This study traces the origin, evolution, and current state-of-the-art of engineering-oriented water-quality management and modeling.Three attributes of polluted water underlie human concerns for water quality: rubbish (aesthetic impairment), stink (ecosystem impairment), and death (public health impairment). The historical roots of both modern environmental engineering and water-quality modeling are traced to the late nineteenth and early twentieth centuries when European and American engineers worked to control and manage urban wastewater. The subsequent evolution of water-quality modeling can be divided into four stages related to dissolved oxygen (1925?1960), computerization (1960?1970), eutrophication (1970?1977) and toxic substances (1977?1990). Current efforts to integrate these stages into unified holistic frameworks are described. The role of water-quality management and modeling for developing economies is outlined.
1. Introduction: Why We Care About Water Quality
The scientist and author, Jared Diamond, articulated how geography influences human societal development in the title of his Pulitzer-winning book
Of these three terms, “death” is the most obvious. Humans clearly care whether water is safe to consume without the risk of immediate or long-term harm. But beyond such public-health concerns, the two other less dramatic characteristics, rubbish and stink, also contribute significantly to how humans value water.
The term “rubbish” reflects the aesthetic importance of water in that humans tend to devalue water when it is visually unappealing.This aversion may have originally been connected with health concerns in that colored, murky or particle-laden water may be unpotable and/or unpalatable. Although this aversion may be health-related, there is no question that humans place higher value on pure, clear water.
At first hearing, the term “stink” suggests another aesthetic quality, as humans have an obvious distaste for malodorous water.However, in the present context, the term is meant to represent ecosystem health. To explain, the “health” of most aquatic ecosystems experienced directly by humans (i.e., while at the beach, fishing, or boating) is often reflected by dissolved oxygen level. When excessive quantities of sewage and other sources of organic matter are discharged into such systems, dissolved oxygen concentrations can drop to zero. This, of course, is an ecological disaster for organisms such as fish that depend on dissolved oxygen for their survival. However, along with such direct biological impacts, anoxic waters also lead to significant chemical changes. In particular, sulfate is converted to hydrogen sulfide gas creating an intense “rotten egg” smell. Thus, the water’s bad odor provides humans with an olfactory signal that the aquatic ecosystem is “sick.”
While this triad of symptoms informs humans that water quality is threatened, when was this awareness translated into remedial action. That is, when did society collectively marshal its resources to solve the water-quality problem? To answer this question, we must turn to nineteenth century England.
2. The Birth of Environmental Engineering
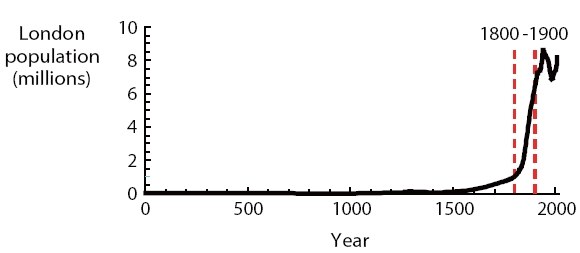
Although some older human cultures which developed infrastructure to provide clean drinking water and sanitation,the roots of modern environmental engineering can be traced to London in the mid-nineteenth century. Due primarily to the economic and demographic impacts of the industrial revolution coupled with the great wealth generated by the British Empire,the population of London grew almost an order of magnitude during the nineteenth century (Fig.1 ). As a result, it was the
world’s largest city from 1830 to 1925.
As is currently the case in many parts of the world, a great deal of this population growth consisted of poor people migrating from the countryside to the city in search of employment.These settlers tended to congregate in densely-populated slums with inadequate water and sanitation infrastructure. Much of their drinking water was drawn from the groundwater via shallow wells, and their sewage was disposed into cesspits or to storm sewers that ultimately fed into the adjacent Thames River.
In 1858, the problem reached an environmental tipping point, as the large quantities of sewage combined with a particularly hot summer created a perfect storm of rubbish, stink, and death. As depicted in Fig. 2,“rubbish,” in the form of dead animals and debris, floated in the Thames. That year was dubbed“The Great Stink of 1858” due to the terrible stench emanating from the river. Doctor William Budd, a pioneer in the study of infectious diseases, stated: “Never before, at least, had a stink risen to the height of an historic event.” The smell was so overwhelming that it threatened the functioning of the House of Commons and the law courts. As public awareness and political pressure grew, a government committee was appointed to develop a solution to the problem.
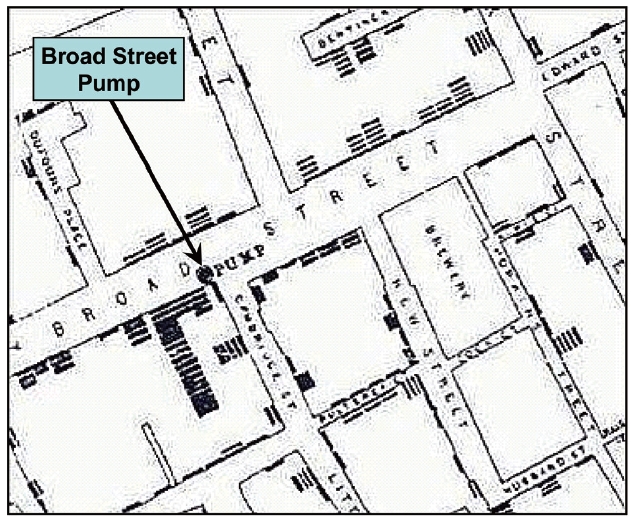
But what about death? During the same period, cholera became widespread in London. At first, the science of the day erroneously posited that the disease was caused by an air-borne“miasma.” In 1854, the London physician John Snow developed a counter-hypothesis that the cholera epidemics were due to contaminated drinking water. A primary line of evidence supporting this hypothesis was a map he developed on which he plotted the deaths for a particular outbreak (Fig.3 ). A precursor of GIS, the so-called “Ghost Map” [2] provided strong circumstantial evidence that the disease was waterborne.
Unfortunately, Snow’s hypothesis was not widely accepted until Robert Koch isolated the cholera organism in the 1880s.Nevertheless, the political awareness and pressure created by the Big Stink was sufficient to stimulate a number of remedial measures, including a major expansion of the London sewer system. Designed by chief engineer Joseph Bazalgette, the sewer expansion was designed to carry most of London’s wastewater to a discharge point well downstream of the city. Although the principle motivation for the new sewer system was to alleviate the stench, ancillary benefits were to improve appearance of the Thames and to greatly diminish the cholera outbreaks.
This huge and expensive sewer project can be considered the birth of modern environmental engineering. That is, it was the first major modern example of the application of the engineering approach to solve an environmental problem. From this point forward, other cities around the world began to consider how infrastructure development could be used to alleviate the rubbish, stink, and death created by urban water pollution.
3. The Roots of Water-Quality Modeling
Although the London sewer project marked the beginning of environmental engineering, it involved the same skill set as classical civil engineering. That is, it used structural engineering and fluid mechanics to design and construct the infrastructure needed to reduce and redirect pollution (i.e., sewers, tanks,treatment systems, etc.). So now we must ask the question: What drew engineers out of the sewers and treatment plants and into the natural environment?
To answer this question, we must again return to London. As stated previously, the initial objective of the London sewer expansion project was to transport the wastewater downstream away from the city center. At first, this approach worked well, as bacterial action and tidal flushing were sufficient to adequately breakdown and disperse the sewage. This ability to absorb pollutants is formally referred to as the receiving water’s “assimilative capacity.” However, as the population continued to grow and expand downstream, the quantity of wastewater eventually overwhelmed the system’s assimilative capacity and waterquality problems reemerged. At this point, it was concluded that some form of wastewater treatment was needed to maintain adequate water quality.
By the early twentieth century, this problem was occurring in many other urban environments with the result that engineers throughout the world began to design urban water and wastewater systems (Fig.4 ). At first, civil engineers became involved in the development of water-treatment plants, water distribution networks, and wastewater collection systems. The design of these projects was fairly straightforward, because the goals were so well defined. The goal was to deliver an adequate quantity of potable water to the urban populace and to safely carry off their wastes.
In contrast, the question of what to actually do with the wastewater was a more ambiguous proposition. At first, municipalities typically discharged their untreated sewage directly to receiving waters. As occurred in London, such action often transformed the receiving waters into large sewers. Hence,wastewater treatment plants began to be constructed. However,it was soon recognized that treatment could range from simple inexpensive sedimentation to costly physical/chemical/biological treatment. In the extreme case, the latter might theoretically result in an effluent that was more pristine than the receiving water. Clearly, both extremes were unacceptable, and some design goal had to be established that would protect the environment adequately but economically.
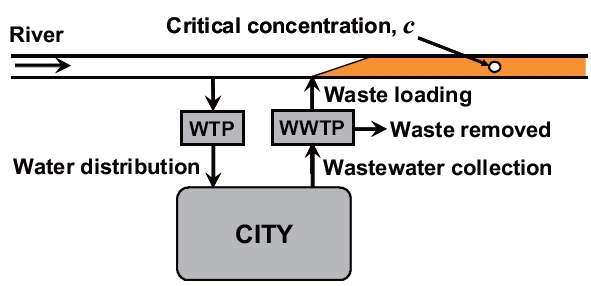
Thus, it was decided that waste treatment should be designed to produce an effluent that induced an acceptable level of water quality in the receiving water. However, to determine this proper level of treatment, it was necessary to establish a linkage between the waste loading and the resulting critical concentration in the water body. Consequently, civil engineers began to develop mathematical water-quality models for that purpose to compute quality (the response) in the receiving water (the system) as a function of treatment plant effluent (the stimulus).Such models can be represented generally as:
According to this equation, the cause-effect relationship between loading and concentration depends on the physical,chemical, and biological characteristics of the receiving water.In essence, the mathematical model offers a quantitative framework to integrate the diverse physical, chemical, and biological information that constitutes complex environmental systems.Beyond solving a particular pollution problem, models provide a vehicle for an enhanced understanding of how the environment works as a unit. Consequently, they can be of great value in both research and management contexts.
The first actual water-quality model was developed by Harold W. Streeter and Earl B. Phelps in 1925 for the Ohio River in the Midwestern United States [3]. The model consisted of a simple analytical equation that could be used to compute the critical dissolved oxygen concentration downstream from an organic wastewater point source. Along with loading, it included terms quantifying the river’s physics, chemistry, and biology (i.e., water velocity, organic decomposition rate, and reaeration rate).Hence, it provided environmental engineers with the quantitative linkage between wastewater loading and receiving-water concentrations. As described next, such modeling frameworks evolved over the following years to address a variety of waterquality problems.
4. The Historical Evolution of Water-Quality Modeling
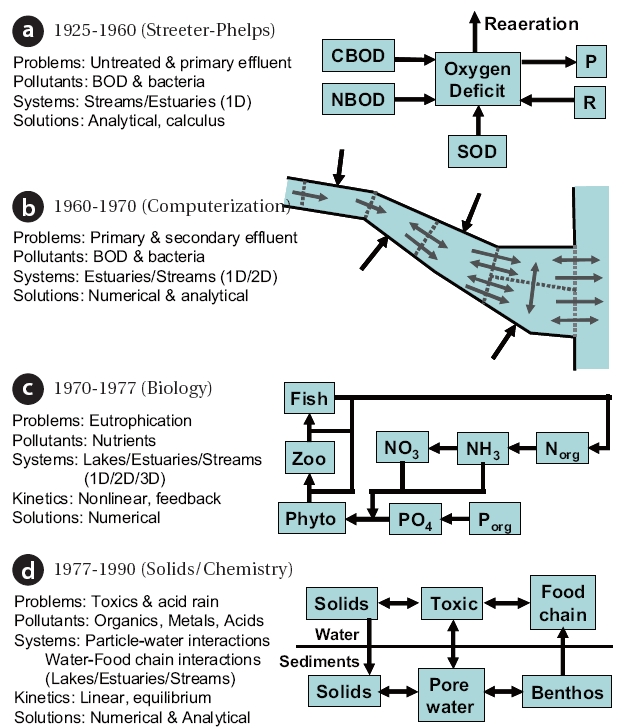
Water-quality modeling has evolved a great deal since its innovation in the early years of the twentieth century. As depicted in Fig. 5,this evolution can be divided into four major phases.These phases relate both to societal concerns and the computational capabilities that were available during each of the periods.
As described in the previous section, most of the early modeling work focused on the urban wasteload allocation problem(Fig.4 ). After Streeter and Phelps [3] seminal work, other investigators[4-7] refined and extended their basic approach to evaluate dissolved oxygen levels in streams and estuaries (Fig.5a). Additionally, bacteria models were also developed [8, 9].Because of the non-availability of computers, model solutions were analytical or “closed form.” This meant that applications were usually limited to linear kinetics, simple geometries, and steady-state receiving waters. Thus, the scope of the problems that could be addressed was constrained by the available computational tools.
In the 1960s, digital computers began to become widely available. This led to major advances in both the models and the ways in which they could be applied (Fig.5 b). The first modeling advances involved numerical expressions of the analytical frameworks [10]. Oxygen was still the focus, but the computer allowed analysts to address more complicated system geometries,kinetics, and time-variable simulations. In particular, the models were extended to two-dimensional systems such as wide estuaries and bays.
The 1960s also brought changes in the ways in which the
models were applied. In particular, the computer allowed a more comprehensive approach to water-quality problems.Rather than focusing on local effects of single point sources, the receiving water could be viewed as a complex system with multiple pollution sources. Tools developed originally in the field of operations research were coupled with the models to generate cost-effective treatment alternatives [11-14]. Although the focus was still on point sources, the computer allowed a more holistic perspective to be adopted.
Another shift occurred in the 1970's (Fig.5 c). Societal awareness moved beyond dissolved oxygen and urban point sources to a more general concern for the environment. An ecological movement was born and, in some quarters, environmental remediation became an end in itself.
The principal water-quality problem addressed during this period was eutrophication; that is, the creation of excessive aquatic plant growth due to the discharge of nutrients. As a consequence,modelers broadened their own scope to include more mechanistic representations of biological processes. Capitalizing on oceanographic research [15, 16], environmental engineers developed elaborate nutrient/food-chain models [17-21].Because of the existing computational capabilities, feedback and non-linear kinetics could be employed in these frameworks.
It should be noted that during this period, major work proceeded in bringing the urban point-source problem under control.In fact, most municipalities in the United States installed secondary treatment for their effluents. Aside from ameliorating the dissolved oxygen problem in many locales, for areas where point-source control was insufficient, this had the ancillary effect of shifting attention towards non-point sources of oxygen-demanding wastes. Because such sources are also prime contributors of nutrients, the emphasis on eutrophication rein-forced concern over non-point inputs.
At face value, the environmental awareness of the early 1970s should have led to an increased reliance on the systems approach to water-quality management. Unfortunately, this was not the case for three primary reasons. First, because eutrophication considers seasonal plant growth, it is a more dynamic problem than urban point-source control. Although systems analysis methods could be devised to optimize such dynamic problems,they are much more complicated and computationally intensive than for the linear, steady-state, point-source problem. Second,the environmental movement fostered an atmosphere of urgency regarding clean-up. A mentality of remediation "at any cost"led to concepts such as "zero discharge" being articulated as a national goal. Third, the economy was booming during this period. Therefore, the economic feasibility of such a strategy was not seriously questioned. As a consequence, the idea of balancing costs and benefits to devise an economical solution waned.Legalities supplanted sound engineering as the basis for most pollution control strategies. Although progress was made during this period, the unrealistic goals were never achieved.
The next stage of model development evolved in the wake of the energy crisis of the mid-1970s (Fig.5 d). Together with increased deficit spending, the energy crisis brought the pollution control effort back to economic reality. Unfortunately, the initial response amounted to an overreaction to the excesses of the early 1970s. Now, rather than an "at any cost" strategy, the public and their representatives had to be "sold" on the efficacy of environmental remediation. Consequently, attention turned to problems such as toxic substances (and to a lesser extent, acid rain) which, although they certainly represented a major threat to human and ecosystem health, could also be marketed effectively in the political arena.
The major modeling advances in this period have been to recognize the prominent role of solid matter in the transport and fate of toxicants [22-25]. In particular, the association between toxicants with settling and resuspending particles represents a major mechanism controlling their transport and fate in natural waters. Furthermore, small organic particles, such as phytoplankton and detritus, can be ingested and passed along to higher organisms [26]. Such food-chain interactions have led modelers to view nature's organic carbon cycle as more than an end in itself. Rather, the food chain is viewed as a conveyer and concentrator of contaminants.
Another advance during this period was stimulated by the problem of acid rain. Just as eutrophication had led to the inclusion of better biology, acid rain led to an upgrade in the characterization of chemistry in water-quality models. In particular,more detailed and mechanistic frameworks were developed to simulate chemical speciation in natural waters [27-29]. Such schemes were valuable in their own right but have also been integrated into broader water-quality modeling frameworks [30,31].
In summary, the evolution of water-quality modeling over the last century has resulted in a variety of modeling constructs that encompass both conventional and toxic pollutants. During the course of addressing specific water-quality problems (such as dissolved oxygen depletion, eutrophication, and toxic contamination),the models have incorporated a comprehensive array of physical, chemical, and biological processes. As described next, this suite of diverse mechanisms can now be integrated to provide a more comprehensive and holistic model representation of multi-faceted water-quality problems.
Over the past 20 years, a number of factors have fostered more holistic water-quality modeling frameworks. Most prominent among these are legislation, scientific developments, and computing advances.
In the United States, a section of the Clean Water Act requires states to list impaired waters for which technology-based regulations and controls are not sufficient to meet water-quality standards. Among other things, the law requires that total maximum daily loads (TMDLs) be developed for these waters. This is a computation of the maximum pollutant load that can be discharged into receiving water to safely meet standards. Because water-quality models are expressly designed to perform such computations, this legislation has stimulated the application of water-quality models addressing a variety of pollutants in a wide range of waterbodies [32]. Although TMDLs typically address a single pollutant, it is also recognized that most water-quality problems are multi-faceted. For example, a stream might be listed because of eutrophication and hence, at face value, require a nutrient TMDL. However, besides excessive plant growth, nutrient overenrichment can have profound effects on a stream’s oxygen and pH levels. Furthermore, other pollutants such as heat and suspended sediments can have a significant impact on how nutrients ultimately affect the biota. Consequently, more comprehensive modeling frameworks that account for multiple pollutants are being developed to more accurately determine TMDLs [33-35].
Significant research advances have occurred in the recent past. In particular, mechanistic characterizations of sedimentwater interactions and hydrodynamics have advanced so that they can be effectively integrated into water-quality modeling frameworks. Aside from the scientific advances involved in developing these mechanisms, their subsequent integration into usable frameworks is made possible by advances in computer technology.
5.3. Computing Advances
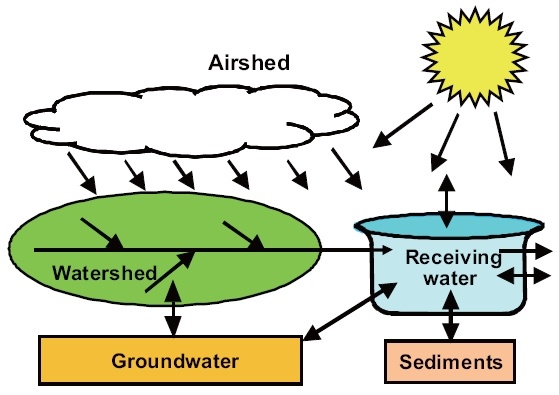
Computer hardware and software have undergone a revolution over the past two decades, which rivals the initial advances made during the 1960s. In particular, graphical user interfaces and decision-support systems are being developed that facilitate the generation and visualization of model output. Further,hardware advances are removing computational constraints that limited the scope of earlier models. Today, two- and threedimensional models with highly mechanistic and nonlinear kinetics can be simulated at a reasonable cost. Additionally, the scope of water-quality modeling has expanded beyond receiving water to encompass bottom sediments, groundwater, the watershed, and in some cases, even the overlying airshed (Fig.6 ).
In summary, the evolution of environmental engineering and water-quality modeling over the last century has resulted in a unified theoretical framework that encompasses both conventional and toxic pollutants. In addition, computer technology and scientific understanding has advanced to the point that
comprehensive systems software can be developed to provide advanced decision support for water-quality managers.
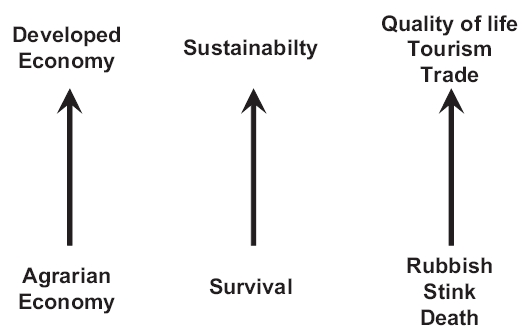
In this article, I have outlined how environmental engineering and water-quality modeling evolved historically in the U.S.and Western Europe. As I hope is clear, less than a century ago,those parts of our planet suffered from comparable levels of“rubbish, stink, and death” suffered by many developing countries today. At first, economic development exacerbated and focused these problems as populations moved from the country to the city. Then, the subsequent growth of stable middle classes coupled with democratic political systems led to societal actions to ameliorate these symptoms.
Fig. 7 provides a depiction of this evolution. As societies move from agrarian to developed economies, the emphasis on water-quality control moves from survival to sustainability. While past concerns for water-quality were driven by “rubbish, stink, and death,” future efforts should be increasingly dictated by higherorder goals such as quality of life, tourism, and trade. Today, developing countries around the world are beginning to recognize that environmental protection must be coupled with economic development. For these countries, cost-effective, model-based control strategies could provide a means to control water pollution while maintaining economic growth. In this sense, the tools and approaches developed over the past century by environmental engineers could greatly contribute to environmentally sustainable development across the planet.