Although lizards are important hosts for hard ticks (Ixodidae), very few studies have been conducted in South Korea. To determine whether or not hard ticks can infest lizards endemic to South Korea, we examined 77 lizards of four species(Eremias argus, Sincella vandenburghi, Takydromus amurensis, and Takydromus wolteri) that were collected at 22 different sites between April and October 2010. We confirmed that all four lizard species can be infested by Ixodes nipponensis larvae or nymphs. Of the 62E. argus examined, we found an average of 12.5 larvae on two lizards and an average of one nymph on one lizard. We found seven nymphs on one S. vandenburghi. We found an average of two nymphs on two of the five T. amurensis and an average of one nymph on four of the nineT. wolteri. Ixodes nipponensis larvae and nymphs were found most frequently on the foreleg axillae (87.8%), followed by the forelegs (7.3%), the eyelids (2.4%),and the ears (2.4%) of the lizards. To the best of our knowledge, this is the first report of I. nipponensis infestations of lizards endemic to South Korea.
Various ectoparasites have been found on reptiles,including ticks, mites, mosquitoes, flies, and leeches(Pough et al. 2004). Because ectoparasites suck lymph and blood from their hosts, they are capable of spreading viruses and bacteria and causing secondary infestations.The typical negative effects of ectoparasites on their hosts include decreased energy, impaired locomotor skills, diminished social activity, reproductive failure,and an elevated risk of mortality for the hosts themselves(Schall and Sarni 1987, Dunlap and Schall 1995, Oppliger et al. 1996, Sorci et al. 1996).
A well-known group of ectoparasites on snakes and lizards are hard ticks (e.g., Ixodidae) (Durden et al. 2002).Larvae and nymphs of the tick species
The life cycle of hard ticks consists of egg, larva, nymph, and adult stages (Sonenshine 1991, Noda et al. 2004, Lee 2009). Hard ticks depend on various terrestrial vertebrates (i.e., rodents, birds, and reptiles) to act as hosts, so that they may feed on their lymph and blood. They also play a key role spreading fungi, viruses, rickettsiae, bacteria, and protozoa among their hosts (Sonenshine 1991). In South Korea, hard ticks infest domestic and wild animals, particularly rodents, birds, skinks (Arthur 1957, Noh 1965, Kim 1970, Kim and Lee 1989, Shim et al. 1992, 1993, 1994, Lee et al. 1997, Eum et al. 2006, Kim et al. 2009), and humans (Kang et al. 1982, Cho et al. 1991, Yoon et al. 1996, Yun et al. 2001). Only a single study has been conducted that relates the
Consequently, this study determined which types of hard ticks infest which endemic lizards. Additionally, we classified the hard tick species with their associated lizard species.
To collect ectoparasitic ticks from lizards, we first captured lizard specimens by hand from the following regions: Kyeonggi-do (Kwangju, Yeoju, Yangpyeong, and Icheon), Kangwon-do (Chuncheon, Wonju, Yeongwol, Pyeongchang, and Gangneung), Chungcheong-do (Jecheon, Chungju, Danyang, Taean, Boryeong, and Seocheon), and Kyeongsang-do (Cheongsong, Gunwei, Sungju, Kimcheon, Gumi, Goryeong, and Hapcheon). This specimen collection period lasted from April to October 2010. Upon catching a lizard, we determined its species and examined it for the presence of hard ticks. In the event that we found ectoparasitic ticks on a lizard’s body, we recorded their number and the locations where they were attached. Next, we measured the lizard’s snout-vent length (in increments of 0.1 cm) and its body weight (in increments of 0.1 g) using a digital vernier caliper (CD-15CPX; Mitutoyo, Kawasaki, Japan) and a digital balance (TMB 120-1; Kern, Balingen, Germany), respectively. Finally, we released the lizards at the site where they were caught. Additionally, we also obtained a sample of Korean skinks (
>
Observation and classification of ticks
Before conducting tick observations, we cleaned the ticks by submerging them in a 10% KOH solution for 1-3 hours. Afterwards, we observed the ticks with either one or both of the following light microscopes: a dissecting microscope (#5424 Stereo Crystal-Pro; Konus, Verona, Italy), or an Eclipse 50i (Nikon, Tokyo, Japan). To investigate the level of detail necessary to construct a classification key for the ticks, we used a scanning electron microscope (SEM). Before this set of observations, the ticks were treated following the method of Park et al. (2008). The ticks were fixed using a 4% glutaraldehyde solution (in 0.2 M phosphate buffer, pH 7.4), washed three times using the same buffer, then serially dehydrated in 50, 60, 70, 80, 90, and 100% ethanol for 30 min at each concentration. Next, the medium was exchanged three times (for 30 min each time) with isoamyl acetate. The ticks were dried using critical-point drying, mounted on a specimen stub, coated with Au-Pd (E-1010; Hitachi, Tokyo, Japan), and observed under a S-3500N low-vacuum SEM (Hitachi) at the Korea Basic Science Institute, Chuncheon, Kangwon. By employing the SEM in our observations, we observed a total of 12 larvae on the Mongolian racerunner (
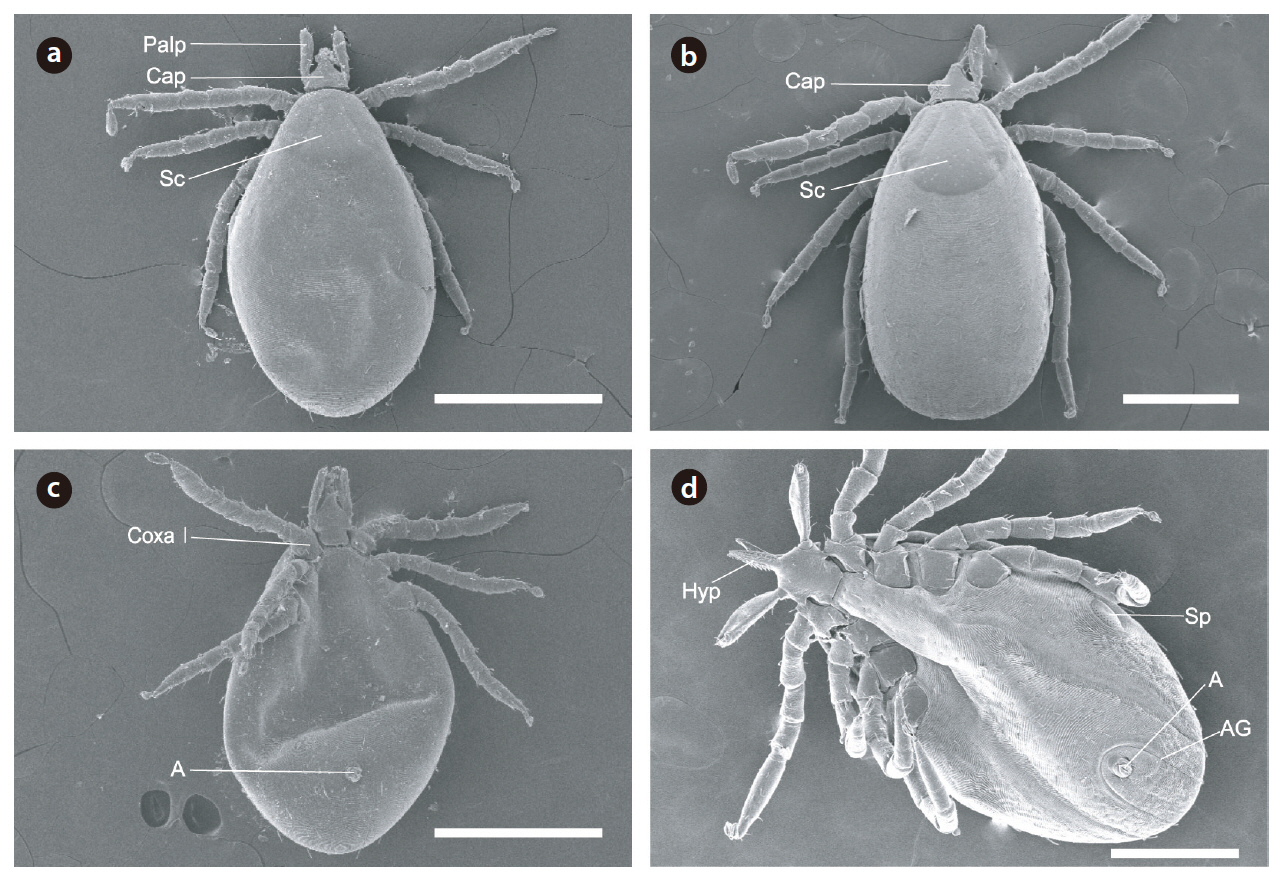
We collected 77 lizards representing four species (
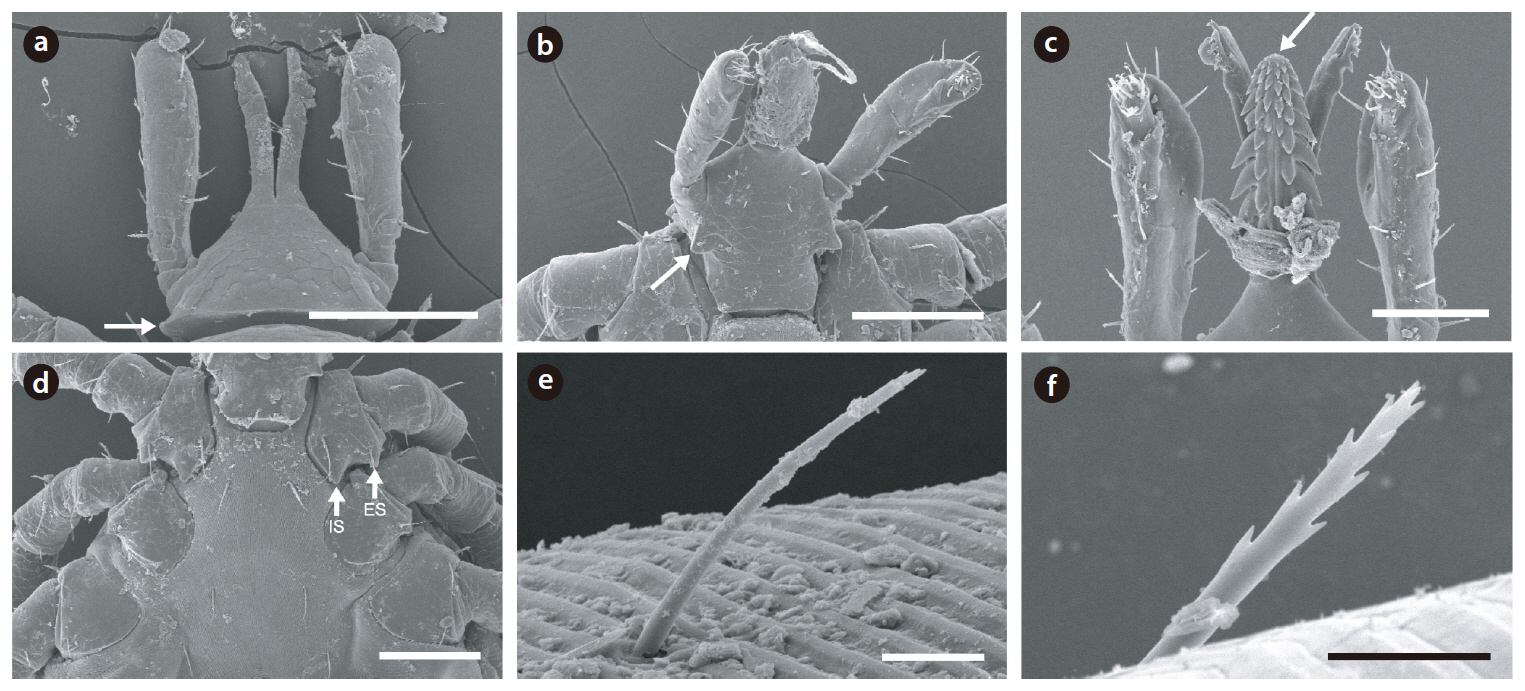
Because the hard ticks had a serrated hypostome on the capitulum at the anterior end of the body, as well as a scutum and an anal groove that rounded the anus anteriorly and was without a festoon, we classified these hard ticks as

Japanese hard tick (Ixodes nipponensis) larvae and nymphs found on lizard species endemic to South Korea
The end of the larvae hypostome was blunt, whereas that of nymphs was sharp. On the hypostome, both larvae and nymphs had 3/3 upper tooth rows and 2/2 lower tooth rows (Figs. 3c and 4c). Both larvae and nymphs had relatively long external and internal spurs on the first coxal plate, whereas the external spurs on the remaining coxae were not extended to the next coxal plate (Figs. 3d and 4d). The postscutal setae were forked, and the forked setae were often observed on other parts of the body such as legs Figs. 3e, 3f, 4e, and 4f). Based on these classification characteristics, the hard ticks collected were all identified as
The 16 ticks collected from Chuncheon, Yeongwol, and Sinan between April and May were all nymphs, whereas the 25 ticks collected in July from
This study identified Japanese hard tick (i.e.,
The infestation rates by larval and nymphal
of plants (Apperson et al. 1993, Durden et al. 2002). Such studies have also indicated that there is a higher chance of infestation by hard ticks when potential hosts are on the ground. Unfortunately, the sample size of each lizard species examined in this study varied widely; consequently, additional studies with larger, more comparable lizard samples will be necessary to compare the Ixodes tick infestation rate in different lizard species.
Consistent with previous findings, most of the ticks found in this study were located on the lizards’ foreleg axillae. Hard ticks are known to be found principally on areas where the skin is weak and well-lined, although they can also be occasionally collected from other parts of the body (Durden et al. 2002). Among the skinks (genus
The larval and nymphal
Although immature larval and nymphal
ticks. As ectoparasites, ticks negatively affect a host’s health and reproduction (Sonenshine 1991, Main and Bull 2000). It seems reasonable to assume that Korean lizards would be negatively impacted by ticks in the field as well. Finally, it is important to pay close attention to such ectoparasites on lizards in captivity, for purposes related to keeping of pets and to conservation.