Characteristics of benthic microalgae and sediment properties were investigated for the intertidal flats of Kwangyang Bay, Korea. Sampling stations were selected every 100 m in the intertidal flats from land-side to open ocean at two different sampling sites. Samples were collected in June 2004, July, September, November, February and May 2005. Sediments properties were measured including temperature, water contents, sediment bulk density, nutrient concentrations in porewater. Chlorophyll a concentrations in surface sediment (0.5 cm) were measured and relationships between the chlorophyll a and various sediment properties were analyzed to identify major mechanisms regulating biomass of benthic microalgae in the intertidal flats using simple linear regression analysis. Sediment chlorophyll a concentrations were maximum during winter and minimum during warm seasons ranging from 4.4 mg m?2 to 81.2 mg m?2. No clear spatial variations were observed for the sediment chlorophyll a in the study sites. Results from regression analysis suggested that benthic microalgae biomass was affected by sediment temperature and nutrients especially ammonium and silicate. Grazing effect was estimated using chlorophyll: pheopigments ratio, indirect indicator of grazing activity, and the positive correlation of the ratio and chlorophyll a implied that microalgae biomass is affected by grazing of zoobenthos although direct measurement of grazing activity is required to determine the importance of top-down controls in the benthic microalgae dynamics.
Intertidal regions have been considered as highly productive systems in the marine environments (Valiela and Teal 1979). In the intertidal flats, the benthic microalgae play an important role as primary producers (Sullivan and Daiber 1975; Rizzo and Wetzel 1985; Underwood and Kromkamp 1999). This primary production of organic compounds is the main food sources for microbial, microbenthic, meiobenthic and macrobenthic organisms abundant in the intertidal flats (Heip
In nutrient dynamics of the intertidal ecosystems, sediments play a role as source through regeneration to the water column deposited on the bottom (Hopkinson 1987) and as sink through benthic nitrogen retention processes including burial in deeper sediment layers and denitrification (Smith
The intertidal flats are located mainly in the west coasts of Korea (83%) and especially 41% of the intertidal flats located in the west and south coasts of Jeonnam Province (http://www.wetland.or.kr). Various studies have been reported focusing on dynamics of sedimentation (Choi 1992; Ryu
The loss of intertidal flats in Kwangyang Bay has increased since 1970s due to reclamation of shallowwater area for construction of large-scaled port and industry facilities. Water surface area was 233 km2 in 1973 and decreased to 193 km2 in 1990 (17% loss) and to 165 km2 (30% loss) in 2003 (Ryu 2003; Choi
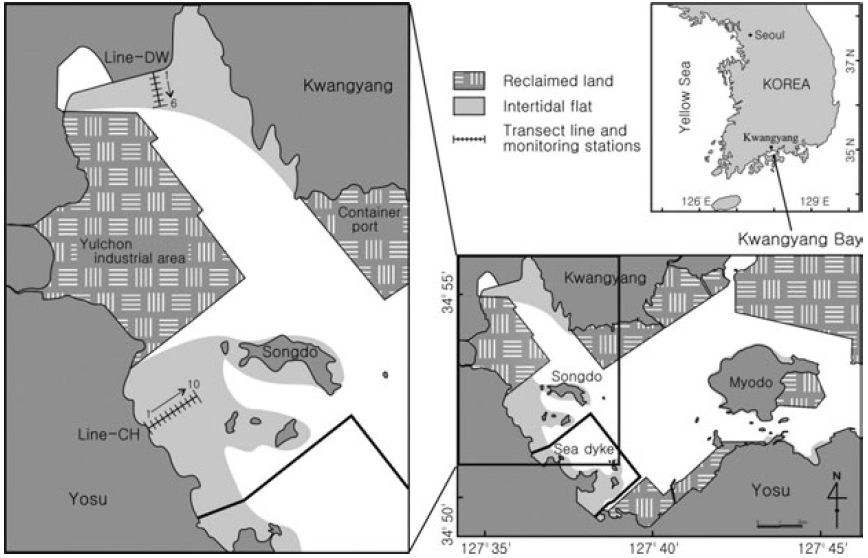
The Kwangyang Bay is located in the central region of South Coast and a semi-closed system surrounded by Yosu Peninsula and Namhae Island (Fig. 1). Study sites were selected from the intertidal flats in the range of 127° 34’-127° 39’ E, 34° 51’-34° 56’ N. The Kwangyang port (container) and Yulchon industry clusters are located in the east and west of the study sites and a sea dyke was constructed for reclamation of coastal regions for 2 years from 2003 to 2004 (Fig. 1). The intertidal flats in west of Kwangyang Bay were mainly reclaimed and study sites are the flats remained in the Kwangyang Bay. Dongcheon stream is extended in the north of study sites and freshwater is introduced especially during wet season. Main tidal channel is located between the Kwangyang Port and Yulchon industry clusters and has been dredged to maintain c.a. 7 m depth for transportation of vessels in the area.
Sediment types of the study sites are grouped in three classes including muddy sand, sandy mud and mud (Ryu 2003). Sediments in the northern regions of intertidal flats (Line-DW) are mainly composed of muddy sand whereas sediments in the western regions (Line- CH) are primarily sandy mud. Muds are abundant in the subtidal zones. Annual average of air temperature and rainfall of the regions is 14.1°C and 1893.5 mm respectively for 10 years from 1991 to 2000. The hydrological data presented in the study were collected by Korea Meteorological Administration.
>
Sample collection and analyses
Samples were collected at the beginning of low tide when the flats were exposed to air in June, July, September, November in 2004 and February and May in 2005. Stations were selected at interval of 100 m from upper to lower regions of the intertidal flats in Dowal-Ri (Line-DW) and Chohwa-Ri (Line-CH) (Fig. 1). Seasonal characteristics were investigated by classifying June-July as summer and September-November as fall. February and May were classified as winter and spring respectively.
Temperature of sediments was measured after inserting the mercury thermometer to the depth of 5 cm in the sediments for 1 or 2 minutes. Interstitial water of surface sediments (1 cm) was collected in-situ to measure ambient nutrients concentrations using a 100 ml syringe. The interstitial water was filtered using 25 mm Acrodisc Syringe Filters (0.45 μm) and inserted into HDPE bottle for freezing. Ambient concentrations of nutrients including nitrite+nitrate (NO2 ? + NO3 ?), ammonium (NH4 +), orthophosphate (PO4 ?3), and dissolved silica (DSi) were measured by the nutrient autoanalyzer (Bran Luebbe). Sediment cores (replicates) were collected using polycarbonate core tube (5 cm x 10 cm) to measure properties of sediments such as water contents (%) and density (mg cm?3) and chlorophyll
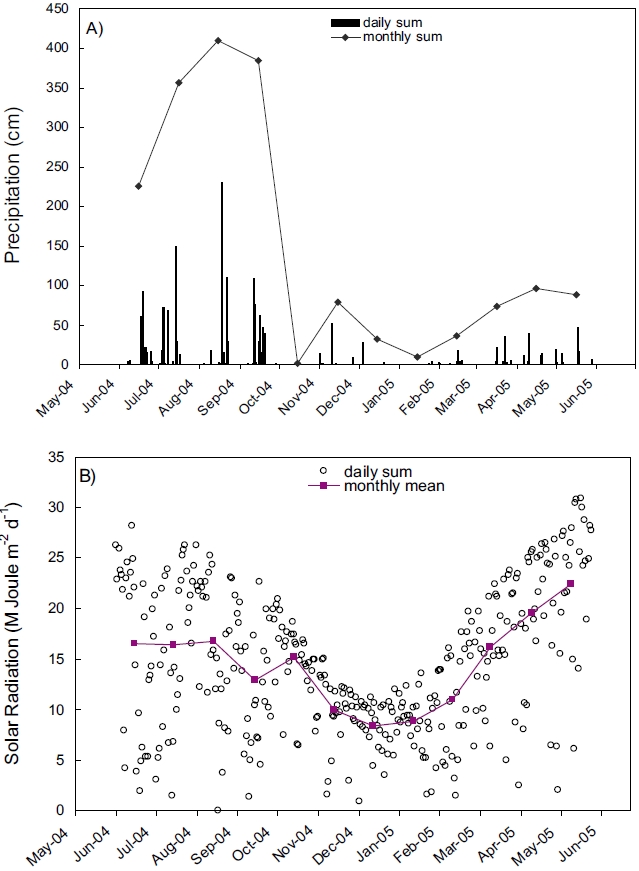
Records of rainfall and solar radiation collected near the Kwangyang Bay area were presented in Figure 2. Precipitation was extraordinarily high during summer compared with other seasons indicating that the study area is influenced by Monsoon climate (Fig. 2A). Solar radiation was also high during summer and low during winter similar to the annual patterns of other areas in mid-latitude (Fig. 2B).
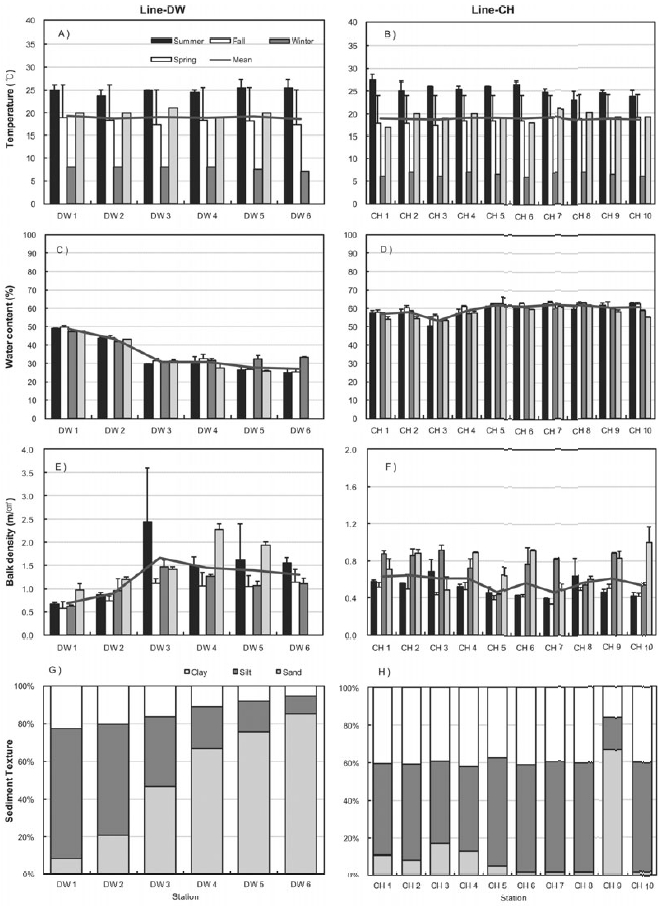
Figure 3 shows the seasonal characteristics of sediments including sediment temperature, water contents, bulk density and sediment texture at the sampling sites of Line-DW and Line-CH. Temperature was high in summer and low in winter and spatial difference was not high between the sampling sites (Figs 3A and 3B). Sediment temperature in the Line-CH was slightly higher than in the Line-DW. Seasonal variation of water contents in the study sites was not evident but water contents decreased as we approach seaward in the Line-DW whereas water contents were remained similar as seaward in the Line-CH (Figs 3C and 3D). Sediment bulk density was generally high during spring and summer whereas density was low during fall and winter in Line- DW (Fig. 3E). In Line-CH bulk density of sediments was relatively high during winter and spring and low during summer and fall (Fig. 3F). Bulk density was mostly low at the landward sites compared with that of seaward sites in Line-DW whereas spatial variation was not evident in Line-CW. Silt and clay were dominant in the sediment texture of the sites near lands but sediment texture was shifted to sand-dominant texture (coarse) as seaward in Line-DW (Fig. 3G). Silt and clay (fine) were dominant in sediment texture of Line-CH except Station CH9 where sand was dominated (Fig. 3H).
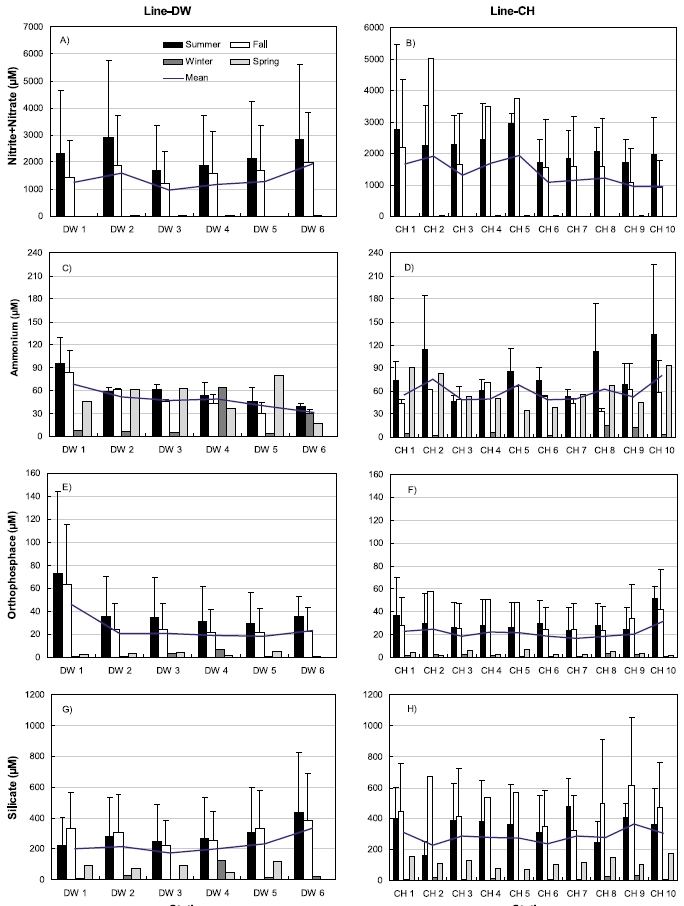
Nutrient concentrations in the sediments including nitrite+nitrate, ammonium, orthophosphate, silicate were shown in Fig. 4. Nitrite + nitrate concentrations were highest during summer and lowest during winter ranging from 1.67 μM (February) to 5767.2 μM (July) in Line-DW (Fig. 4A). Similar pattern was observed for nitrite+nitrate ranging from 0.57 μM (February) to 5461.2 μM (July) in Line-CH (Fig. 4B). No evident spatial variation was observed in Line-DW but the concentration slightly decreased as seaward in Line-CH. Ammonium concentrations were high during spring, summer and fall whereas the concentrations were low during winter in the Line-DW except Station DW4 (Fig. 4C) and similar seasonal pattern was observed in the Line-CH (Fig. 4D). Ammonium concentrations were decreased as seaward in Line-DW whereas no clear trend was detected in Line- CH. Orthophosphate concentrations were highest during summer and lowest during winter ranging from 0.68 μM (February) to 143.9 μM (July) in Line-DW except DW4 (Fig. 4E). Similar pattern was observed for orthophosphate ranging from 0.49 μM (February) to 76.4 μM (September) in Line-CH (Fig. 4F). The concentration in Line-DW was slightly decreased as seaward but no evident spatial variation was observed for Line-CH. Silicate concentrations were highest during summer or fall and lowest during winter except Station DW4 ranging from 3.4 μM (February) to 830.7 μM (July) in Line-DW (Fig. 4G). Silicate was maximum mostly during fall and mini-mum during winter ranging from 3.3 μM (February) to 1052.2 μM (June) in Line-CH (Fig. 4F). No evident spatial variations were observed in Line-DW and Line-CH.
>
Variations of benthic microalgae biomass (chlorophyll a)
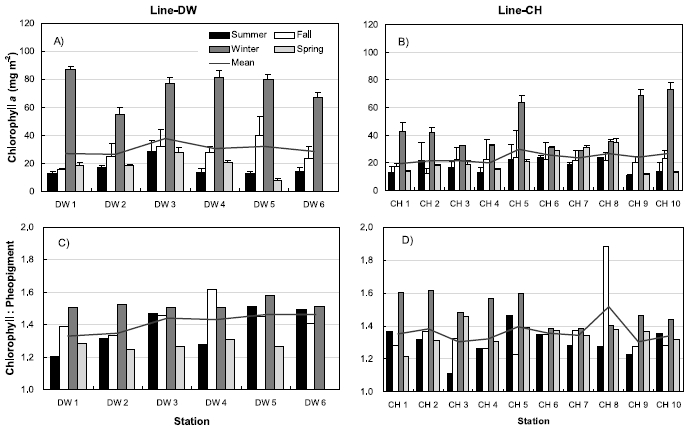
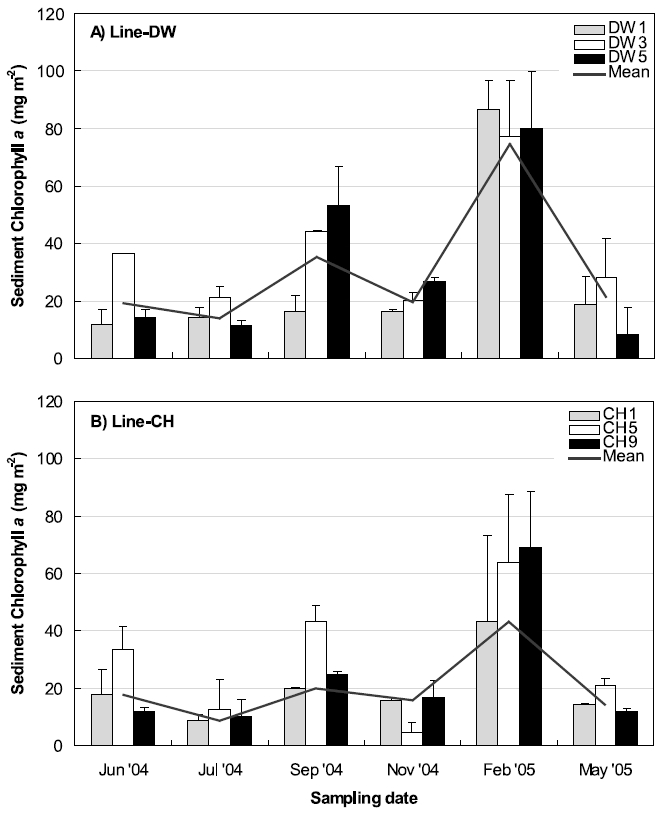
Figure 5 shows the seasonal variations of sediment chlorophyll
Three stations were selected between land boundary and boundary of open ocean at Line-DW and Line-CH respectively to compare the sediment chlorophyll
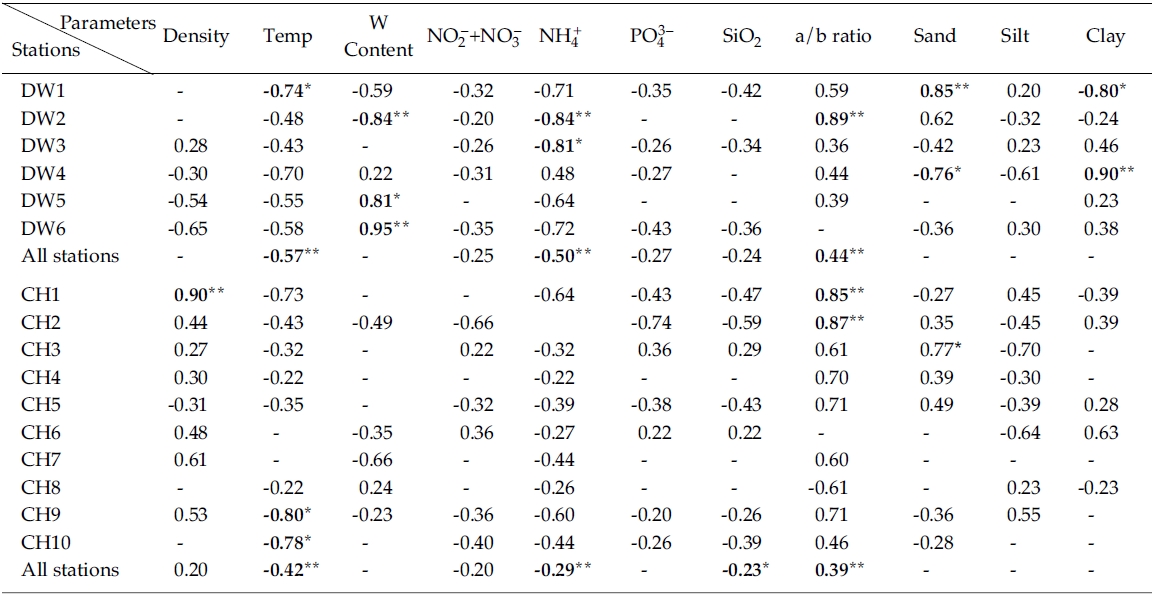
Results from simple linear regression analysis on chlorophyll
Negative correlation was observed between nitrate + nitrate and in Line-DW and similar relationship was generally observed in Line-CH except Stations CH3 and CH6 where positive correlation was detected. Significant negative correlation was observed between ammonium and chlorophyll
The ratios of chlorophyll:pheopigment were positively correlated with in Line-DW and similar relation was detected in Line-CH except Station CH8, suggesting that benthic microalgae were influenced by physical environmental stress and/or biological interaction such as grazing. Percentage of sand in sediment texture was significantly and positively correlated with chlorophyll
Multiple regression analyses indicated that sediment temperature, water contents, chlorophyll:pheopigment, clay % and ammonium were responsible for 64% of the variation in sediment chlorophyll
Sediment microalgae play an important role both as food for benthic organisms and a source of nutrients for the overlying water column after decomposition (Middlelburg
de Jonge (1985) reported that the diatoms were dominant in mud coating sand grains since mud rather than in the bare parts of the sand grains systems since mud serves as a substratum for diatoms on the tidal flats. Lower rates of primary production were observed at coarse grain-sized sediments (greater exposure to water energy) than fine grain-sized sediments (Cook
Nutrient concentrations in the Line-CH were generally higher than those in Line-DW (Fig. 4). This difference may be resulted from different water contents (Figs 3C and 3D) caused by gradient of grain size in sediment between the Line-DW and Line-CH (Figs 3G and 3H). A more stable habitat with higher concentrations of reduced nutrients such as ammonium has been reported to be favored by benthic microalgae within soft sediments (Saburova and Polikarpov 2003). The preference of microalgae to the stable habitat with high levels of nutrients was not observed in this study since chlorophyll
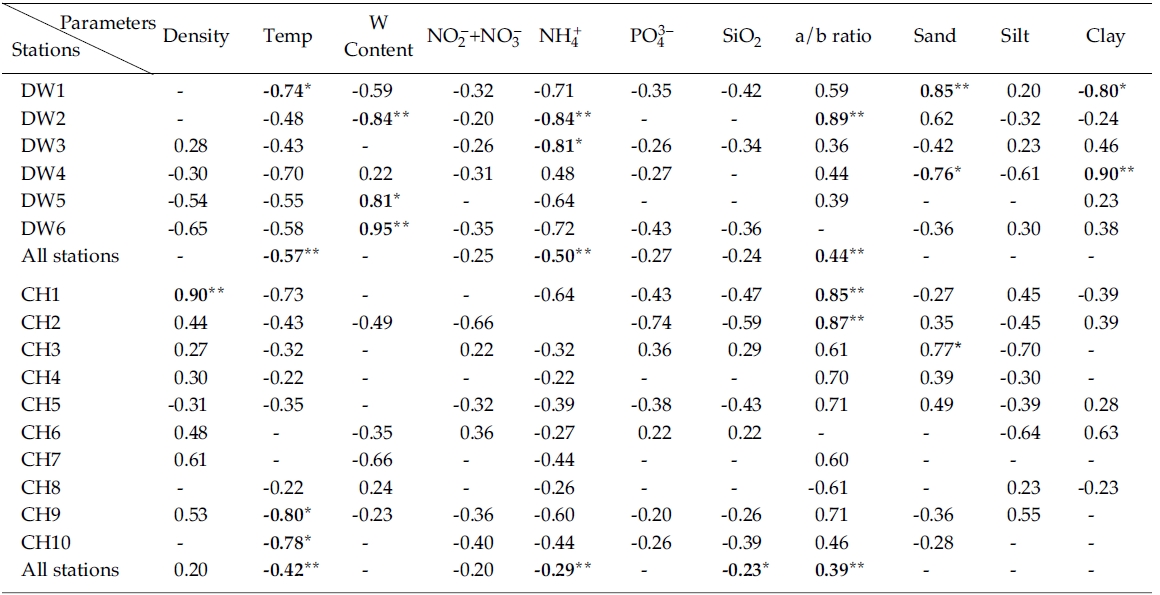
Results (R2) of simple linear regression analyses of sediment chlorophyll a vs. various sediment properties from sampling sites. R2 values less than 0.2 were omitted and denoted by ‘-’ (Negative values denote negative relationships)
Aquatic and benthic microalgae biomass is controlled by top-down such as grazing by consumers as well as bottom-up mechanisms including physical and chemical variability (Goldfinch and Carman 2000; Worm
Algal colonized sediments can utilize nitrate, and thus reduce nitrogen fluxes to the water column (Tyler