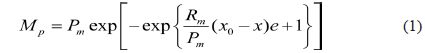Due limited natural resources and increased greenhouse gas emissions, substitution of fossil fuels with renewable bio-energy has been encouraged United Nations Framework Convention on Climate Change (UNFCCC). According to the United Nations, by the year 2050, up to 77% of the world’s energy demand could be supplied by renewable energy sources [1]. However, because biomass has gained economic interest, further expansion of biogas production is increasingly dependent on the exploitation of new sources of biomass.
Biomass is composed of carbon-rich materials including plant residues, animal excrements and bio-waste from households and industry [2]. Unused or discarded biomass residues from agriculture are a potential energy resource, even though those materials can be a source of green house gas (GHG) emissions causing climate change. The potential energy production from crop residues and animal wastes is globally estimated at about 34 EJ (exajoule = 1018 joules) out of a total 70 EJ [3]. Biomass is a renewable energy resource derived from all organic materials produced by both human and natural activities. It is a complex mixture of carbohydrates, fats and proteins. Plant residues are regarded as the main source of biomass because they usually contain 40%-50% cellulose, 20%-35% hemi-cellulose, and 15%-30% lignins. Therefore, biomasses like the plant residues of grapeseed, rice, barley and wheat could serve as alternative bio-energy resources.
The grand total of generated waste from livestock, agro-industrial waste and crop residues is estimated at 58,010 kt yr-1 in Korea. Potential methane production from agricultural wastes including livestock, crop residues and agro-industrial wastes is estimated at 436 kt yr−1 [4]. Anaerobic digestion (AD) for methane production using crop residues in the agricultural sector is becoming necessary in areas with limited natural resources like Korea. Over the past decade, AD of biomass originating from agriculture to produce methane gas has attracted much attention from government authorities, academia, and industry in Korea.
AD is a biological process that converts the solid or liquid biomass into a gas in the absence of oxygen. Many studies on AD in Korea have focused on swine manure and food wastes as substrates [5-6]. AD has many environmental benefits including the production of a renewable energy carrier, nutrient recycling and the reduction of waste volumes [7-9]. Therefore, different types of organic wastes such as sewage sludge, industrial wastes, slaughterhouse waste, fruit and vegetable wastes, manure and agricultural biomass have been successfully digested under anaerobic conditions. Each of these wastes has been digested separately or co-digested with other wastes [10-12].
However, not many papers have been published on the mono-digestion of crop residues [13-16], while thermophilic AD has been mainly the subject of more recent studies.
The organic loading rate and hydrologic retention time (HRT) are two major parameters used for sizing a digester, and their optimum values are specific to each substrate as well as the digester temperature [17]. A temperature phased anaerobic digestion (TPAD) system has been shown to be a reliable and effective means of sludge stabilization that achieves bioconversion and methane production rates higher than existing mesophilic anaerobic systems [18].
Therefore, the current study was done to determine the optimum loading rate and to predict the maximum potential methane production during mesophilic and thermophilic AD with crop residues. The work was considered as a pilot scale test for various temperatures and loading amounts in a continuously operating phased anaerobic digestion system.
Seeding sludge was taken from an anaerobic digester in a local waste water treatment plant. Once collected, the seeding sludge was stored in a refrigerator at 4℃ for one week before its volatile solids (VS) content was analyzed. Then, it was preheated to 35℃ for 24 h, and inoculated with substrates. The VS concentration of the seed microorganisms was 0.05%.
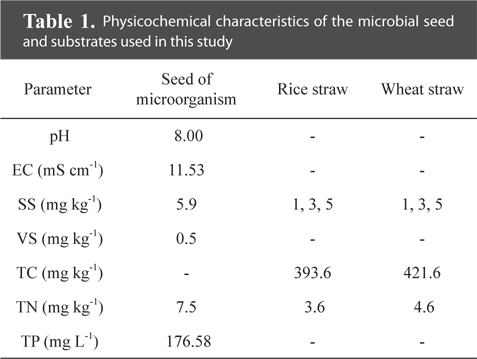
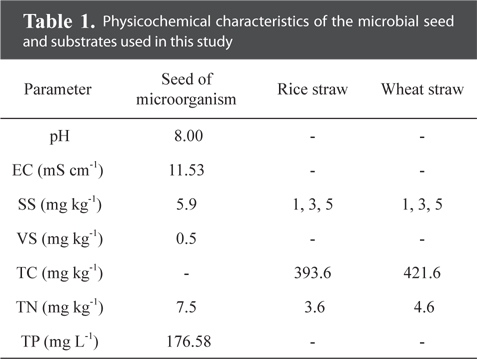
Rice and wheat straws were used as substrates in this study. The straws from the experimental field of the National Academy of Agriculture Sciences (NAAS) were ground with an electric blender and passed through a 2 mm sieve. Substrates were added to 200 mL of microbial seeding sludge to give final biomass loading rates of 1, 3 and 5% (w v−1). The physicochemical parameters, pH, total suspended solids (TSS), VS, total nitrogen (T-N), and total phosphorus (T-P), were determined according to Standard Methods [19]. The physicochemical characteristics of the substrate are presented in Table 1.
[Table 1.] Physicochemical characteristics of the microbial seed and substrates used in this study
Physicochemical characteristics of the microbial seed and substrates used in this study
The experimental design was a randomized factorial design with three replications. The main plots were the loading amounts of the crop residues: 1%, 3%, and 5% based on the dry weight. The sub-plots were the incubation temperatures: thermophilic (55℃) and mesophilic (35℃) AD. After placing the substrate and microbial seed sludge in the bottles, the headspace of each bottle was flushed with N2 gas for two minutes, degassed after three hours with a glass syringe, and sealed tight with a clamp. The bottles were then placed on a shaker at 40 rpm at digestion temperatures of 35℃ and 55℃.
The biogas production was periodically measured with a 20-200 mL glass syringe during the entire digestion periods [20]. At the same time, the concentration of methane in the produced gases was determined. The methane content in the biogas was measured with gas chromatography (GC; Varian CP3800, USA) and a thermal conductivity detector and a 1.0 m × 2 mm stainless steel packed column with N2 gas as a carrier. The temperatures of the detector and column were maintained at 189℃ and 40℃, respectively.
In general, biological phenomena are seldom clearly explained by building a mechanistic model. In this case, responding to a surface methodology, an empirical model or a statistical analysis can be formulated to elucidate the basic mechanisms underlying a complex system and thus provide better guidance for the process design and control [21]. In this study, the effects of the loading amounts and crop residues at each digestion temperature on methane production of an anaerobic digester were analyzed with a modified Gompertz model [22] shown below eq (1):
, where
All the parameters in the above equation were estimated by performing non-linear regression analysis with a Newtonian algorithm to minimize the sum of the squared errors (SSE) between the experiment and estimation. How well these parameters fit was validated by SSE, coefficient of determination (R2), standard errors (SE), 95% confidence limits, t-values and F-values using SigmaPlot version 11.0 (Systat Software Inc., San Jose, CA, USA).
The modified model provided in eq (1) was applied to fit the cumulative methane production profiles. Additionally, how well these parameters fit was validated with the model R2 values. The cumulative methane production curves from the two crop residues and three loading rates at each incubation temperature were well described by eq (1). All the model R2 values were less than 0.01 (Tables 1 and 2), suggesting that the modified model is statistically significant. Although the hydrogen production curve was previously fitted to a modified Gompertz equation, which was used as a suitable model for describing the hydrogen production in a batch system [23-25], it was also observed that the Gompertz model was appropriate to predict methane production with crop residues at different loading amounts and digestion temperatures.
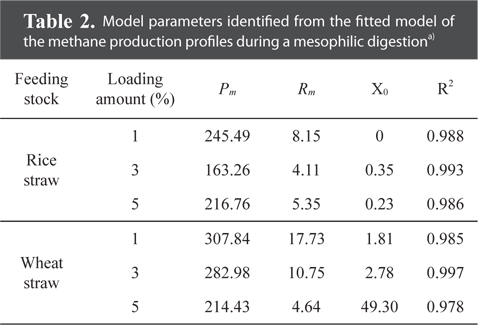
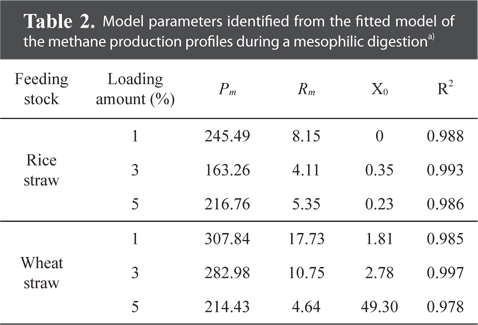
Model parameters identified from the fitted model of the methane production profiles during a mesophilic digestiona)
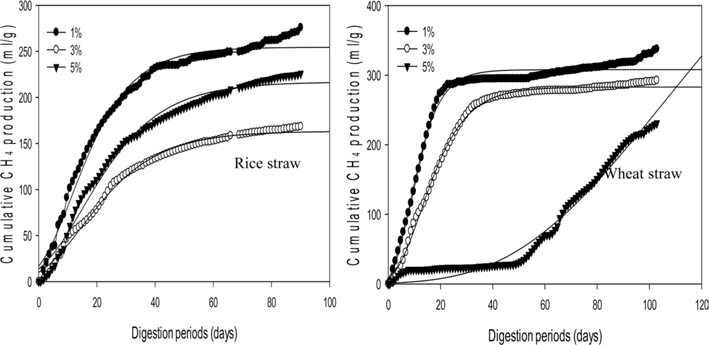
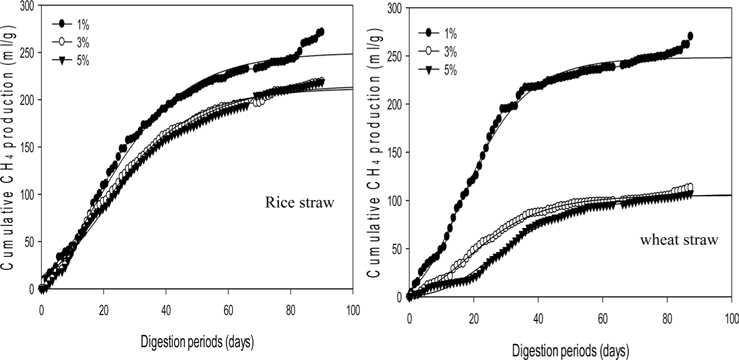
Fig. 1 shows that the cumulative methane production potential varied from 163.3 to 307.8 mL g−1 during a mesophilic AD and from 105.3 to 250.0 mL g−1 during a thermophilic AD with different applied loading amounts. These values were higher than the reported maximum values from co-digestion with cow manure and crop residues as grass, sugar beet top and straw: 268, 229, and 213 mL g−1 VS fed, respectively [26]. Moreover, these values are comparable to the methane yields of 160-260 mL g−1 VS for the batch degradation of wheat straw (values depend on the experimental conditions and particle size) presented in a review by Gunaseelan [27].
The maximum cumulative methane production potentials were 245.5 mL g−1 for a rice straw substrate and 307.8 mL g−1 for a wheat straw substrate during a mesophilic AD (Fig. 1). During a thermophilic AD, the results showed a potential of 250.0 mL g−1 of methane production for the rice straw substrate and 248.3 mL g−1 for the wheat straw substrate with a 1% loading amount (Fig. 2). The cumulative methane production from the reactor with only the wheat straw substrate was greater than that of the rice straw substrate at both digestion temperatures, but the loading rates for both crop residues did not have a consistent trend. The higher cumulative methane production from the reactor with the wheat residue could be attributable to the higher carbon content of the raw material (Table 1).
From the biogas production data, the current methane contents were 6.5% lower for the mesophilic AD and 17.9% for the thermophilic AD than the results reported by Lianhua et al. [28]. Wheat and rice straw substrates without pretreatment showed biogas production yields of 18.8 and 14.0 mL g−1 VS, respectively, with a 4.4% VS substrate concentration at 37℃, which was 1.7 and 1.2 times lower than the current results in dry unit weight [29]. Chandra et al. [30] claimed that the maximum biogas production yield occurred only during the initial 20 d of the digestion period for the rice straw substrate. However, the maximum biogas yield was observed within 10 d, and HRT lasted twice as long when compared with our result.
The activity of methane producing bacteria would be decreased if excess crop residues were provided (Table 2 and Fig. 1). The yield of methane production from the wheat straw substrate was greater than that from the rice straw substrate (Table 2), suggesting that the methanogenic activity might be affected by the carbon contents from the crop residues (Table 1). Ultimate methane production potentials during a mesophilic AD ranged from 163.3 to 245.5 mL g−1 for the rice straw substrate and from 214.4 to 307.8 mL g−1 for the wheat straw substrate (Table 2). The lag phase time for the rice straw substrate was shorter than that for the wheat straw substrate; however, the higher the loading rates were, the more prolonged the lag phase time was for the wheat straw substrate.
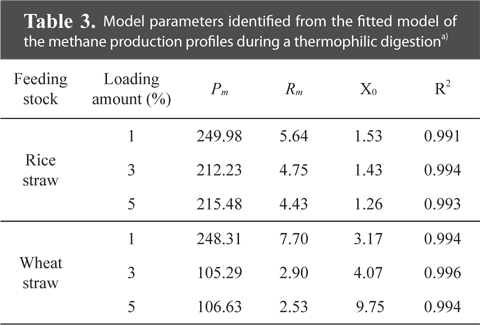
The highest ultimate methane production potential was 250.0 mL g−1 for the rice straw substrate and 248.3 mL g−1 for the wheat straw substrate during a thermophilic AD (Table 3). These values for the ultimate methane yield from wheat for both digestion temperatures were approximately similar to the values of 385.8 and 483.3 mL g−1 previously measured for the maximum biogas yields (based on the fresh weight), during a mesophilic and thermophilic AD, respectively [31]. Again, the lag phase time for the rice straw substrate was shorter than that for the wheat straw substrate with higher loading rates prolonging the lag phase time for the wheat straw substrate.
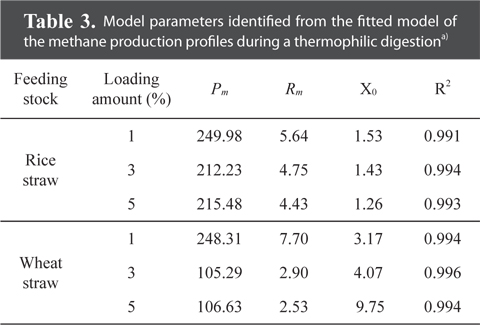
Model parameters identified from the fitted model of the methane production profiles during a thermophilic digestiona)
Overall, the study results strongly indicate that a 1% biomass loading rate for both crops and temperatures could be used as the optimum loading amount and feeding stocks. Through the kinetic model for the surface methodology, methane production, fitted to a modified Gompertz equation, showed ultimate methane potentials of 245.49 mL g−1 for the rice straw substrate and 307.8 mL g−1 for the wheat straw substrate during a mesophilic AD. During a thermophilic AD, the ultimate methane production potentials were 250.0 mL g−1 for the rice straw substrate and 248.3 mL g−1 for the wheat straw substrate with a 1% loading amount.
Overall, these research results strongly indicate that a 1% biomass loading amount for both crops and temperatures could be used as the optimum loading rate. For further study, these experimental results could be applied to the continuous operation of the pilot scale TPAD system with crop residues.