Panax ginseng is one of the most popular natural tonics that have been used in oriental countries. Ginseng showed anti-tumor activities in slow growing tumors but not in rapidly growing tumors1). Ginseng also inhibited tumor angiogenesis and metastasis2,3). In an epidemiological study, the intake of ginseng reduced the incidence of human cancer4). However, its mechanisms of action have not been elucidated yet.
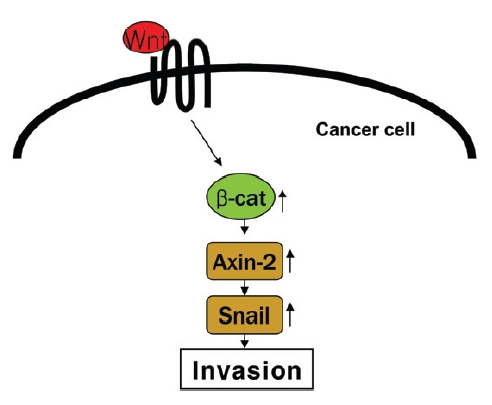
The canonical Wnt signaling pathway has a dominant role in regulating epithelial-mesenchymal cell interactions critical for morphogenesis5,6). In this signaling system, Wnt ligands bind to a coreceptor complex consisting of a seven-transmembranedomain, Frizzled receptor and one of the LDL receptor-related proteins (LRPs), LRP5 or LRP67). A signaling cascade is then engaged that leads to the formation of a bipartite complex between the adhesion molecule, β-catenin, and transcription factors of the TCF family. In turn, the β-catenin-T-cell factor(TCF) complex activates expression of a cohort of target genes hat impact on cellular function. Although the TCF complex as an important role in regulating normal cell function, increasing evidence indicates that canonical Wnt signaling can be disrupted in various cancerous states8,9).
Accumulating evidence indicates that hyperactive Wnt signaling occurs in association with the progression of human cancer. As a consequence of engaging the canonical Wnt pathway, a TCF transcriptional complex is generated, which has been postulated to trigger the epithelial-mesenchymal transition (EMT), which convert epithelial cells into migratory mesenchymal cells10). Recent study has demonstrated that canonical Wnt signaling engages tumor cell dedifferentiation and tissue-invasive activity through an
Based on the grown environment and the cultivate method, the commercial trade ginseng is classified into three grades of ginseng, cultivated ginseng( CG), mountain cultivated wild ginseng(MCWG) and mountain wild ginseng(WG). The CG is cultivated artificially in forms and contributes the major quantity of ginseng in the current market. The WG grows in the natural environments, vegetating in the deep mountains and MCWG can be considered as mimicry WG which is seeded and grown up in the forest and mountain.
In this study, we examined the effect of MCWG on the transcription of the human
1. Preparation of ginseng extracts
Cultivated ginsengs(CG) used in this experiment were 5 years of age(Fig. 1-a). Mountain cultivated wild ginseng(MCWG) used in this experiment were of 10 years old(seeded in 1999) grown at ChonBangNongSan in Choongnam, Korea, (Fig. 1- b). Wild ginseng(WG) used in this experiment were collected from Changbai Mt. in July, 2007. They are about 20-40 cm long, dried weighs about 10- 20g with the approximate age of 20~50 years(Fig. 1-c).
Selected dried ginseng, CG, MCWG and WG(5g) were added with 80% ethyl alcohol 500㎖ and refluxed for 3hours, 3times to obtain extraction. Then this extraction was suspended in a distilled water for using experimental studies.
2. Cell culture and Cell lines
Human cancer cell lines were obtained from the Korean Cell Line Bank (KCLB, Korea) and were maintained in the recommended medium containing 10% heat-inactivated fetal bovine serum (FBS), 100 U/ml penicillin, 100 μg/ml streptomycin and 0.25μg/μl amphotericin B at 37℃ under a humidified 5% CO2 atmosphere.
RNA was extracted from various ginsengs treated cells using Easy-spin RNA Extraction kit (iNtRON, Biotech, Korea) according to the manufacturer’s instructions.
4. Reverse transcriptase-polymerase chain reaction (RT-PCR) analysis.
The reverse transcription reaction was performed on 2μg of total RNA using a SuperScript II First-Strand Synthesis system (Invitrogen) with random primer. PCR was carried out in solution containing 1X PCR buffer (TaKaRa, Japan), 200μM each deoxynucleotide triphosphate, 2.5pmol of each primer, 1 unit of ExTaq polymerase (TaKaRa, Japan). The housekeeping gene β-actin served as an internal control to confirm the success of the reverse transcription reaction. The PCR products were subjected to 2% agaroseg elelectrophoresis.
Volume of the reaction mixture was made up to 20μl. Real-time quantitative PCR was performed using SYBR GreenER qPCR SuperMix reagents (Invitrogen) and a Bio-Rad iCycler. Relative transcript quantities were calculated using the ΔΔCt method with β-actin as the endogenous reference gene amplified from the samples(Table 1.).
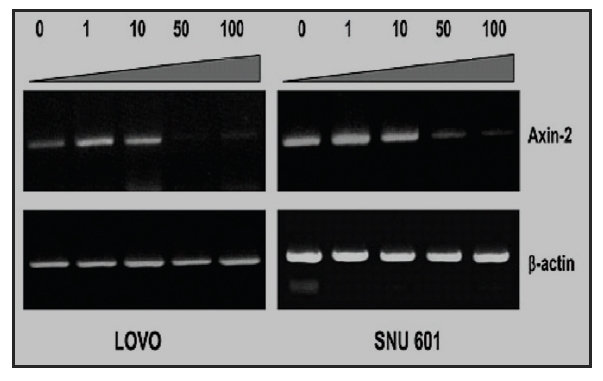
1. Effect of mountain cultivated wild ginseng on the expression of AXIN2 Gene
Recent study11) has demonstrated that canonical Wnt signaling engages tumor cell dedifferentiation and tissue-invasive activity through an AXIN2- dependent pathway(Fig. 2). Thus, the effect of cultivated wild ginseng on the expression of the
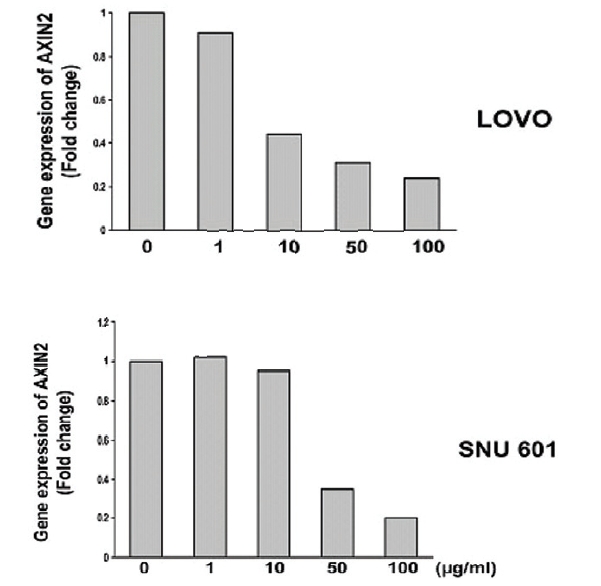
Quantitative real-time PCR results confirmed a dose-dependent decrease in
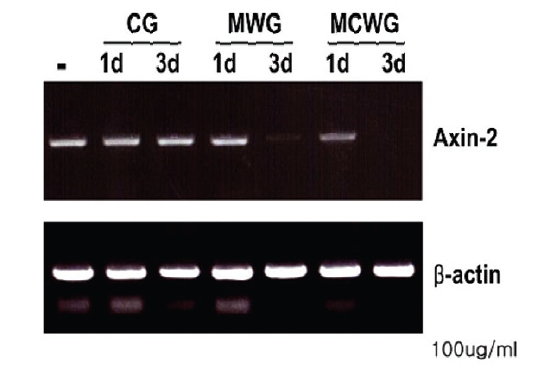
2. A comparison analysis of three types of ginseng on the expression of AXIN2 gene
Both of MCWG and WG are considered superior to CG. We therefore effects of these three types of ginseng in the expression of
3. Effect of WG on the expression of metastasissuppressor genes
Maspin (mammary serine protease inhibitor, 42 kDa) is a metastasis-suppressor gene first described by Zou et al in 1994 by subtraction of cultured breast cancer cell and normal breast tissue mRNAs12). Maspin has been thought to inhibit carcinoma invasion, metastasis, and angiogenesis. In addition, it has been reported that maspin induced apoptosis of neoplastic cells13) and expression of maspin are suppressed as the carcinoma progresses in breast carcinoma and prostatic carcinoma14,15).
As shown in Fig. 6, RT-PCR analysis revealed that the expression of two metastasis-suppressor genes, maspin and nm23 was not affected by the treatment of ginseng extracts in LOVO cells.
Metastasis is the major cause of death for cancer patients with solid tumors, due mainly to the ineffectiveness of current therapies once metastases begin to form. Metastasis is defined as the formation of progressively growing secondary tumor foci at sites discontinuous from the primary lesion20). In cancer, it is believed that epithelial tumor cells may be able to convert differentiated epithelial cancer cells into de-differentiated cells that possess more mesenchymal characteristics21). This epithelial-mesenchymal transition (EMT) phenotype in cancer has been associated with a decrease in tumor growth, increased resistance to apoptosis, increased motility and invasiveness, and enhanced metastatic ability22). These phenotypic transitions are reversible, and it is hypothesized that once tumor cells have reached their destination, they may transform back into an epithelial phenotype in order to facilitate tumor growth in the secondary site23). EMT is an important process in tumor development, and several studies suggest that the Wnt/β-catenin signal pathway may play an important role in EMT .
The present study focuses on identifying the mechanism that underlies the anti-metastasis activity of ginseng. The study shows, for the first time, that ginseng can repress the expression of AXIN2 gene that play key roles in the downsream signaling of the neoplastic EMT process. These results showed us a novel mechanism of ginseng’s antimetastasis activity and could provide a molecular link between ginseng intake and its inhibitory effects on metastasis.

Primer for RT-PCR