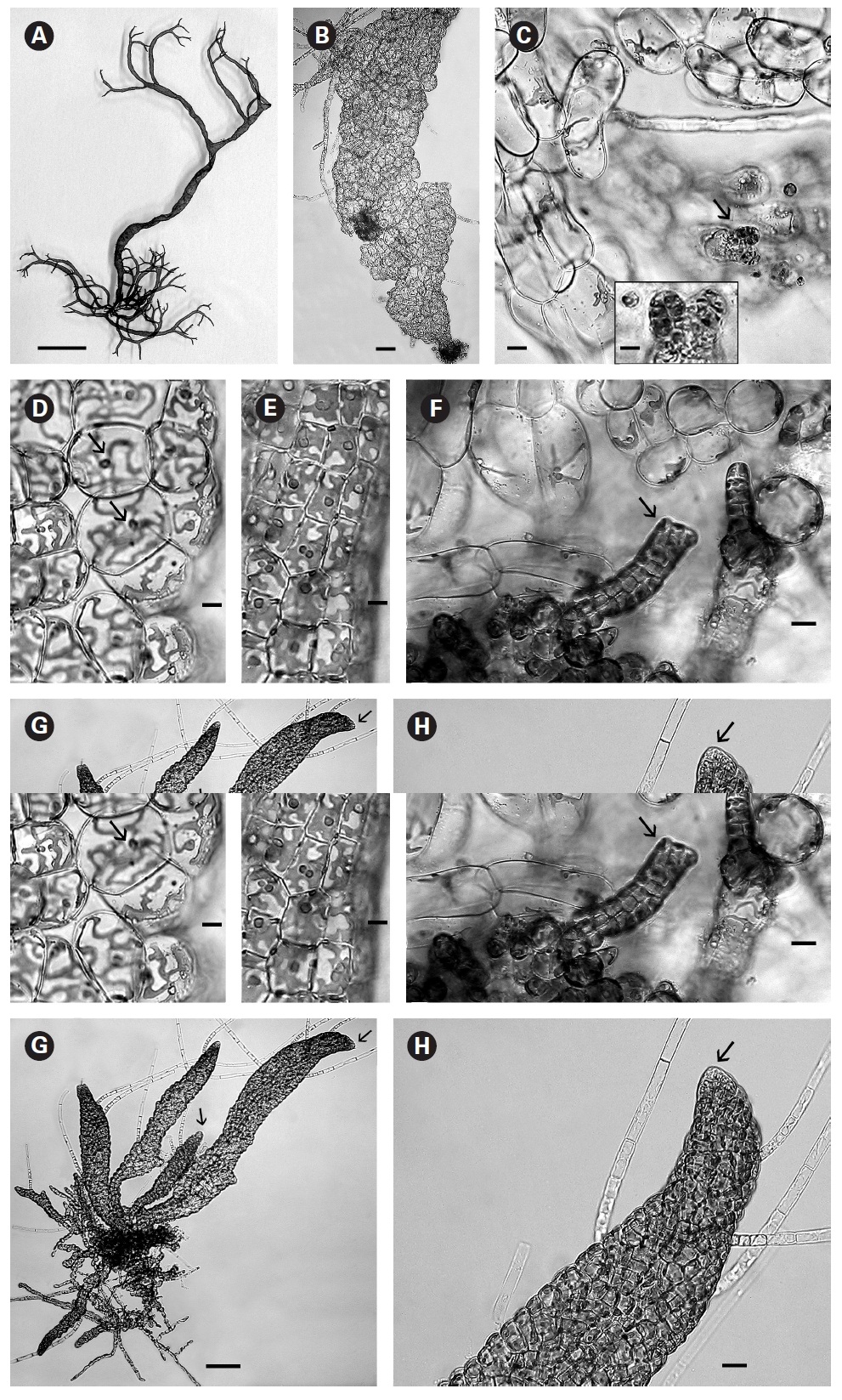
In culture fully developed fronds reached 6 cm in length and a width up to 2 mm with 4-5 orders of dichotomous branching occurring primarily in the upper half of the developing shoots. Some adventitious secondary branching developed at irregular intervals along the lower shoot. From the small basal mat many secondary shoots arose (Fig. 1A). Based on these morphological features it seems closest to
When first observed in rough culture
In brighter light (> 20 ㎛ol photons m-2 s-1) smaller rectangular cells developed with larger chloroplasts in the cortex. In some cells paired pyrenoids possibly indicated the onset of cell division (Fig. 1E). A few small (< 1 ㎛ diameter) colourless bodies, presumed to be physodes, similar to those observed by Feldmann (1949) in
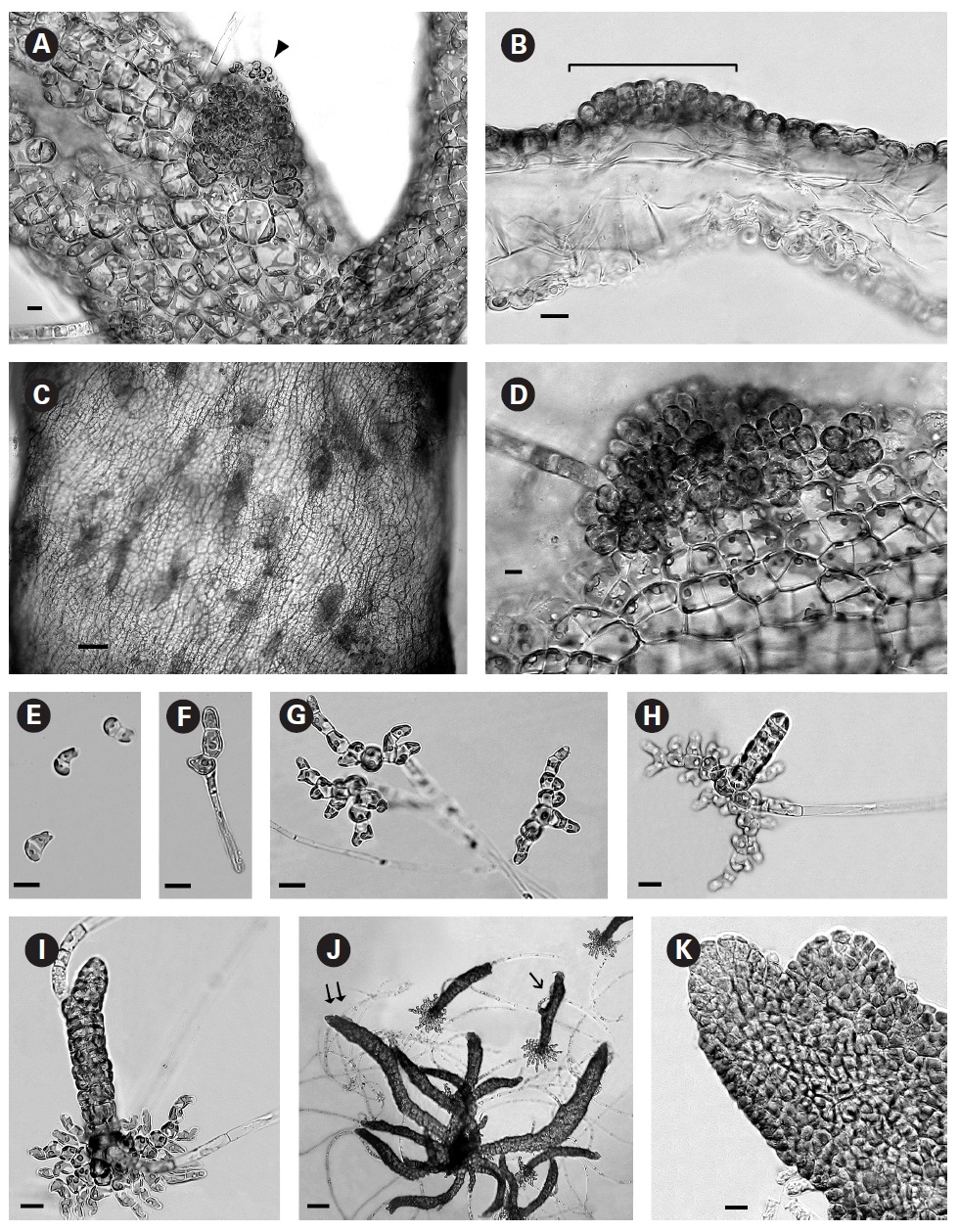
When thalli with loosely coherent filaments were transferred from low light to brighter light on the shaker numerous, compact dark brown shoots developed (Fig. 1F). After 15 days the shoots reached 300 ㎛ long. At 25 days they became about 800 ㎛ long and 100 ㎛ wide with a well-defined medulla and cortex and phaeophycean hairs scattered along the axis (Fig. 1G). The older and longer shoots shifted from a cylindrical form to slightly compressed fronds and all had a hollow center enclosed by the large colourless medullary cells. The apex had a single apical cell and compact cortex with numerous phaeophycean hairs (Fig. 1H). In larger thalli with a compact cortex small, round plurangial sori developed in the mid-regions (Fig. 2A). In cross section, older wider shoots (1.2 mm) were about 120-140 ㎛ thick, with a medulla of large colorless cells and a cortex of a single cell layer (Fig. 2B). Also visible were plurangial sori about 10 cells or more wide (Fig. 2B). Sectioning caused partial collapse of the medulla so the hollow center was not evident. No hyphae were seen in the medulla of longitudinal sections, near the base of older fronds, as was described by Børgesen (1914) for
In older compressed fronds the elongate and mature linear sori followed the slightly oblique pattern of cortical cells along the axis and were generally 100-300 ㎛ long and 60-100 ㎛ wide (Fig. 2C). The plurangial sori were usually two cells deep and in surface view each plurangium appeared to be composed of four cells quadrate to rectangular in shape and somewhat variable in size, 8-15 ㎛ wide and 12-20 ㎛ long (Fig. 2D). Visible within each sorus were several colorless phaeophycean hairs about 5-10 ㎛ wide at the base (Fig. 2D) and up to 1 mm long, before abscising at one or more points along the length.
The zoospores discharged readily and were about 6-8 ㎛ long with two heterokont flagella, a single peripheral chloroplast bearing a pyrenoid and lateral anterior eyespot. In bright light the spores settled on the glass and germinated rapidly. At day 1 (20 hours old) most sporelings were 2 cells long (8-19 ㎛) (Fig. 2E) and did not show evacuation of the original spore that is seen in germination of other Scytosiphonaceae (e.g.,
The shoots of clumped plants appeared to grow faster than solitary ones, measuring 0.5 to 1.2 mm in length. Phototropic growth of the erect shoots of solitary fronds was evident throughout development (Fig. 2J). At some late stage in development shoots appeared to switch from single celled to multi-celled apices (Fig. 2K) but it was difficult to determine when. Cell divisions were primarily in the apical to subapical regions.
Our
phylogenetic placement of our sample does not group with other samples of
Our combined morphological, life cycle and molecular phylogeny studies suggest that the plant collected in Mexico is
Reproductive patterns in various genera of the Scytosiphonaceae are variable in different taxa and different regions.
Only plurangia occurred in our cultured isolate of
Ascocysts were not observed in
The type species
A key (Table 1) was constructed to aid in the identification of the seven
The phylogenetic relationships seen in this study, based on