Diatom classification can be made according to two groups based on morphology (Schütt 1896, Simonsen 1979), though recent insight on diatom classification has been debated (Williams and Kociolek 2011). Most planktonic diatoms in the marine environment consist of morphologically centric diatoms, which are classical concept following Simonsen (1979), such as
A master plan entitled “The Project on Survey and Excavation of Korean Indigenous Species” at the National Institute of Biological Resources (NIBR), under the Ministry of Environment of Korea, was established in 2006. We conducted the survey and excavation of diatoms in brackish and marine waters of Korea from 2006 to the present. Lee et al. (2012) investigated the newly recorded diatoms in Korea in the first conclusive study of the species, reporting 60 species in 39 genera with illustrations based on light microscopy and scanning electron microscopy. This is the second serial study for newly recorded pennate diatoms in Korea during the investigation of “Survey and Excavation of Korean Indigenous Species”. Twenty species of diatom are for the first time documented in this study.
The specimens were collected from 46 sites from December 2008 to November 2012 from brackish and coastal waters of Korea (Table 1). All samples were collected by using 20-μm mesh-sized phytoplankton nets with vertical and/or horizontal towing, and the collected net samples were immediately fixed with 4% neutralized formalin. To observe the fine structures and characteristics of each diatom, we removed the cell organelles and organic matter using concentrated HCl and saturated KMnO4 (Hasle and Fryxell 1970). Some acid-cleaned materials were affixed to permanent slides that were mounted in Pleurax. The permanent slides were observed by means of light microscopy (LM): Axioskop 40 (Zeiss, Jena, Germany) with an AxioCam MRc 5 (Zeiss) and Eclipse 80i (Nikon, Tokyo, Japan) with and DS-Fi1 (Nikon) digital camera. The remaining cleaned materials were mounted on the aluminum stubs and coated with gold-palladium to examine the fine structures using scanning electron microscopy (SEM) (JSM-5600LV; Jeol, Tokyo, Japan). The dimensions of diatoms were measured from micrographs using ImageJ ver. 1.32 software. Diatom identifications were mainly based on the studies of Hendey (1964), Round et al. (1990), Hasle and Syvertsen (1996), Krammer and Lange-Bertalot (1997, 1999, 2004) and a number of references, including the original description for each taxon.
[Table 1.] Sampling information in Korean coastal waters
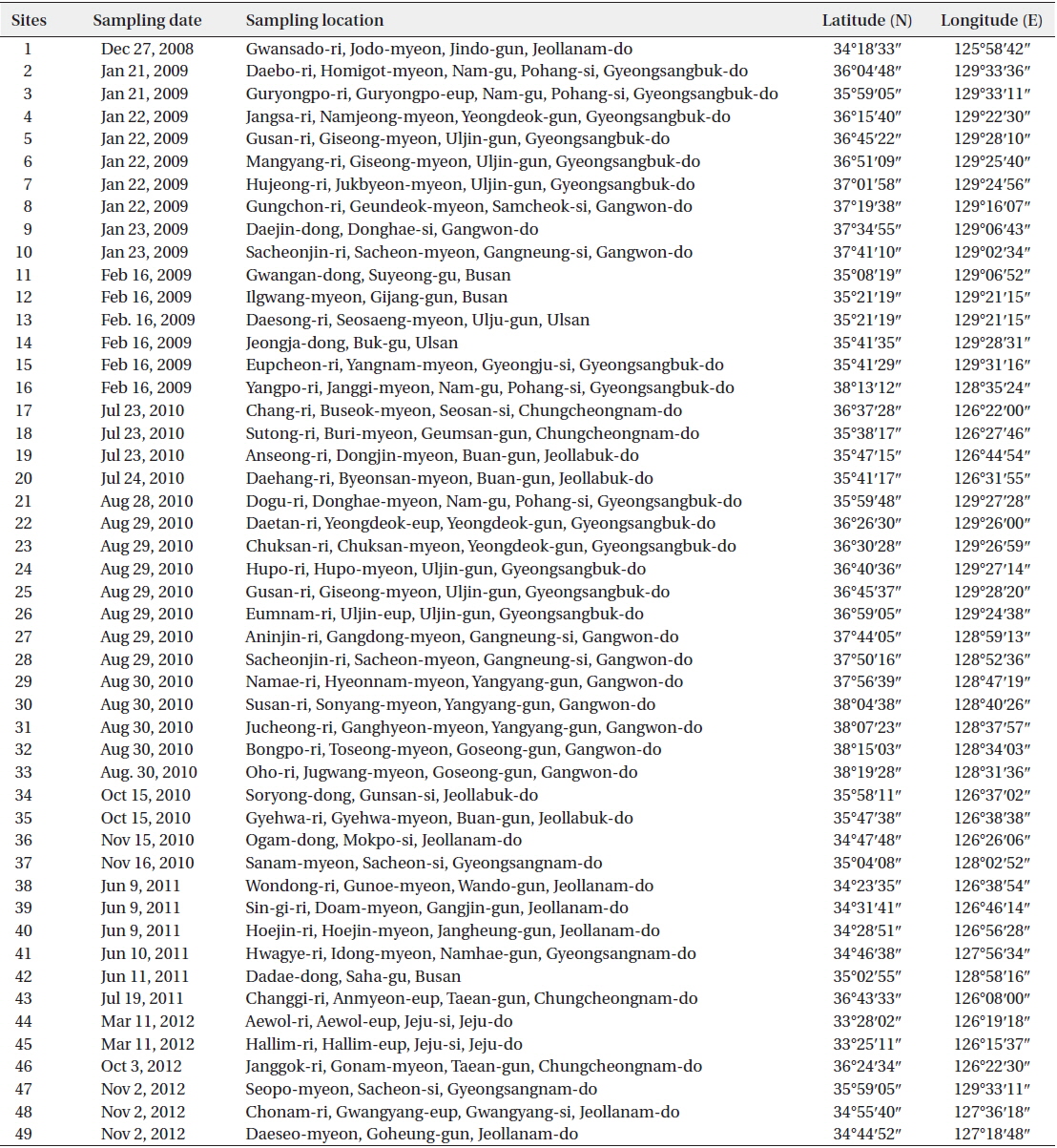
Sampling information in Korean coastal waters
>
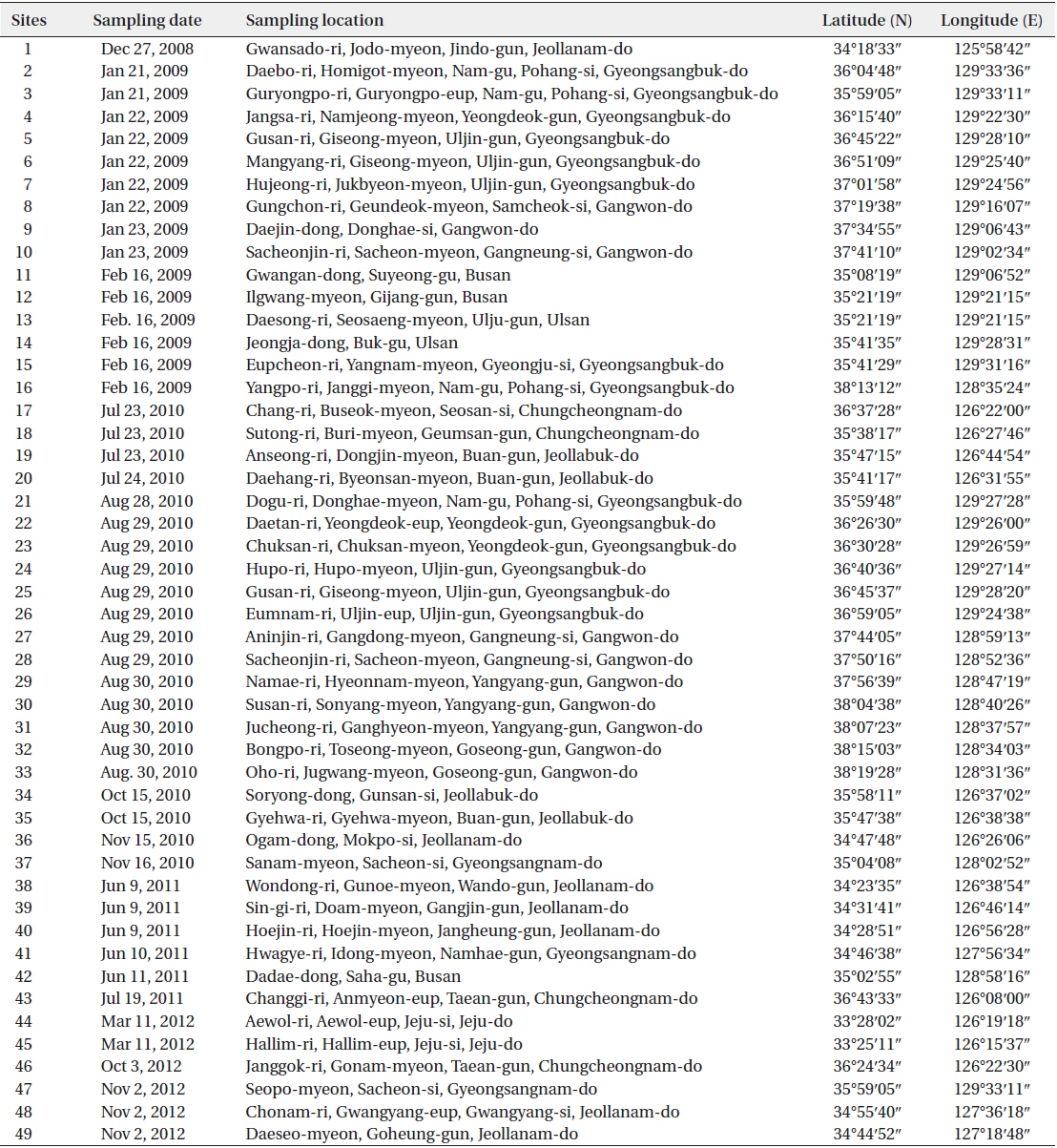
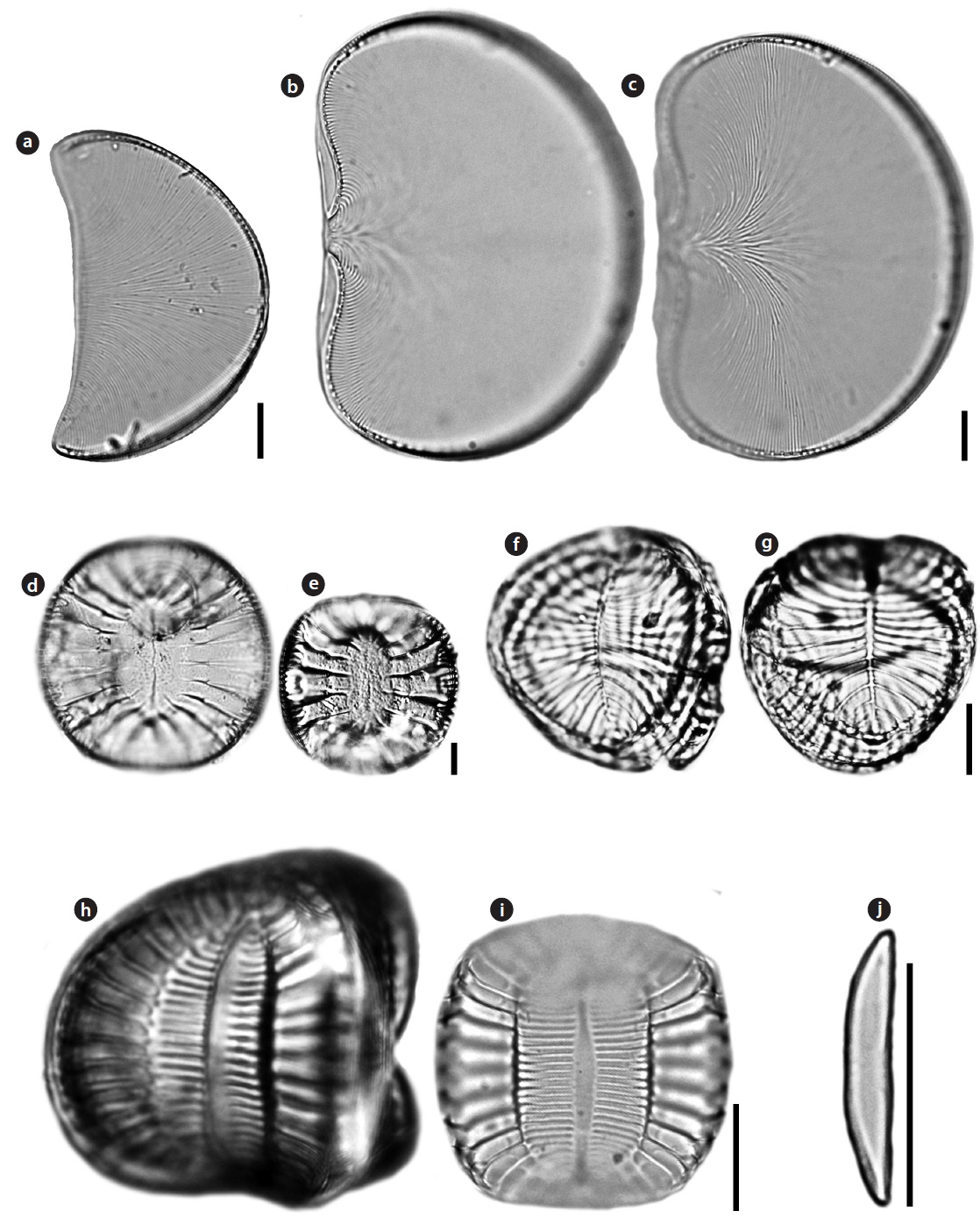
Order Achnanthales Silva Family Cocconeidaceae Kutzing
Cocconeiopsis wrightii (O’Meara) Witkowski 2000 (
Basionym:
Synonyms:
References: Riaux-Gobin and Compère 2004, p. 65, pl. III, figs 16–22, pl. IV, figs. d, e.
Specimen examined: NIBRDI0000130085.
Valves elliptical to almost rounded and slightly convex, length 12.8−22 μm, width 8.5−16 μm. Striae radiate, composed of rounded or slightly elongated areolae, 8−11 in 10 μm. One or two rows of areolae smaller near the margin. Raphe straight. End nodules rounded and distant from the valve apices, central endings pear-form and distant, central area transapically expanded and connected to an H-shaped area.
Korean distribution:
[Table 2.] Check list of new recorded diatoms in this study

Check list of new recorded diatoms in this study
>
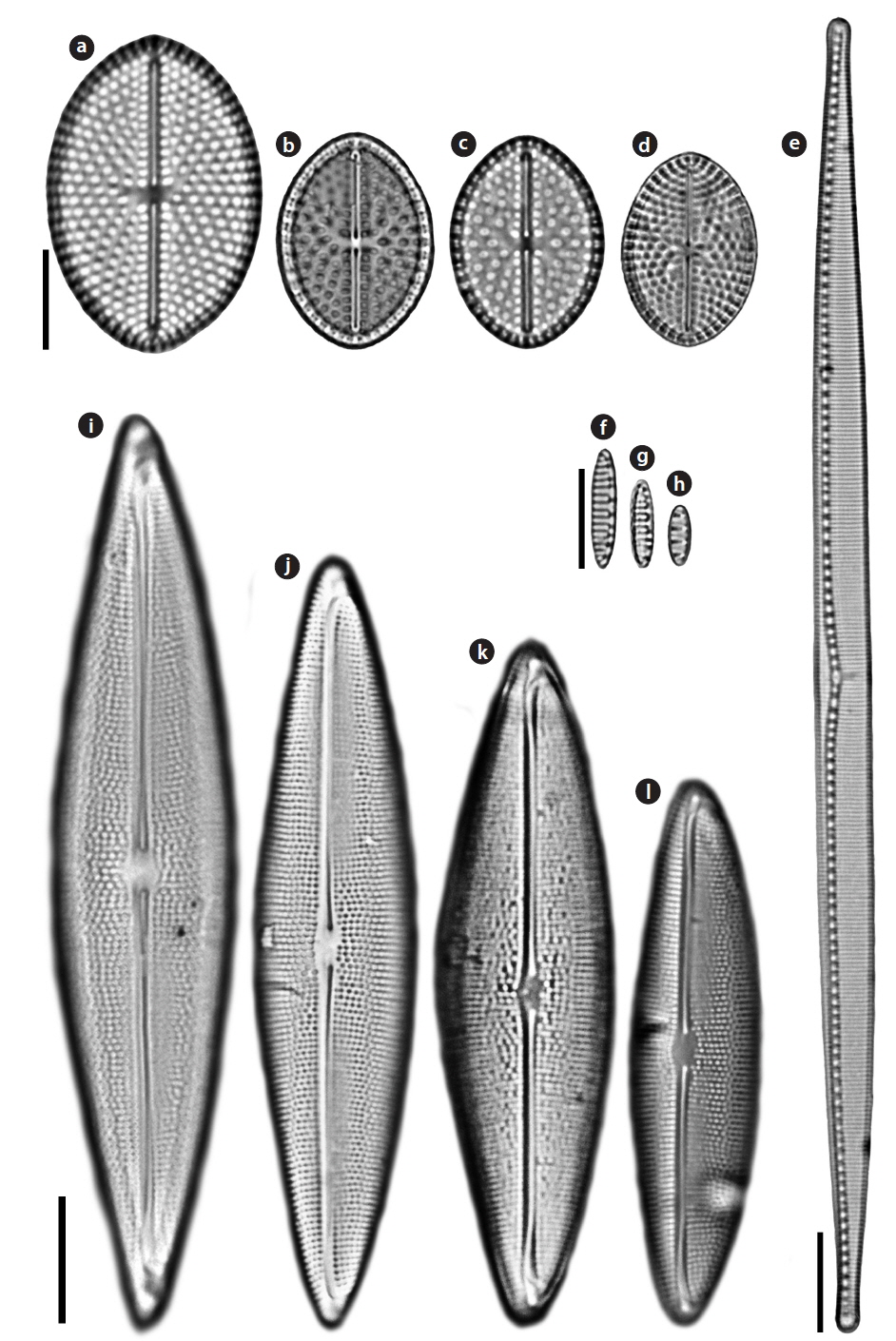
Order Bacillariales Hendey Family Bacillariaceae Ehrenberg
Nitzschia improvisa Simonsen 1960 (
References: Lange-Bertalot and Krammer 1987, p. 44, 60; Krammer and Lange-Bertalot 1997, pl. 2l.
Specimen examined: NIBRDI0000130087.
Valves linear with capitate pole. Length 117.5 μm, width 6.25 μm. Striae very clearly visible in LM and areolae resolvable, 24−25 in 10 μm. Raphe system fibulate, marginal. Fibulae square, regularly spaced, 8−10 in 10 μm. Raphe straight with distinct constriction in central area.
Korean distribution:
Nitzschia valdestriata Aleem & Hustedt 1951 (
References: Krammer and Lange-Bertalot 1997, p. 121, pl. 84, figs. 9-12.
Specimen examined: NIBRDI0000130092.
Valves bilaterally symmetrical, elliptical to linear-elliptical, with bluntly rounded poles. Length 6.1−12.1 μm, width 2.4−2.8 μm. Striae very clearly visible in LM, 15 in 10 μm, but areolae unresolvable. Raphe system fibulate, marginal. Fibulae square, regularly spaced. Central raphe endings absent. Raphes of the two valves lie at opposite sides.
Korean distribution:
>
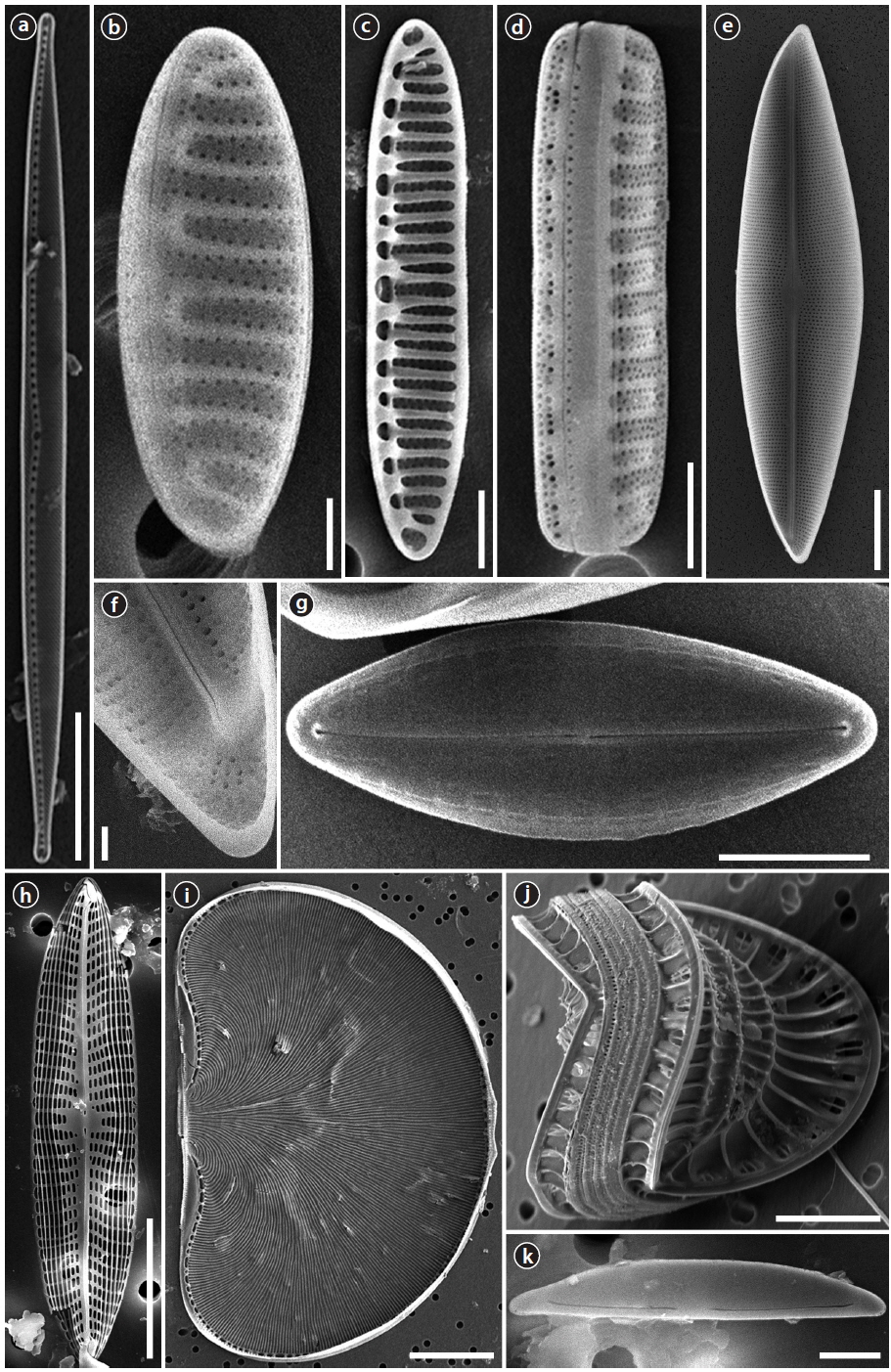
Order Naviculaes Bessey Family Berkeleyaceae D.G. Mann
Parlibellus hamulifer (Grunow)
Basionym:
References: Hustedt 1961, p. 311, fig. 1430; Cox 1988, p. 24.
Specimen examined: NIBRDI0000130093.
Valves rhombic-lanceolate with sub-acute apices. Length 44.5–60.7 μm, width 11.2–15.7 μm. Striae poroidal, 19–21 in 10 μm, more or less parallel and throughout the length of the valve. Raphe straight and slightly curved in central area, extending almost to the apices, deflected to one side at the poles helictoglossae.
Korean distribution:
Parlibellus rhombicus (Gregory)
Basionym:
Synonyms:
References: Cox 1988, p. 25, figs 17, 33-38.
Specimen examined: NIBRDI0000130094.
Valves rhombic-lanceolate with acute apices. Length 34.6–48.0 μm, width 8.9–10.8 μm. Striae poroidal, 26–29 in 10 μm, radial in valve centre. Raphe straight and slightly curved in central area, extending almost to the apices, deflected to one side at the poles helictoglossae.
Korean distribution:
Navicula diabolica Geissler & Gerloff 1963 (
References: Krammer and Lange-Bertalot 1999, p. 210, pl. 78, fig. 28.
Specimen examined: NIBRDI0000130095.
Valves linear-lanceolate to rhombic with bluntly rounded ends. Length 5–6 μm, width 2–2.5 μm. Raphe filiform, axial congruent with the media rib, very narrow, linear, central area and other structures in the LM not accurately differentiable. Striae parallel, 35 in 10 μm, Areolae fully developed, distributed only in the margin. Raphe straight, terminal end with a small helictoglossa internally.
Korean distribution:
Navicula pavillardii Hustedt 1939: 635 (
References: Hendey 1964, p. 204; Krammer and Lange-Bertalot 1999, p. 118, pl. 39, figs 4, 5.
Specimen examined: NIBRDI0000130096.
Valves linear to lanceolate sometimes in the middle concave edges and wedge-shaped, lean, fairly acute to obtuse rounded ends. Length 36.9−57.7 μm, width 7.7–8.2 μm. Striae continuous radially around 12–14 in 10 μm, areolae fairly coarse to 28 in 10 μm. Raphe straight. Central raphe ending slightly expanded. Polar raphe fissure hooked to one side.
Korean distribution:
>
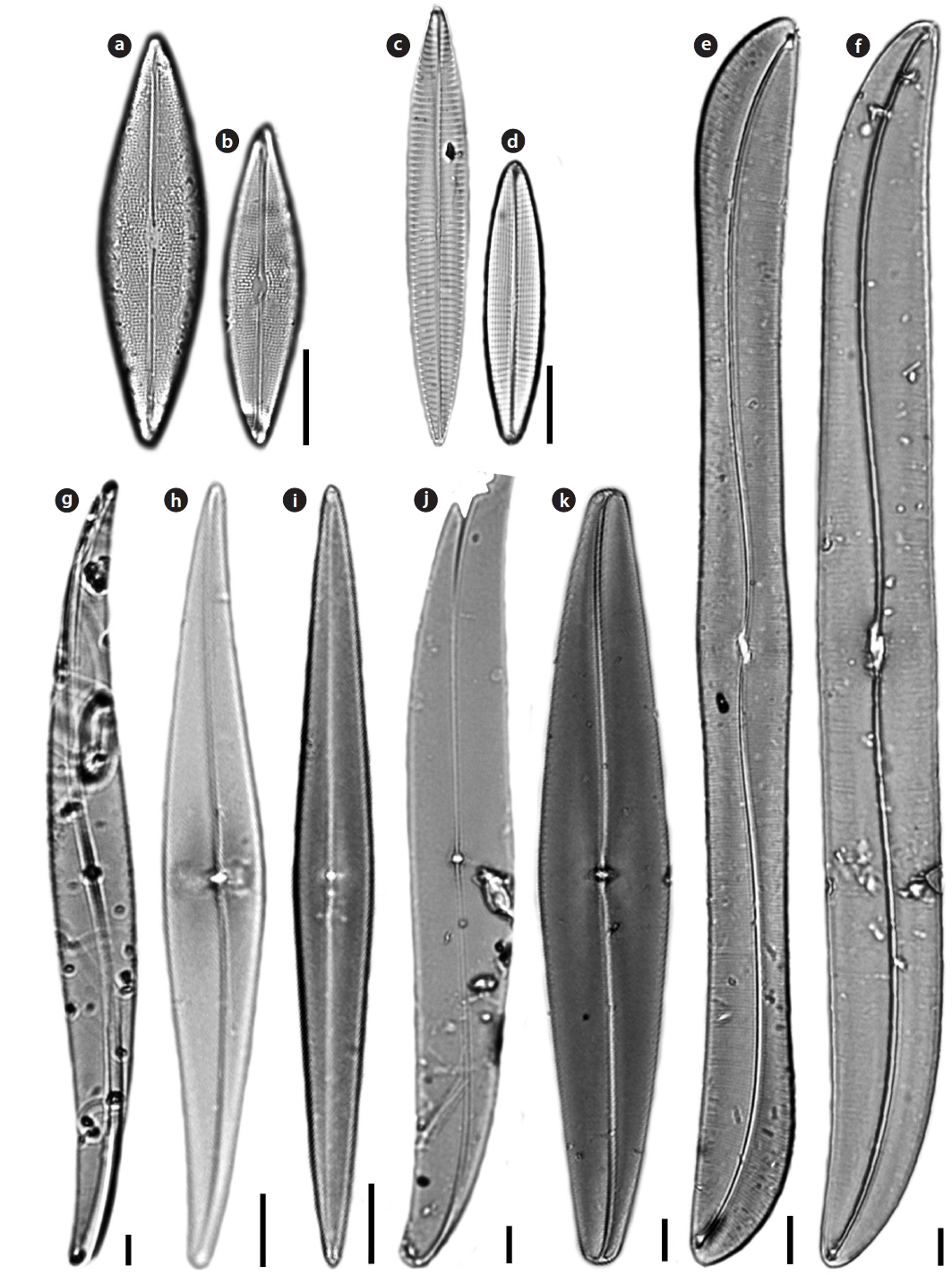
Family Pleurosigmataceae Mereschowsky
Gyrosigma subtile (Brebisson) Reid 2003 (
Basionym:
Synonym:
References: Kützing 1849, p. 87; Pritchard 1861, p.917; Kuntze 1891, p. 919; Reid and Williams 2003, p. 139, figs 57–61.
Valve vaulted, linear, sigmoid, tapering to obtuse apices. Length 335–400 μm, width 30–40 μm. Transverse striae 16–26 in 10 μm, longitudinal striae 14–16 in 10 μm. Raphe central, with double curvature. Central area transapically offset, with flattened central bars. Central hyaline area large. A row of pores, arranged in an arc, occurs above the helictoglossa.
Korean distribution:
Gyrosigma turgida (Stidolph)
Basionym:
Synonym:
References: Stidolph 1980, p. 385; John 1983, p. 115, pl. 48, figs. 1–3; Stidolph 1988, p. 380, figs. 6-10.
Specimen examined: NIBRDI0000130097.
Valves linear, sigmoid, oblong, gently inflated at the center. Length 148–256 μm, width 18-19 μm. Transverse striae 20–25 in 10 μm, longitudinal striae 19–20 in 10 μm. Raphe undulate, eccentric. Central area transapically offset, with smooth flattened central bars. No hyaline area at the apex. A series of minute pores surrounds the apex of the valve beyond the terminal raphe fissures and the helictoglossa.
Korean distribution:
Pleurosigma decorum
Synonym:
References: Smith 1853, 63, pl. 21, fig. 196; Griffith and Henfrey 1856, p. 302; pl. 11, fig. 26; Kuntze 1891, p. 919.
Specimen examined: NIBRDI0000130098.
Valves lanceolate, sigmoid, with sides between the middle and the end one convex and the opposite barely concave, vaulted and with sub-acute apices. Length 200–310 µm, width 20-28 µm. Transverse striae 18–20 in 10 µm, longitudinal striae 15–18 in 10 µm. Central area conspicuous, saddle-shaped, internally short and raised, with central bars surrounding the central nodule. Terminal endings finishing in helictoglossa turned by a siliceous ridge.
Korean distribution:
Pleurosigma inflatum
Synonym:
References: Shadbolt 1854, p. 16, pl. 1, fig. 9; Brébisson 1854, p. 255, pl. 1, fig. 7; Smith 1856, p. 96.
Specimen examined: NIBRDI0000130099.
Valve only slightly sigmoid, broadly lanceolate, with acutely rounded and slightly rostrate apices. At valve center slightly vaulted, towards the apices more strongly vaulted. Length 148−150 µm, width 16−26 µm. Transverse striae 22−30 in 10 µm, longitudinal striae 22−30 in 10 µm. Central area slightly convex, with thin central bars. Central hyaline area large.
Korean distribution:
Pleurosigma patagonicum (Ferrario & Sar) Sterrenburg & Sar 2012 (
Synonym:
References: Ferrario and Sar 1990, p. 201, figs. 1–6; Sar et al. 2012, p. 248, figs. 59–76.
Specimen examined: NIBRDI0000130102.
Valves narrowly lanceolate to sub-rhomboidal, straight, tapering gradually towards the acute apices, slightly vaulted near the ends. Length 98-152 µm, width 10−17 µm. Transverse striae 20−25 in 10 µm, longitudinal striae 15−16 in 10 µm. Central area very small, orbicular, symmetric, funnel-shaped. Internal apices with one or two isolated pores in apical position and two rows of slits placed laterally at the last section of raphe-sternum towards the helictoglossa. No central hyaline area.
Korean distribution:
Pleurosigma speciosum
Synonym:
References: Smith 1852, p. 6, pl. 1, fig. 3; Griffith and Henfrey 1856, p. 302, pl. 11, fig. 28; Kuntze 1891, p. 919.
Specimen examined: NIBRDI0000130103.
Valves linear and slightly attenuated at the apices, which are obtuse. Length 268−270 µm, width 28−33 µm. Transverse striae 16−17 in 10 µm, longitudinal striae 10−11 in 10 µm. Central bars smooth and slender and of approximately equal length. Central hyaline area adjacent to the central bars.
Korean distribution:
Pleurosigma williamsii
References: Reid 2002, p. 80, figs. 9-17.
Specimen examined: NIBRDI0000130104.
Description: Valves lanceolate, barely sigmoid, gently curving to obtusely rounded ends. Length 136−192 µm, width 22−34 µm. Transverse striae 18−20 in 10 µm, longitudinal striae 1213 in 10 µm. Central bars smooth and of approximately equal length. No central hyaline area next to the central bars. Extra siliceous thickening at the apices and valve margin, giving a hyaline area around the edge of the valve.
Korean distribution:
>
Order Surirellales D.G. Mann Family Auriculaceae Hendey
Auricula complexa (Gregory) Cleve 1894 (
Basionym:
References: Gomes et al. 1989, p. 6, fig. 1; Ruck and Kociolek 2004, pl. 2, fig. 2; Teanpisut and Patarajinda 2007, p. 122, pl. 2, fig. 31.
Specimen examined: NIBRDI0000130105.
Valve with ear-shaped outline. Length 60.0 μm, width 35.6 μm. Striae flabellate, radiating from ventral margin to dorsal raphe canal, 16−18 in 10 μm. Costae frequently bifurcated. Raphe canal is eccentric, present on slightly raised keel along the dorsal margin. Mantle well-developed on dorsal side only, becoming almost absent at the ventral margin.
Korean distribution:
Auricula flabelliformis M.Voigt 1960 (
References: Ruck and Kociolek 2004, pl. 2, fig. 5, pl. 6, fig. 3.
Specimen examined: NIBRDI0000130106.
Valve with shellfish-shaped outline. Length 82−115.7 μm, width 56.7−75.7 μm. Striae flabellate, radiating from center of the ventral margin to dorsal raphe canal, 16−18 in 10 μm. Costae frequently bifurcated. Raphe canal surrounded valve margin, present on slightly raised keel along entire valve.
Korean distribution:
>
Family Surirellaceae Kutzing
Campylodiscus ambiguus Greville 1860 (
References: Schmidt 1875, pl. 18, figs. 23–26; Ruck and Kociolek 2004, p. 163, pl. 36, figs. 1–7, p. 165, pl. 37, figs. 8–12, p. 167, pl. 38, figs. 13−18.
Specimen examined: NIBRDI0000130107.
Valve circular to sub-circular, saddle-like, with barely noticeable ribs, 45−107 µm in diameter. A circular and shallow navel in the middle of the valve depression. Central area marked by two ridges that expand the length of the axis. The central ridges and the area between them are hyaline without any striations.
Korean distribution:
Campylodiscus decorus
References: Schmidt 1875, pl. 14, figs. 4–5; Stidolph 1980, p. 403, pl. 14, fig. 10; Lobban et al. 2012, p. 469, pl. 64, figs. 5–6.
Specimen examined: NIBRDI0000130108.
Valve circular to sub-circular, saddle-like, with barely noticeable ribs, 28 µm in diameter. Central area narrow and marginal ribs expand toward the center. Infundibula absent, single tubes connect the interior cell to the canal raphe. Fenestral openings have one or two fenestral bars each. Narrow ribs extending from keel to very narrow lanceolate. The pores in this species are resolvable in LM.
Korean distribution:
Campylodiscus samoensis Grunow in
References: Schmidt 1875, pl. 15, figs. 19–20; Schmidt 1897, pl. 207, fig. 14; Stidolph et al. 2012, pl. 21, fig. 16.
Specimen examined: NIBRDI0000130109.
Valve circular to sub-circular, saddle-like, with barely noticeable ribs. Infundibula visible around the margin of the valve. Central area marked by two ridges that extend the length of the apical axis. The central ridges and the area between them are transapically striated.
Korean distribution:
>
Order Thalassiophysales D.G. Mann Family Catenulaceae Mereschkowsky
Catenula adhaerens (Mereschkowsky) Mereschkowsky 1903 (
Basionym:
References: Sundbäck and Medlin 1986, p. 284, figs. 1–23.
Specimen examined: NIBRDI0000130110.
Valves bilaterally symmetrical, elliptical to linear-elliptical, with rounded apices. Apices slightly deflected to the ventral part. Length 7−21 μm, width 1.5−3.5 μm. Striae very fine, unresolvable. Raphe close to the ventral part, slightly deflected toward the dorsal part. Central raphe endings slightly expanded. Terminal raphe endings distant from the apices.
Korean distribution:
The number of diatoms has been estimated approximately 100,000 species in over 1,000 genera (Mann 1999, Fourtanier and Kociolek 1999, 2003). Despite of the high species diversity in the world, there have been approximately 1,000 diatom species in Korea (Lee et al. 1995, Lee 1995, Lee et al. 2012, Joh 2013). The diversity of diatoms in Korea is relatively low compared to the world record. In the present study, the new recorded pennate diatoms imply several reasons why the taxa have not been recorded in Korea. First, some genera such as
Second, some species is probably confused to the morphologically similar taxa.
Third, several species only occurred from specific habitat. Most pennate diatoms have been mainly reported to live in diverse substrates such as plants, macroalgae, hard rock, and sediments (Round et al. 1990, Joh 2013).