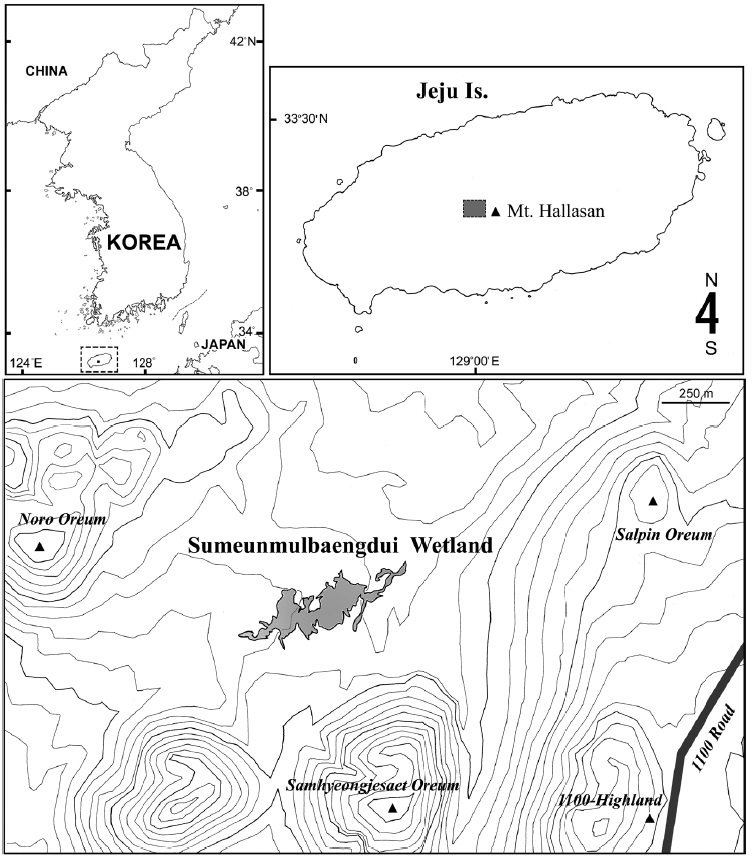
Jeju Island, the largest island in South Korea, is dominated by Mt. Hallasan, a 1,950 m high shield volcano and the highest mountain in the country. As the island mainly consists of basaltic and trachytic lava deposits, and rain water readily percolates through the underlying volcaniclastic layers, natural surface water bodies are not common on the island (Smith et al., 2014). A few mountain wetlands exist between the secondary (parasitic) cones (called ‘oreum’ in the local Jeju dialect) on the flank of the volcano. Recently an alpine wetland, Sumeunmulbaengdui, was reported from the northwestern hillside of Mt. Hallasan near the 1,100-Highland, at a height of 980 m above sea level between Samhyeonjesaet oreum (1,114 m), Noro oreum (1,070 m), and Salpin oreum (1,076 m). Sumeunmulbaengdui was registered as a Ramsar Wetland in 2015 and is the largest alpine wetland in South Korea, having an area of 43,602 m2 and a circumference of about 2,474 m (Ko et al., 2014). On 11 Sep 2010, the junior author (C.Y.C.) participated in a preliminary investigation of the wetland sponsored by the National Institute of Biological Resources, Korea, and obtained a large amount of small aquatic invertebrates, including copepod specimens.
Among the copepods obtained, the most noticeable and dominant species was a harpacticoid species belonging to the genus
Several specimens of both sexes of
Copepods were collected from an alpine wetland on Mt. Hallasan, Sumeunmulbaengdui (33°21′25-28″N, 126°27′ 43-57″E, 980 m altitude; about 90 cm in depth at its deepest point) (Fig. 1).
Collections were made with a hand net and a dipnet, both with a mesh size of 64 μm. Samples were fixed in the field by immediately adding about 10% solution of formalin (equivalent of a 4% solution of formaldehyde in water by weight). In the laboratory, specimens were sorted from the samples under a zoom-stereomicroscope (Leica M165C, Wetzlar, Germany) at high magnifications (20-120×) with a micropipette, and transferred to about 80% ethanol to which glycerine was added for long-term preservation.
Methods for dissection and double-coverglass preparation using H-S slides followed those in the latest paper of the junior author (Chang, 2012). Drawings and measurements were made at 630× or 1,000× magnification using a drawing tube mounted on a differential interference contrast microscope with Normaski optics (Leica DM2500).
Holotype specimen is deposited in the National Institute of Biological Resources (NIBR), Incheon, Korea, and paratype specimens are kept temporarily in the collection of the junior author (C.Y.C.) at the Department of Biological Science, Daegu University (DB), Korea.
The descriptive terminology follows Huys and Boxshall (1991). Abbreviations used in the text and figure legends are as follows: enp-1 to enp-3 or exp-1 to exp-3, the first to third endopodal or exopodal segments of each leg.
>
Bryocamptus jejuensis n. sp. (
Non
Type locality. Sumeunmulbaengdui wetland (33°21′27″N, 126°27′53″E; at 980 m altitude), Mt. Hallasan, Jeju Island, South Korea.
Material examined. Holotype ♂ (NIBRIV0000552737), undissected, ethanol-preserved, from Sumeunmulbaengdui, 11 Sep 2010 (coll. C.Y. Chang). Paratypes: 2♀♀ (DB20056, 20057), 2♂♂ (DB20058, 20059), dissected on slides; 4♀♀ (DB20060), 2♂♂ (DB20061), ethanol-preserved, collection details same as for the holotype.
Additional material: 1♀, 1♂, dissected on slides, from 1100-Highland, 13 February 1988 (coll. C.Y. Chang).
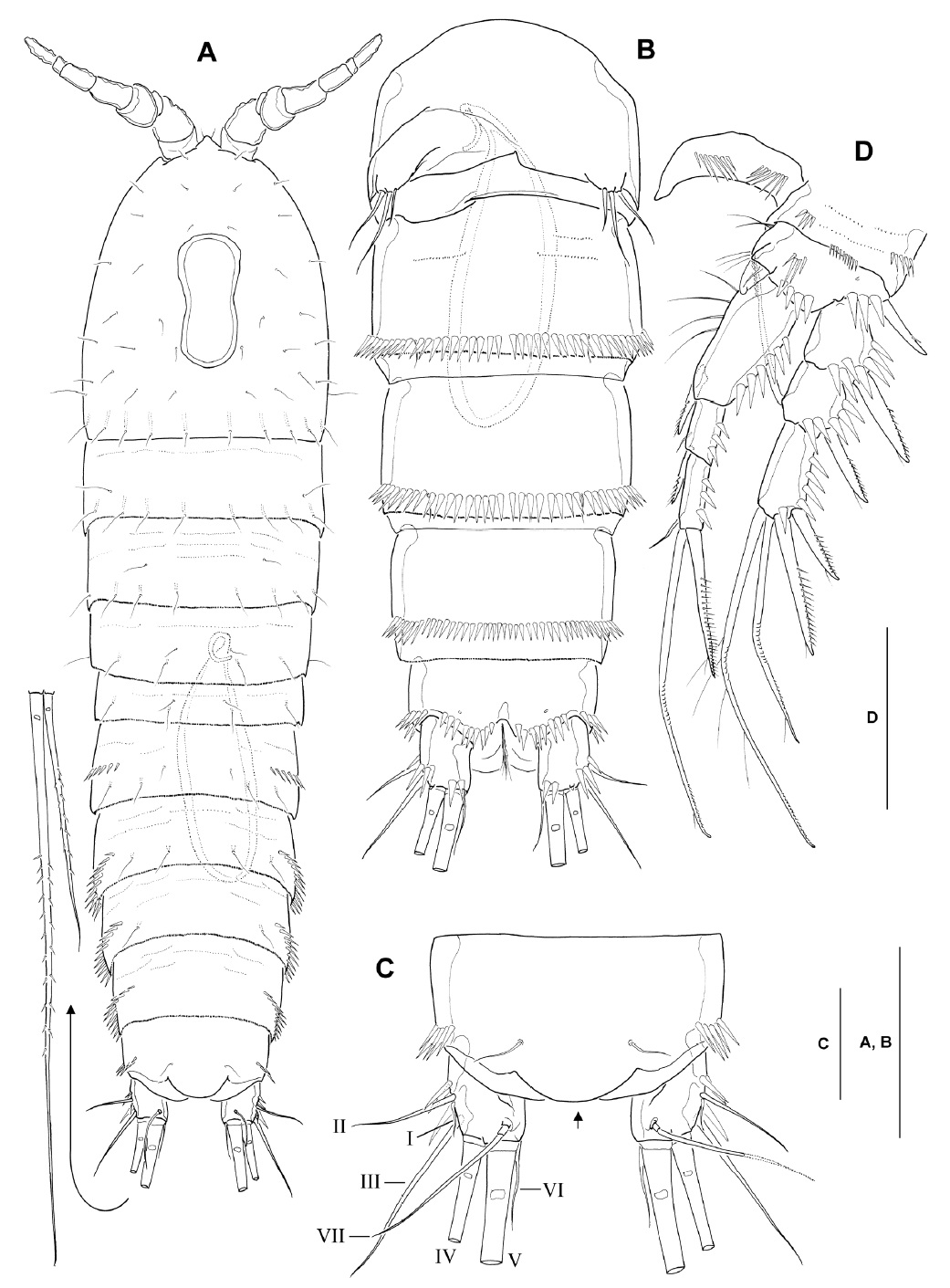
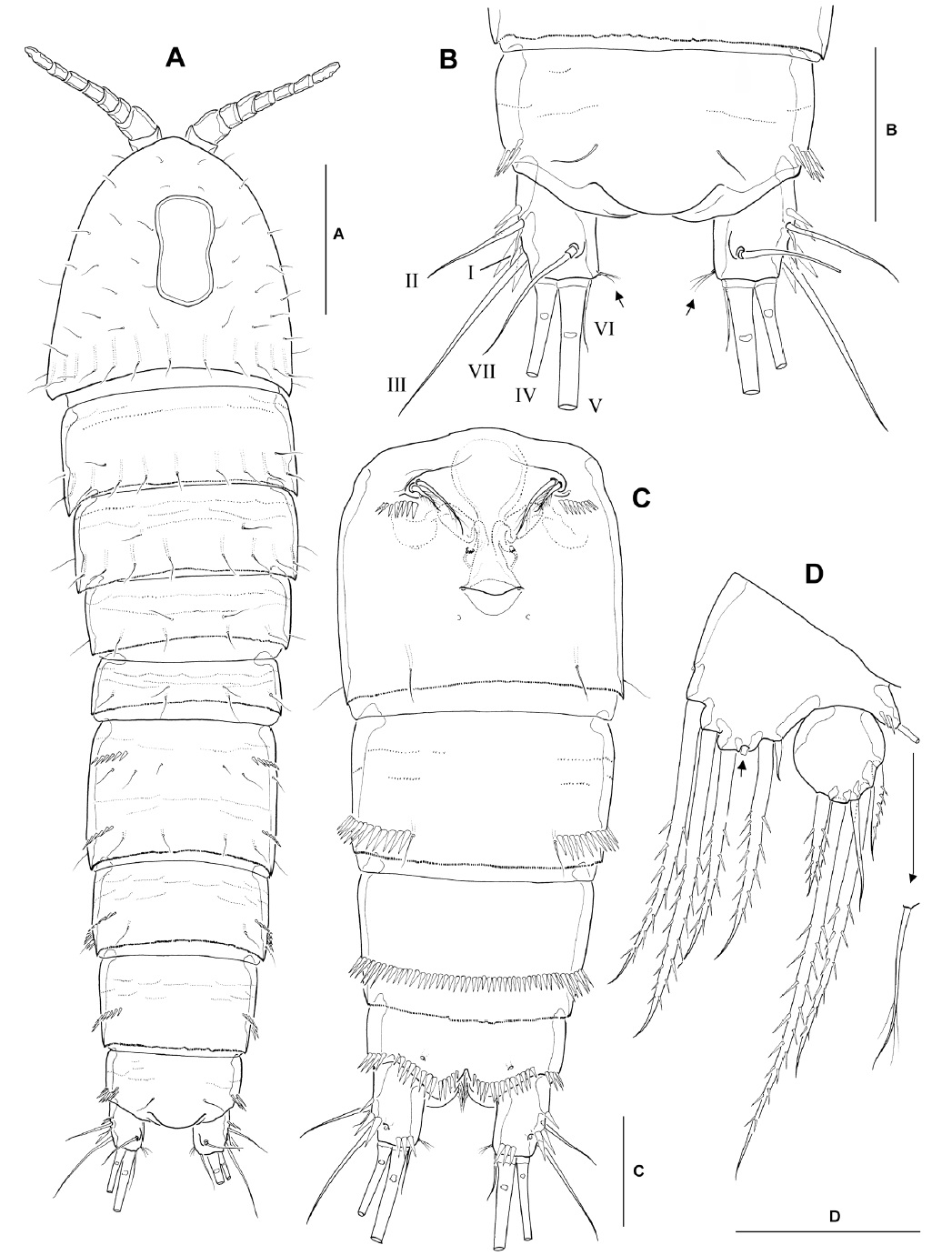
Description. Male: Body (Fig. 2A) cylindrical; body length ranging from 536 to 583 μm (mean 564 μm, n=6; holotype 570 μm), excluding caudal setae. Prosome about 1.2 times longer than urosome, slightly narrowing posteriorly. Rostrum weakly developed, blunt, slightly protruding anteriorly, with 2 dorsal sensilla subdistally; not defined at its base. Cephalothorax with scattered sensilla, with peanut-shaped integumental window mid-dorsally. Free prosomites with 1 pair of sensilla dorsolaterally, and transverse row of long sensilla along posterior margin of each somite. Urosomites (excluding fifth pedigerous somite) with dorsolateral spinules along posterior margin. Genital somite and remaining urosomites with well-developed continuous row of strong spinules laterally and ventrally along posterior margin (Fig. 2A, B). Spermatophore (Fig. 2A, B) elongate, ellipsoidal (143×45 μm), with coiled basal neck.
Anal somite (Fig. 2B, C) with smooth dorsal surface, except for paired sensilla anterior to anal operculum; armed with transverse rows of strong spinules laterally and ventrally along posterior margin of somite. Anal operculum convex with smooth posterior margin (Fig. 2C, arrow).
Caudal rami (Fig. 2A-C) subparallel, truncate, 1.3-1.4 times longer than wide in ventral view, with smooth inner margin; ventral surface with 1 oblique row of 3 sharp spinules near outer margin at bases of setae II-III, and 2 strong spinules ventromedially, at base of inner terminal caudal seta (seta V); armed with 7 setae. Seta I short, arising slightly ventral to seta II (Fig. 2C). Seta II relatively long, naked, located near middle of outer margin of ramus. Seta III naked and slender, situated at outer distal corner of ramus, not flanked by dorsal spinules at its base. Seta IV pinnate, with fracture plane in basal part, slightly shorter than half the length of seta V (Fig. 2A). Seta V pinnate between anterior quarter and anterior two-thirds, slightly less than half the body length. Seta VI short, naked and slender, slightly longer than ramus. Seta VII situated anterior to seta V, naked, with basal socket, about twice longer than ramus.
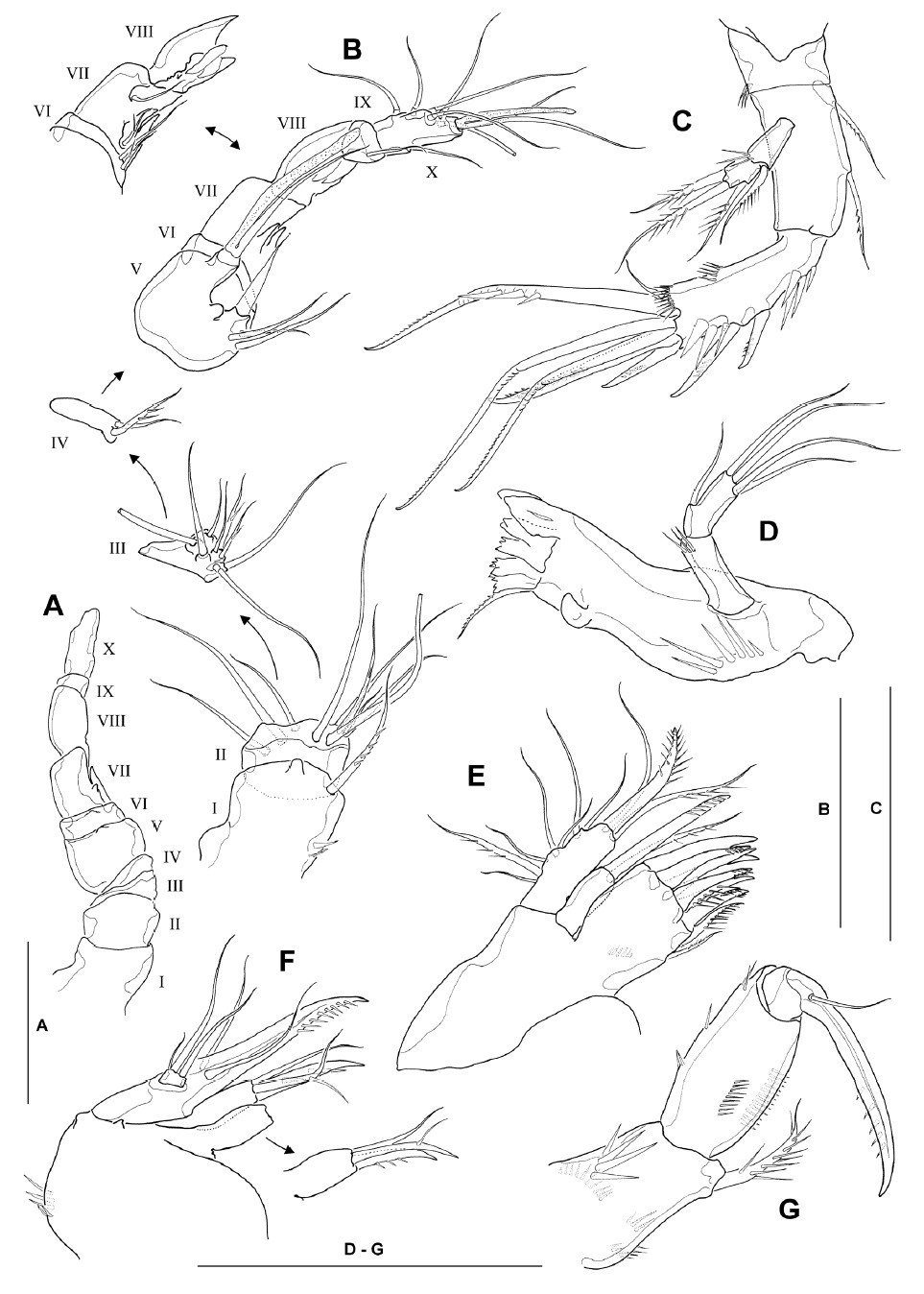
Antennule (Fig. 3A, B) subchirocerate, about two-thirds as long as cephalothorax, 10-segmented; geniculation located between segments 7 and 8; anterior margin of segment 1 with 4 spinules proximally and 1 long pinnate seta distally; segment 2 with membranous insert dorsally, bearing 9 slender naked setae; segments 3-4 relatively short, with 8 and 2 setae, respectively; segment 5 bulbous, bearing 1 long aesthetasc distally, its tip nearly reaching to distal end of penultimate segment and its base fused to seta of approximately the same length; segment 6 with 2 minute setae proximally; segments 7 and 8 with spiniform projections (modified armature elements) on anterior margin; segment 9 small, bearing 1 slender seta distally; segment 10 armed with 6 posterior, 1 anterior and 2 apical setae (the latter fused at base to aesthetasc). Armature formula: 1-[1 pinnate], 2-[9], 3-[6+2 pinnate], 4-[1+1 pinnate], 5-[3+1 spine+(1+aesthetasc)], 6-[2], 7-[2+2 modified], 8-[2 modified], 9-[1], 10-[7+(2+aesthetasc)].
Antenna (Fig. 3C), coxa small, with spinules at outer distal corner; allobasis 1.7-1.8 times as long as wide, with 2 pinnate setae on abexopodal margin; free endopodal segment armed with 2 lateral spines and 6 distal elements, comprising 2 unipinnate spines and 3 geniculate setae, the outermost of which being fused at the base to a minute seta; exopod 2-segmented, comprising cylindrical exp-1 with 1 inner distal pinnate seta and exp-2 with 1 inner and 2 apical pinnate setae.
Mandible (Fig. 3D), with well-developed coxal gnathobase bearing 5-6 bicuspidate and multicuspidate teeth and 1 pinnate seta along distal margin; with strong spinules near base of palp; palp well-developed, comprising basis and endopod; basis cylindrical, with setules near inner distal corner; endopod about 2.4 times as long as broad, with 1 lateral and 4 apical naked setae.
Maxillule (Fig. 3E) with praecoxal arthrite bearing 7 unguiform elements and 1 pinnate seta around distal margin, and 1 pinnate seta on distal inner margin; coxal arthrite with cylindrical endite bearing 1 pinnate spine and 1 slender seta apically; exopod and endopod completely fused to basis, bearing 1 pinnate and 6 naked setae laterally, and 1 bipinnate spine, 1 pinnate seta and 1 naked seta apically; origin (exopodal, endopodal or basal) of lateral setae indeterminable.
Maxilla (Fig. 3F) armed with 2 syncoxal endites, each bearing 1 unguiform spine and 2 setal elements; allobasis produced into strong unipinnate claw, flanked by 1 anterior and 1 posterior naked seta proximally; endopod a minute segment bearing 1 short and 2 well-developed naked setae distally.
Maxilliped (Fig. 3G) subchelate; syncoxa armed with 1 stout bipinnate seta at inner distal corner, surface with 4 spinular rows; basis about 1.8 times longer than wide, with longitudinal row of spinules on both anterior and posterior surfaces near medial margin; spinule row on posterior surface consisting of 12-14 spinules; few spinules present along outer margin; endopod bearing 1 strong, curved claw with 1 minute accessory seta arising from its base.
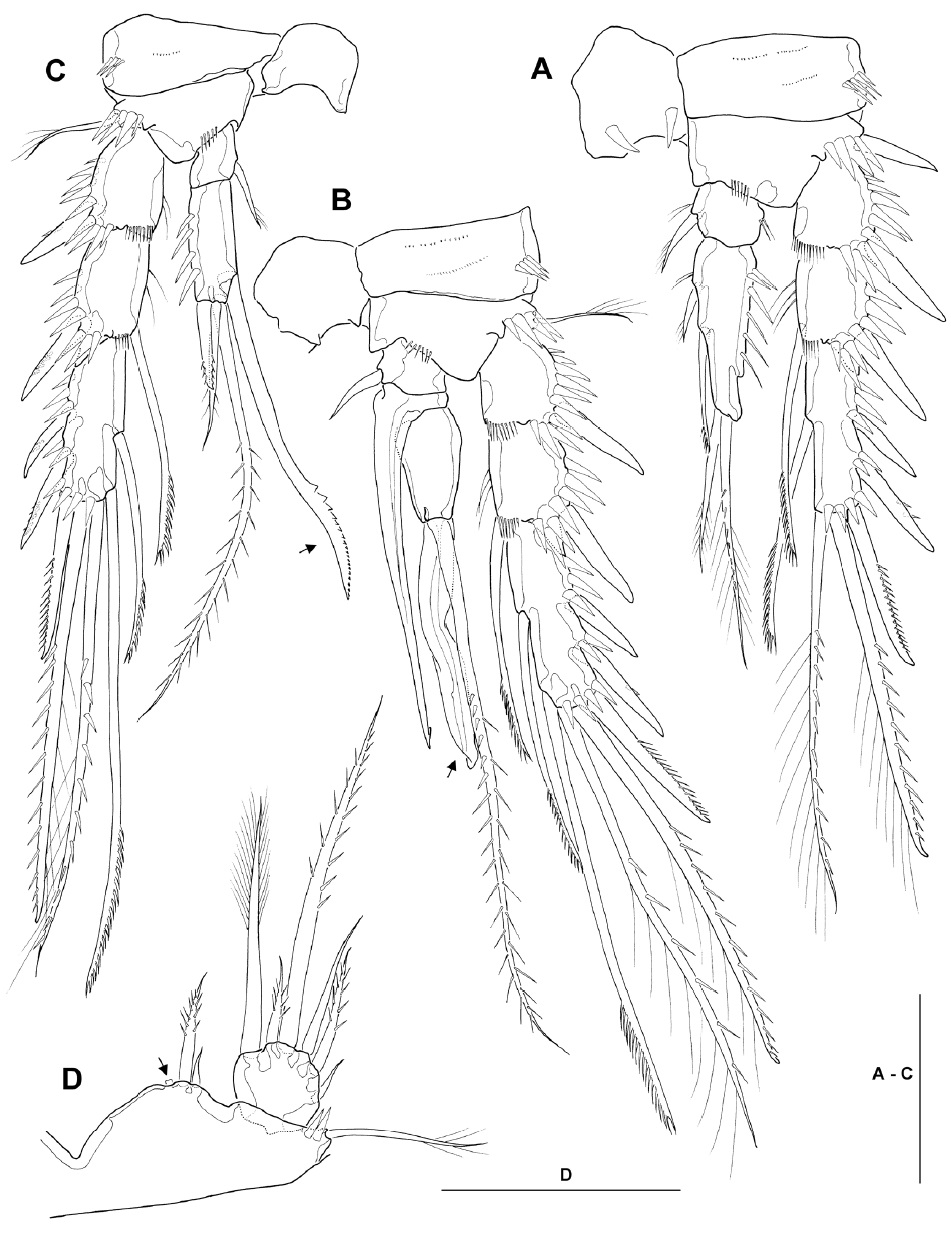
Leg 1 (Fig. 2D) with 3-segmented rami; all segments with strong spinules along outer margin. Intercoxal sclerite with paired spinule rows consisting of 7 sharp spinules each on anterior surface. Coxa with 2 groups of spinules near medial margin and lateral margin, respectively, about 12 fine setules in middle of posterior margin, and 2 transverse rows of minute spinules. Inner distal element of basis represented by stout short spine with blunt tip slightly recurved outwardly, not reaching to proximal quarter of enp-1. Enp-1 about 2.1 times as long as wide, reaching just beyond posterior margin of exp-2, with short, pinnate seta near inner distal corner; enp-2 with stout, pinnate seta near inner distal corner; enp-3 with 1 short inner seta subdistally, and 1 unipinnate spine and 1 long geniculate seta distally. Exp-1 with stout outer spine; exp-2 not elongate, with dorsally recurved inner distal seta, serrated in distal third; exp-3 with 2 outer spines and 2 apical geniculate setae.
Legs 2-4 (Fig. 4A-C) with small enp-1, each bearing inner distal seta; exp-1 lacking inner seta. Leg 2 (Fig. 4A), anterior surface of intercoxal sclerite with pair of sharp, incisor-like spinules near concave free margin; basis with strong, naked, outer spine; enp-2 elongate, about 2.8 times as long as broad, with distinct notch along distal outer margin, bearing 2 pinnate setae on inner margin and 2 plumose apical setae, of which outer one about 1.5 times as long as inner one, and ornamented with secondary spinule row; exp-3 with 3 spines along outer margin, one spine and one seta apically (both with mixed ornamentation as figured) and 1 pectinate seta along inner margin. Leg 3 (Fig. 4B), intercoxal sclerite with paired spinous processes on concave free margin; enp-2 short, inner distal margin produced into spinous apophysis, proximal part broad and hollow inside, narrowing posteriorly to form minute barb, reaching almost to end of modified spine of enp-3; enp-3 pyriform, apex with long, strongly pinnate seta and long modified spine, shown as ribbon-like process, with a long, narrow incision or groove as figured; exp-3 armed with 3 spines along outer margin, 2 setae apically (both with mixed ornamentation as figured) and 2 pectinate setae along inner margin. Leg 4 (Fig. 4C), enp-2 bearing 1 outer distal spine, 2 apical plumose setae (inner one about 3 times as long as outer one), and 1 inner distal spinous seta with curved serrated inner margin in distal third; seta/spine armature of exopod same as in leg 3.
Leg 5 (Fig. 4D), baseoendopod with weakly produced endopodal lobe, reaching to just beyond middle of inner margin of exopod, bearing 1 short outer and 1 stout inner setae, flanking 1 membranous tube-pore (Fig. 4D, arrow); exopod slightly wider than long, L/W about 0.9, bearing 6 setae in total, comprising (from inner to outer) distally plumose seta, short bipinnate inner distal seta, long bipinnate apical seta, slender, naked outer seta, bipinnate outer seta, and minute, naked, outer seta.
Female: Body (Fig. 5A) cylindrical, slightly tapering posteriorly; body length ranging from 664 to 710 μm (mean 683 μm, n=6). Genital double-somite completely fused both dorsally and ventrally (Fig. 5A, C), without trace of subdivision by subcuticular chitinous rib; paired transverse rows of 7-8 spinules situated posterior to leg 6; copulatory pore located on ventral midline, immediately posterior to genital apertures; pair of long sensilla present near posterior margin of genital double-somite. Third (antepenultimate) urosomite with dorsolateral and ventrolateral spinules along posterior margin; fourth (penultimate) urosomite with continuous ventral spinule row (Fig. 5C). Anal somite (Fig. 5B, C) nearly same as in male, including smooth posterior margin of anal operculum.
Caudal rami (Fig. 5B) slightly longer than in male, 1.3-1.4 times as long as wide in ventral view; setular tuft present along distal inner margin of ramus (arrowed in Fig. 5B; absent in male).
Seta/spine armature of legs 1-4 as follows (as for figures of female legs 1-4, see Lee and Chang, 2006):
Leg 2, enp-3 with long apical plumose seta, about 2.3 times as long as adjacent subapical seta. Leg 3, enp-3 with 2 equally long apical setae. Leg 4, enp-3 bearing 5 elements in total, comprising 1 subapical spine, 1 short, pinnate outer distal, 1 elongate, plumose inner distal, and 2 inner pinnate setae.
Leg 5 (Fig. 5D), baseoendopod with well developed endopodal lobe, reaching slightly beyond middle of exopod, with 4 stout spiniform, bipinnate setae, flanked by 1 minute, naked outermost seta, remaining prominent gap between inner third and fourth pinnate setae (arrowed in Fig. 5D); exopod nearly circular, with naked inner margin, bearing 5 setae in total, comprising 1 short bipinnate innermost seta, 1 longest, apical, bipinnate seta, 1 slender, naked, subapical seta, and 2 bipinnate outer setae.
Remarks. Four subgenera are currently recognized in the genus
Among the other members of the subgenus
In
Wilson (1958) remarked that
Lee and Chang (2006) reported five species of the genus