Thirty populations of Chondrus crispus Stackhouse from Nova Scotia were collected during the years 1993 to 2011.Taken from estuaries, wave exposed open coasts, high intertidal rock pools and shallow subtidal habitats, the populations were evaluated for relative abundance of tetrasporophytic and gametophytic life history phases. Over 2,800 thalli were characterized using the resorcinol-acetal test to distinguish the kappa- and lambda-carrageenan containing fronds of gametophytes and tetrasporophytes, respectively. These populations had 77 ± 5% gametophytes (mean ± 95% confidence interval), with most populations having gametophyte : sporophyte ratios ranging from 2 : 1 to 9 : 1. No population had a dominance of tetrasporophytes, although two populations had 1 : 1 ratios. A meta-analysis of our data along with previously published accounts showed no significant changes in gametophyte dominance with respect to hypothesized gradients of wave exposure, salinity, or water depth. Significant changes occurred in ratios at five sites where replicate sampling occurred in different years. We conclude that C. crispus in Maritime Canada has a natural ratio of 3 : 1 or greater in stable conditions, and that lower ratios represent recovery from disturbance in which bare substratum is created that is subsequently colonized by carpospores from remaining gametophytic thalli.
The life history of
Like other members of the Gigartinaceae,
Here we present an empirical study of life history phase variation over wide temporal and spatial scales in Nova Scotia. Our objective was to determine if environmental factors could be resolved as causal agents in determining the relative abundance of gametophytic and sporophytic thalli. Several previous studies suggested that wave exposure, salinity or water depth could influence relative dominance in the life history phase of
#Dedicated to the memory of Jack L. McLachlan ? friend and mentor
Populations of
As a test of reproducibility of results from a given site / collection, two separate analyses were carried out. In the 62first, two collections of 100 fronds were processed from an aquaculture facility (Scotian Halibut Ltd., Woods Harbour, shelburne Co.) based on fronds originally isolated from an apparently asexual population in Basin Head, Prince Edward Island (Sharp et al. 2010), and grown in mass culture for two years. These fronds have only ever produced cystocarps and the two analyses separated by eight months showed 99 and 100% gametophytes. The single frond segment not conclusively a gametophyte gave an ambiguous result and was scored accordingly. In a second experiment, two independent lots of 100 fronds were processed from a larger collection of fronds from Tor Bay collected in October 1996. These samples gave 83% and 89% gametophytes, values not significantly different from each other (
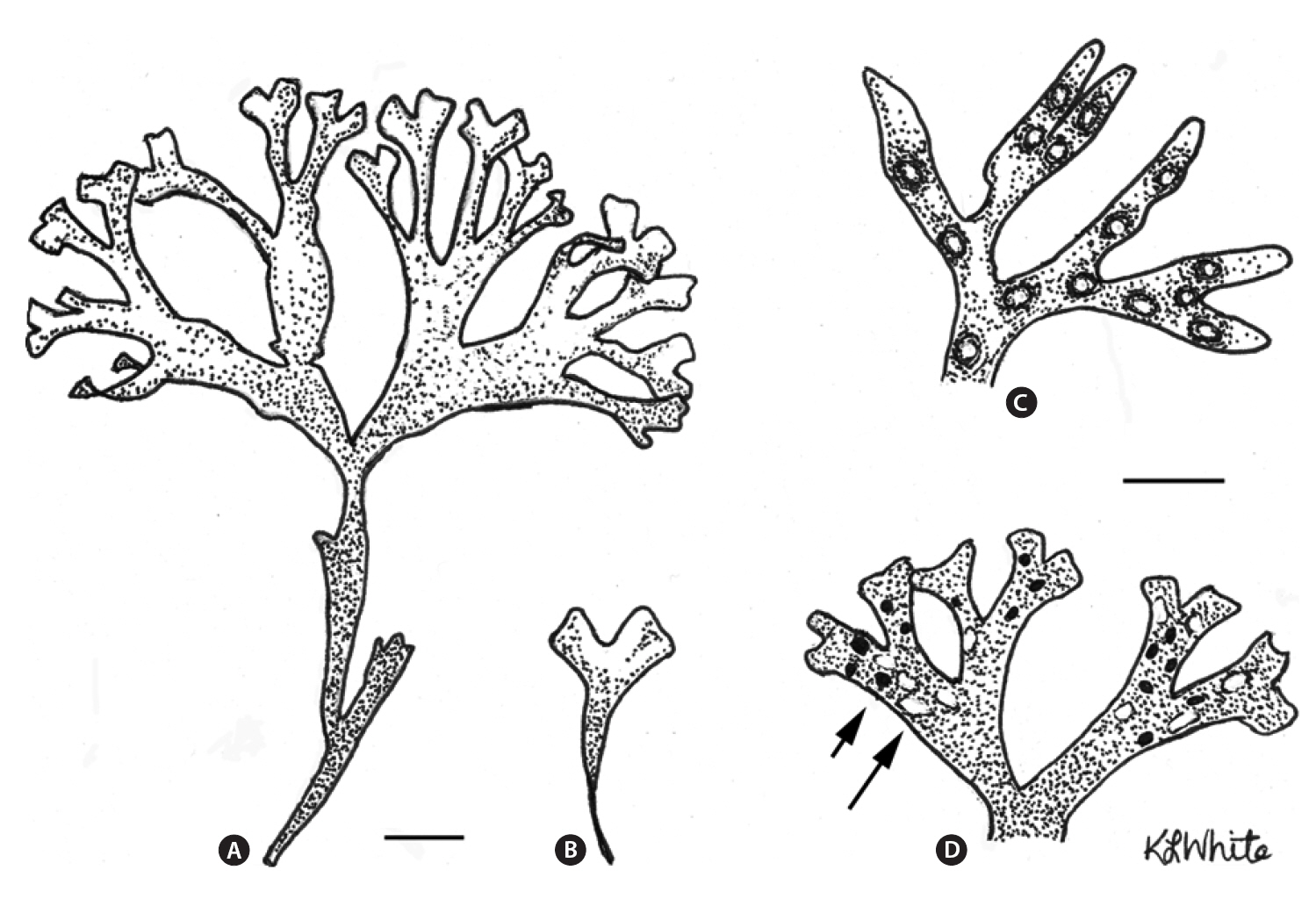
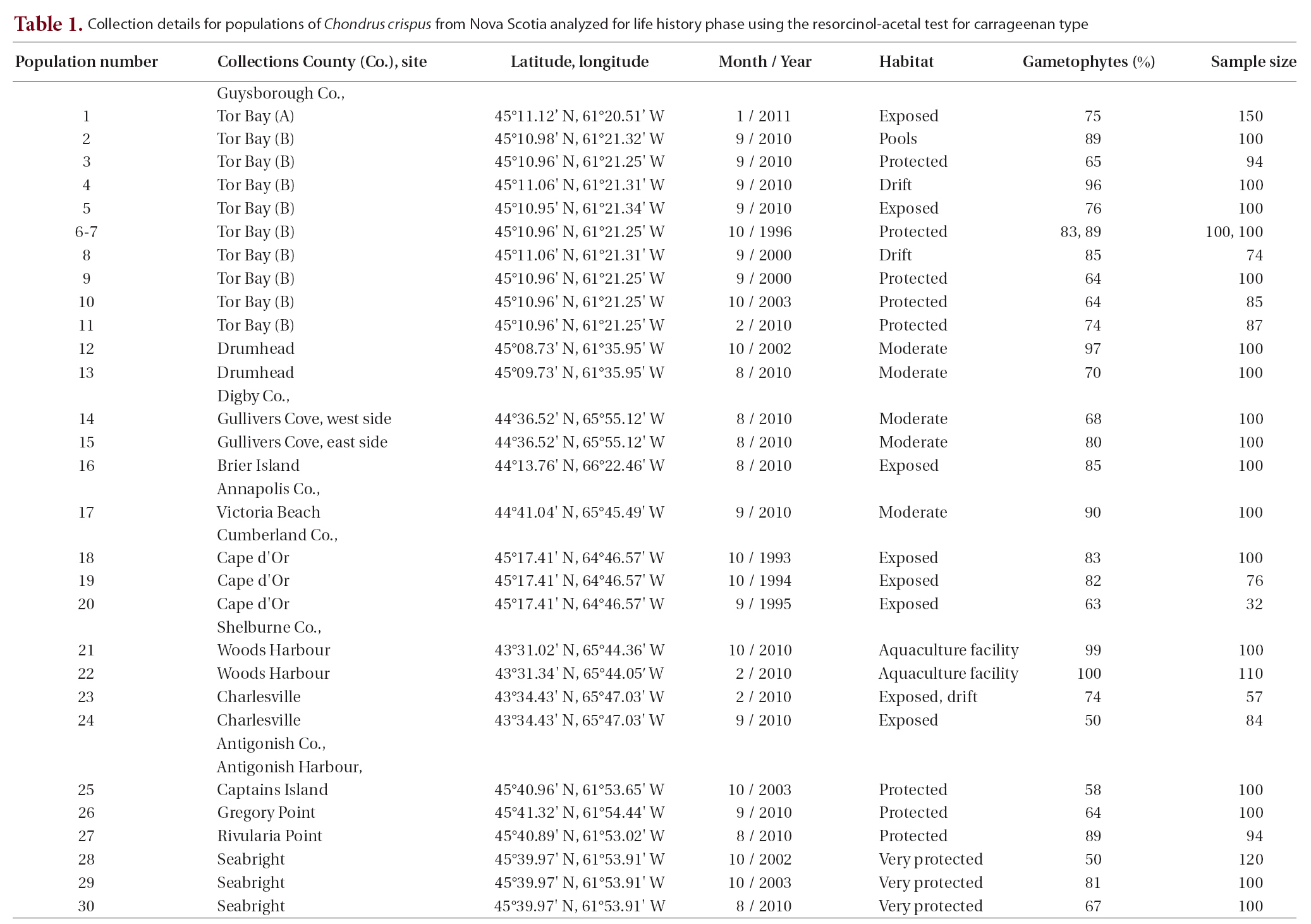
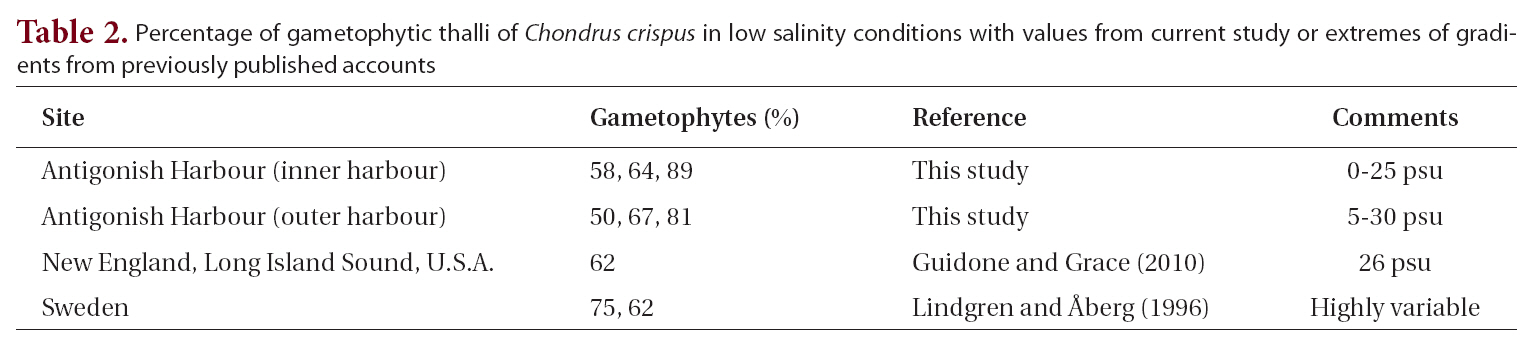
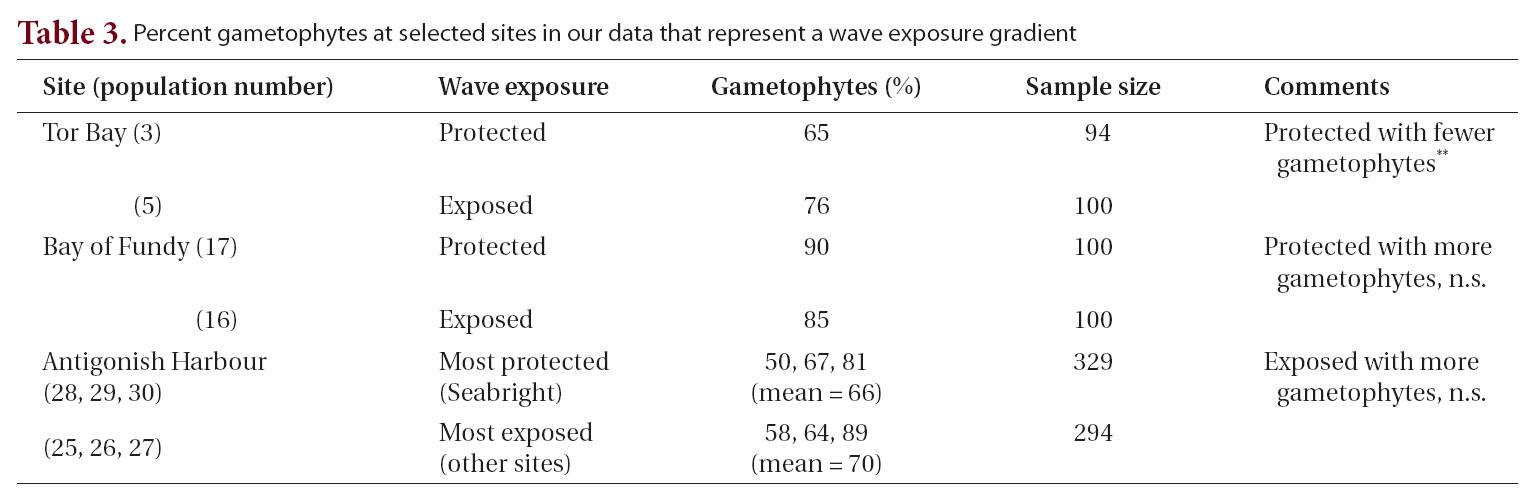
Life history phases of fronds of
The mean percent gametophytes of the 30 populations processed was determined along with the 95% confidence interval. A series of single sample t-tests (Zar 1999) showed that this value (76.4 ± 5.2, mean ± 95% confidence interval) was significantly greater than 70 (p < 0.05). Accordingly, we ignored the question of which life history phase was dominant in a given population, as it was patently the case that gametophytes dominated these Nova Scotia populations. The evaluation of our data in combination with previously published data was to look for patterns of differential gametophyte dominance that could be related to particular environmental gradients, i.e., wave exposure, salinity and water depth. Furthermore, if environmental factors were the primary cause of different life history ratios, then we would predict that the ratios of gametophytic to sporophytic thalli would remain relatively constant at a given site. This was done at five sites, with four of these consisting of collec-
tions from three different years and time spans of two to 20 years.
To determine if different environmental variables affected the relative abundance of gametophytes, we selected relevant populations from our data set along with previously published accounts. We then evaluated the extent to which differences in ratios were present along gradients of salinity, wave exposure, and depth. To evaluate hypotheses that gradients of abundance were present, we conducted a series of
The 30 populations from six counties of Nova Scotia collected over a 20-year period from diverse habitats showed extensive variation in the ratio of gametophytic to tetrasporophytic fronds. While the mean was 77% gametophytic fronds (with 95% confidence intervals ± 5%), most samples had 67-89% gametophytic fronds (Table 1). Only three populations were below 60% gametophytes, and none of the collections showed a dominance of tetrasporophytes. Thus most of the samples ranged from a ratio of about 2 : 1 to 9 : 1 (gametophytes : sporophytes). Use of
The lowest value of 50% gametophytic fronds was found twice. One site was a wave exposed shore at Charlesville (September 2010). These fronds were collected near the upper limits of their distribution on the shore from beneath a canopy of
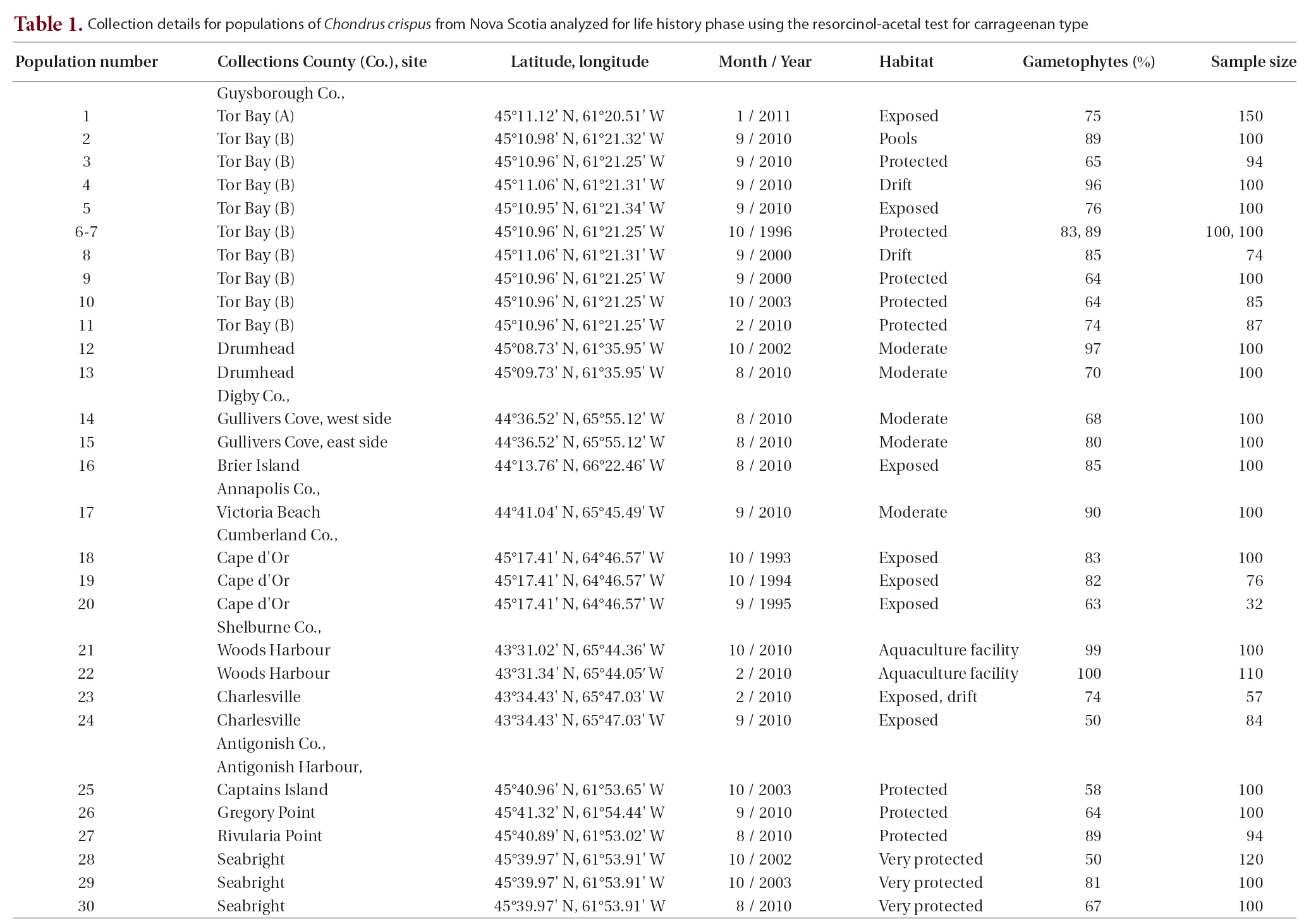
Collection details for populations of Chondrus crispus from Nova Scotia analyzed for life history phase using the resorcinol-acetal
be representative of the general community at Charlesville as evidenced by a collection seven months previous (i.e., February 2010) taken from the drift that gave 74% gametophytes (Table 1). The second collection of 50% gametophytes came from an estuarine site in Antigonish Harbour; this population was significantly lower than two other collections from the same site taken in different years (i.e., 67%, 81%).
Another low value for gametophyte dominance (i.e., 58%) came from near Captains Island, in Antigonish Harbour. This is an estuarine site adjacent to the main channel through the harbour. The
The possibility that estuarine, or low salinity conditions might influence gametophyte:sporophyte ratios was examined by taking all of our samples from Antigonish Harbour where salinities can vary from 0-28 psu and combining these with the values from low salinity samples in the literature (Table 2). These nine values were then compared with the overall mean from our complete data set of 30 samples. The mean of the low salinity sites was 68 ± 12% (mean ± standard deviation). A t-test showed no significant difference between these populations and the overall mean in our entire analysis. A further attempt
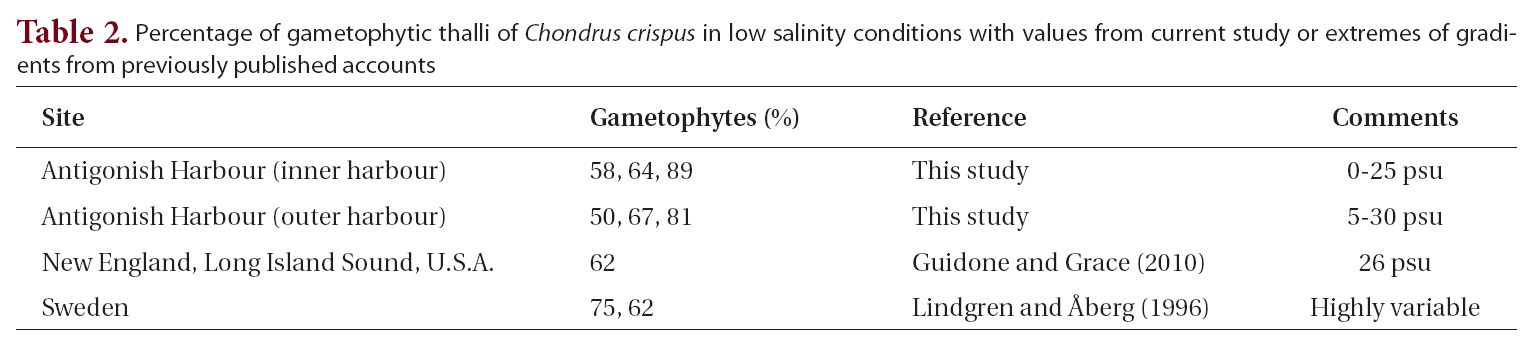
Percentage of gametophytic thalli of Chondrus crispus in low salinity conditions with values from current study or extremes of gradients from previously published accounts
to resolve a salinity gradient within Antigonish Harbour, performed by separating populations from the more saline outer harbour (58%, 64%, 89% gametophytes) from the more brackish inner harbour (50%, 67%, 81% gametophytes), showed no apparent trend with salinity.
The possibility that life history phase was modified by wave exposure was examined in three sets of populations from Tor Bay, the Bay of Fundy and in Antigonish Harbour (Table 3). Within each region the pairs of populations (wave protected vs. wave exposed) were collected in the same season. At Tor Bay where the two populations were selected along the wave exposure gradient of Heaven and Scrosati (2008), the protected site had fewer gametophytes (significant at p < 0.05) whereas the difference in values from the other sites was not significant.
The most intensively sampled location temporally (i.e., 1996-2011), spatially (ca. 1 km) and ecologically (i.e., wave protected to wave exposed, rock pools to low intertidal and drift) was at Tor Bay, a fully marine area on the Atlantic coast. Values ranged from 64 to 89% gametophytes for attached populations, and two drift collections had values of 85% and 96% gametophytes (Table 1). Additional samples from Tor Bay by Scrosati et al. (1994) and Scrosati and Mudge (2004
Other sites (Drumhead, Charlesville) along the Atlantic coast of Nova Scotia showed a similar pattern of strong gametophytic dominance as well as significant differences in the proportions of gametophytes among collections. The two collections from Drumhead (70% and 97% gametophytes) are consistent with other values from the Atlantic coast of Nova Scotia by Bhattacharya (1984) (90-93% gametophytes) and McLachlan et al. (2011) (58-92% gametophytes).
Our collections from fully marine sites in the Bay of Fundy showed variation from 63-90% gametophytes, i.e., ca. 2 : 1 to 9 : 1. Significant differences occurred with small spatial separation (i.e., east and west sides of Gullivers Cove, 80% and 68% gametophytes, respectively) and with temporal sampling over one year (63 to 83% gametophytes).
Gametophytic thalli clearly dominate in Nova Scotian populations of
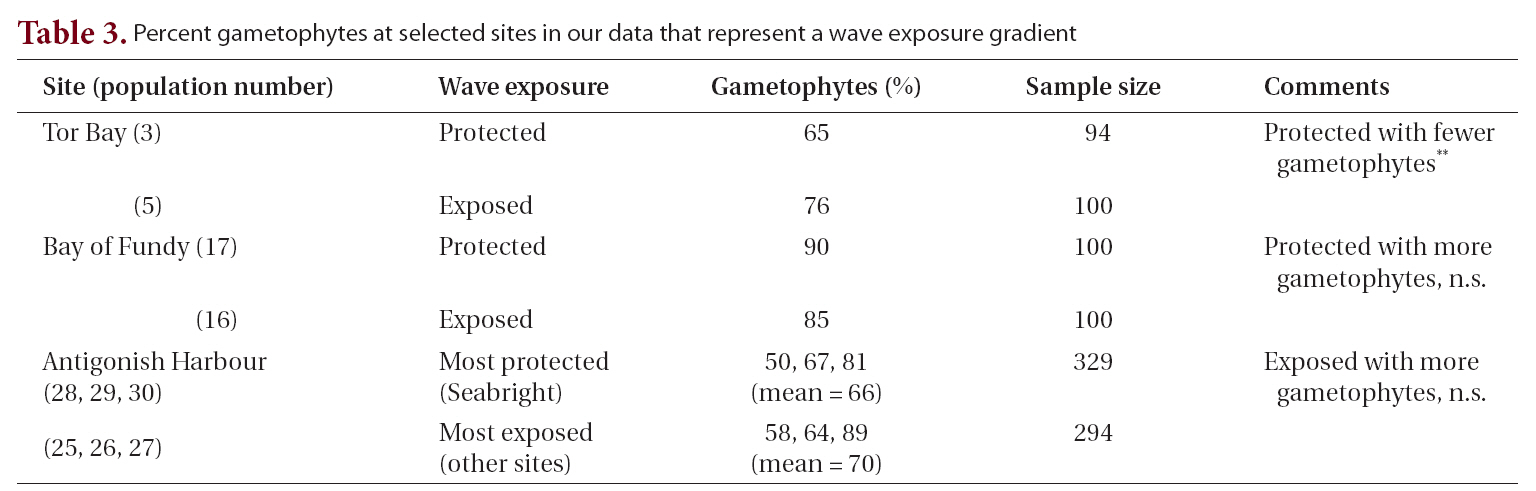
Percent gametophytes at selected sites in our data that represent a wave exposure gradient
son and Prince 1973). The underlying assumption here is that female gametophytes (i.e., carpospore producing) and tetrasporophytes have equivalent fecundities. This assumption is consistent with results of Bhattacharya (1984, 1985) and Ferna ndez and Menendez (1991). Given the similarities in size and shape of cystocarpic and tetrasporophytic fronds, and the apparent absence of biomechanical properties leading to selective frond removal that might be associated with carrageenan differentiation in
Developing a realistic model that explains life history demography of
Populations collected from the drift high in gametophytes may be explained by the tendency of gametophytic plants to degenerate more readily than tetrasporophytes. Bhattacharya (1984) found survivorship of tetrasporophytes to be greater than cystocarpic plants observed simultaneously. A population of
Two other studies of
Wave exposure has been implicated as a regulatory mechanism for abundance of life history phases in
The possibility that there might be a depth gradient for abundance of gametophytic thalli in Irish moss was proposed by Craigie and Pringle (1978). While they found an increase in tetrasporophytes with depth in Nova Scotia and Prince Edward Island, more comprehensive sampling by Lazo et al. (1989) did not uncover this pattern. This pattern was also not apparent in the subtidal sampling of Chopin et al. (1988), who found an increase in tetrasporophytes in a bed of
Given that conspicuous gametophyte dominance is the rule in
Our explanation is consistent with the conclusion of Lazo et al. (1989) and McLachlan et al. (2011) to account for the near equality and occasional predominance of tetrasporophytes in Prince Edward Island. Here new space for colonization is being produced because the sandstone is so friable and whole thalli are being removed along with the underlying substratum. Parker and McLachlan (1990) also showed that large proportions of the
A second mechanism that could be responsible for tetrasporophyte dominance involves the periphery of a population that is undergoing gradual spread. Such spread might be a consequence of environmental change (e.g., climate change or competitive interactions). In such situations the marginal sites would have a greater spore rain of carpospores from the gametophyte dominated population as in Bellgrove and Aoki (2008). This could account for the tetrasporophyte dominated populations at the deeper edges of beds in Prince Edward Island (Craigie and Pringle 1978). It could also explain the 50% gametophytes from the September 2010 population at Charlesville. This population was collected from the upper margin of the
Over time, we suggest that, barring a high frequency of catastrophic events, that populations that were initially tetrasporophyte dominated, would subsequently return to the 2 : 1 (or higher) ratio that characterizes most populations. In light of the inability to relate relative abundance of a life history stage of
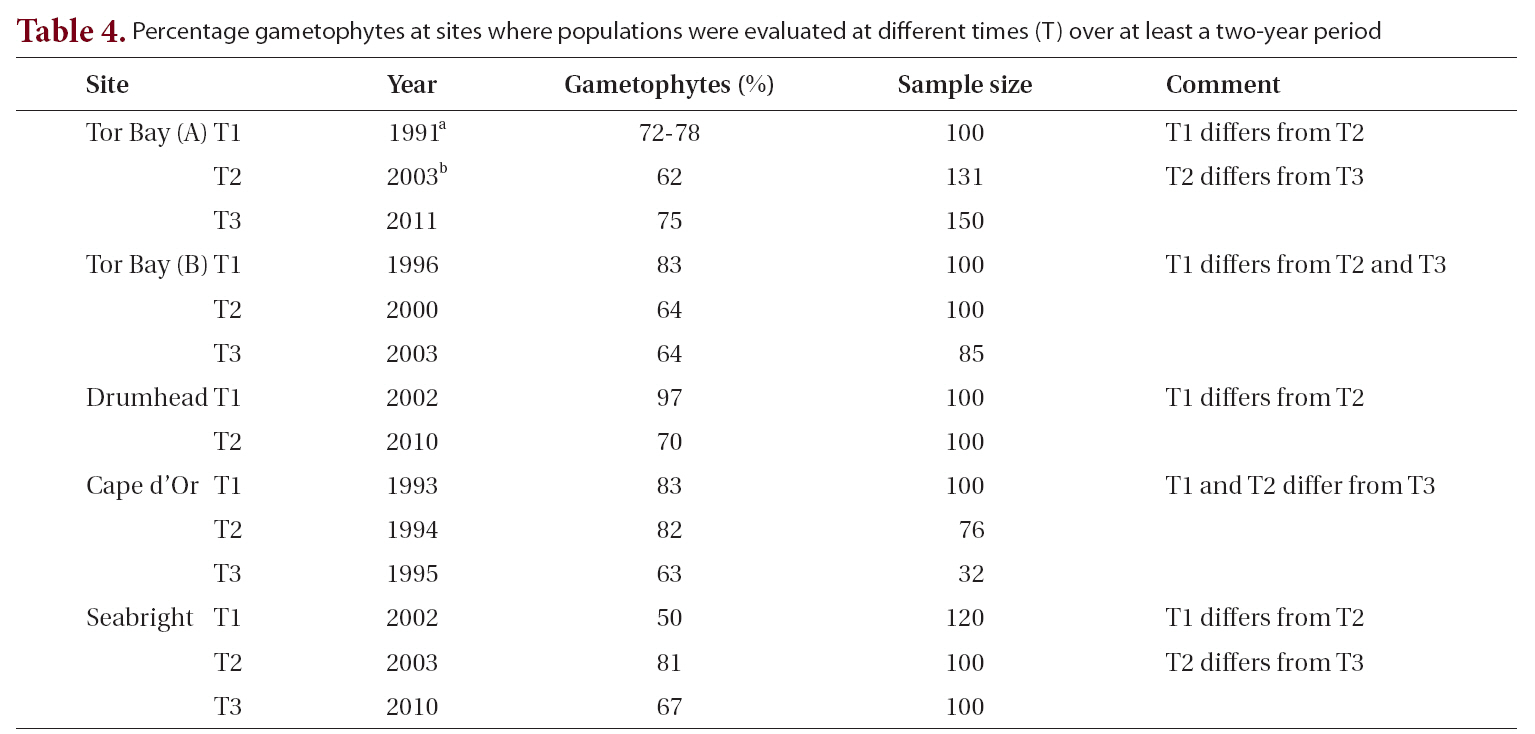
Percentage gametophytes at sites where populations were evaluated at different times (T) over at least a two-year period
gradients to which they are being related. This is consistent with the temporal variation we found in that all five sites sampled in more than one year showed significant differences among years (Table 4). Our study shows that changes in periods as short as one year may lead to significant changes in the proportion of life history phases, and that these changes may be greater in extent than differences associated with environmental gradients.
We suggest a series of environmental and biotic factors that might account for these stochastic changes. First, storm action can severely modify the lower intertidal zone by displacing boulders into or out of the habitat or even just turning them over. Secondly, ice scour from pack ice can effectively remove a large expanse of intertidal algae. With respect to biotic factors, abundant sea urchins can effectively remove the entire flora from the subtidal zone. This will leave intertidal populations of