The silkworm,
The anti-diabetes mechanism of silkworm powder and its extracts was found to effectively inhibit α-glucosidase in the human small intestine. Related to this factor, the major functional component of silkworm powder is 1-deoxynojirimycin (1-DNJ), an intestinal α-glucosidase inhibitor present in mulberry leaves (Asano, 2003) and sericulture products such as silkworm powder (Asano
According to previous data, the concentration of 1-DNJ in most commercial mulberry and sericulture products is known to be as low as approximately 0.2% (Asano
In this study, we wanted to provide a basis for understanding 1-DNJ content in various silkworm species. Furthermore, our goal was to a database for silkworm genetic resources utilizing morphological and, genetic characteristics data.
Silkworm resources were collected from the Sericulture and Apiculture Division for the Department of Agricultural Biology, RDA in Suwon, Republic of Korea. Both spring and autumn reared silkworms 270 varieties were used for the DNJ (Japanese 90; Chinese 88; European 33; Tropicals 9; Korean 2; Commercial Varieties 8; and Miscellaneous species; 40). The freeze-dried 5th instar, 3-day-old silkworms (Ilshin Lab Co., Ltd) were ground and used within 48 h.
>
Sample preparation procedures
About 0.1 g of larva powder was mixed with 10 mL H2O, followed by vigorous shaking for 30 min, and then treatment with sonication for 1 h. After extraction with 60oC for 1 h, the sample was centrifuged at 4,000 rpm for 20 min, and then the supernatant was collected. The pellet was treated again by repeating the above steps, and then supernatants were combined and diluted to 100 mL by adding distilled water. This crude DNJ extract was mixed with 10 μL 0.4 M borate buffer, and 100 μL FMOC-CI (9-fluorenylmethyl chloroformate); the mixture was reacted on 40oC for 1 h. Finally, 10 μL 0.1 M glycine was added to this sample, and then adjusted to 1 mL with 0.1% acetic acid.
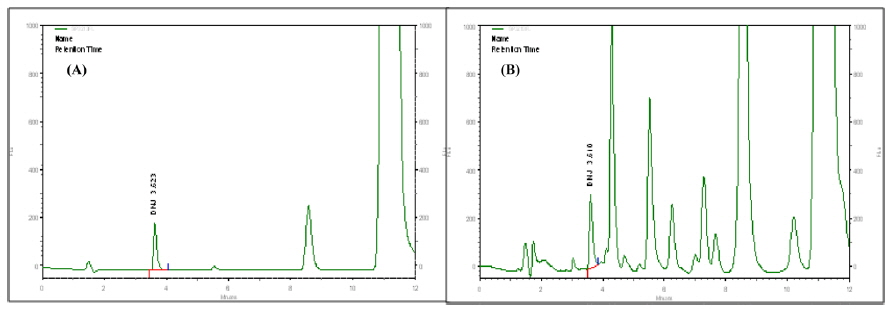
In order to measure DNJ within the analytes, high performance liquid chromatography(HPLC, SHISEIDO SP3203), fluorescence detector (Em 254, EX 322) and a C18 (100×4.6 mm, ID 3 μm) column were used. The absorbance of the effluent was monitored at 254 nm and flow rate was 1 mL/min. The mobile phase consisted of solvent A (acetonitrile) and solvent B (0.1% aqueous acetic acid). The initial solvent condition was solvent A: solvent B (20:80, v/v) for 16 min. After each analysis, a gradient was used on solvent A: solvent B (40:60, v/v) for 16 min, A:B (80:20, v/v) for 16.1, A:B (80:20, v/v) for 20 min, and A:B (20:80, v/v) for 20.1 min. The standard material was 1-deoxynojirimycin hydrochloride (SIGMA, 10 mg) and the DNJ content was calculated (Fig. 1.).
>
DNJ content of 270 silkworm varieties
There were many reports on DNJ in
Using data acquired from this study, we built a database of silkworm and mulberry resources with morphological and, genetic characteristics data, moreover, certain bioactive compounds have been receiving increasing attention. We analyzed DNJ content in addition to the base genetic information using 270 varieties of silkworms. All silkworms were the freeze-dried 5th instar species that were 3 days old.
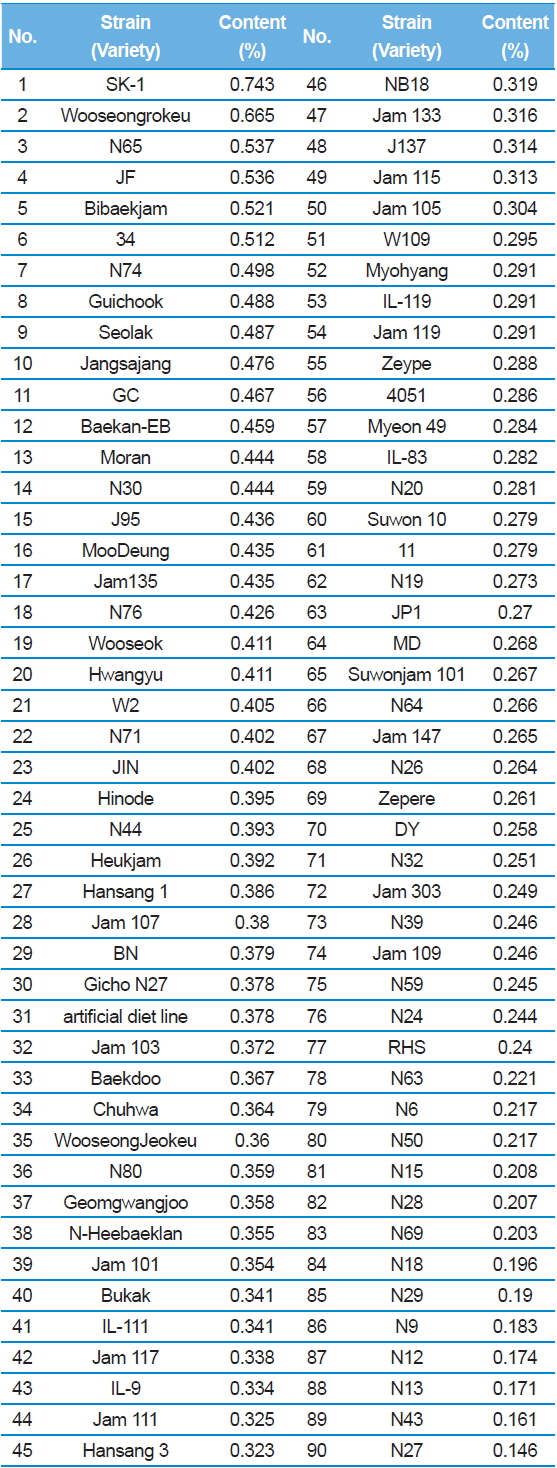
Results showed that, SK-1 had the highest DNJ content with 0.743% (Table 1). In contrast, the N27 had the lowest DNJ content on 0.146%. Yatsunami
[Table 1.] 1-DNJ contents for Japanese silkworm varieties
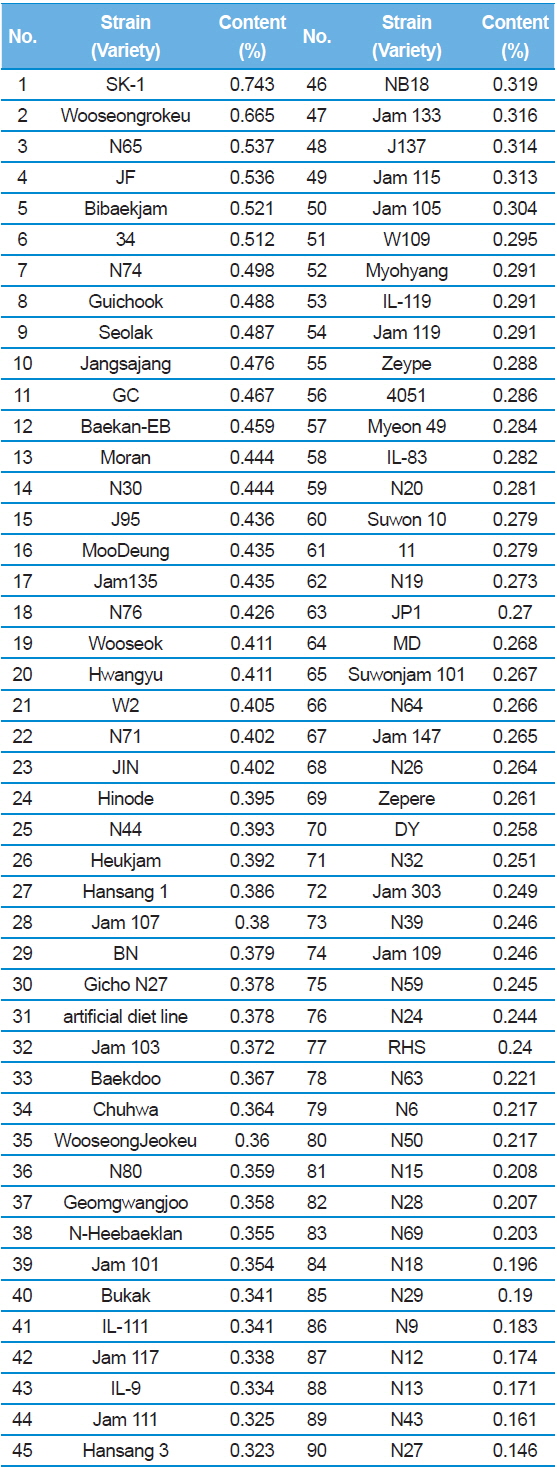
1-DNJ contents for Japanese silkworm varieties
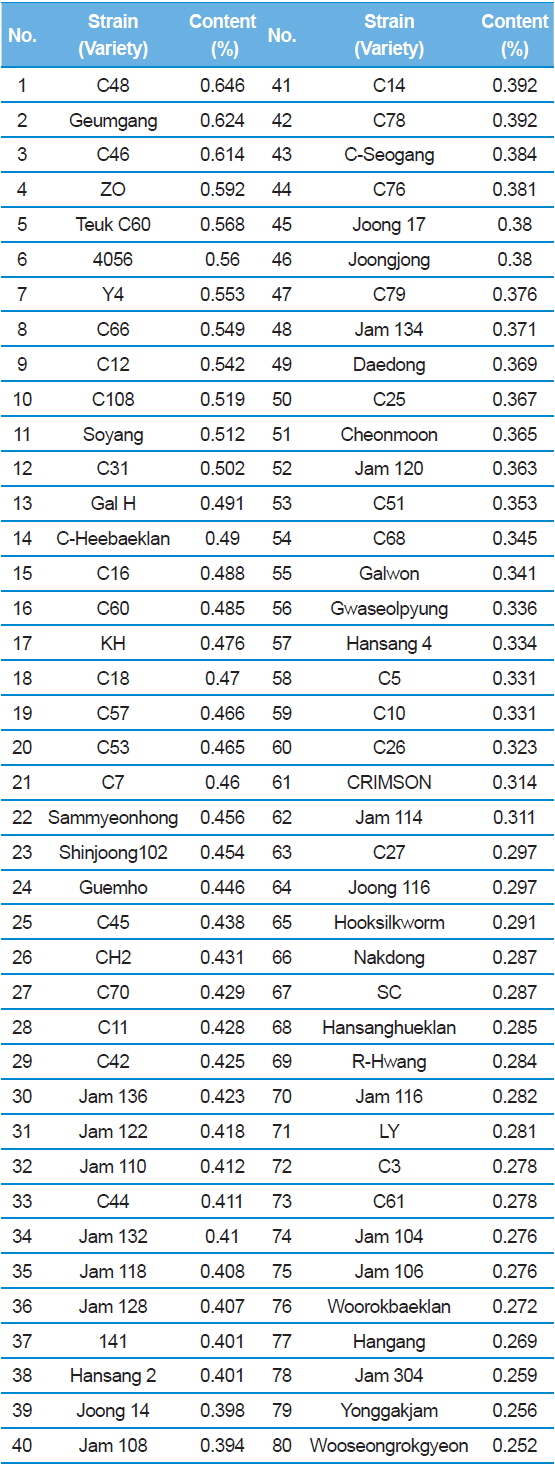
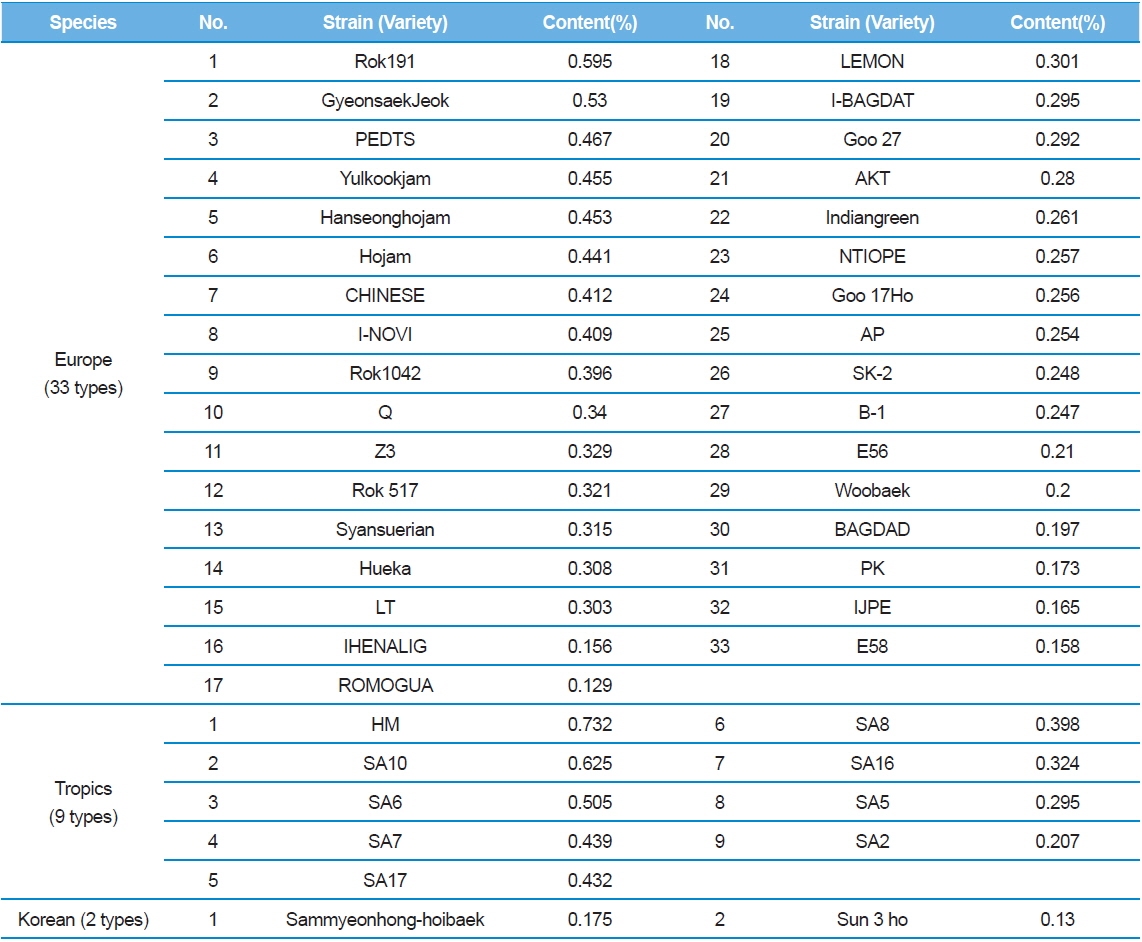
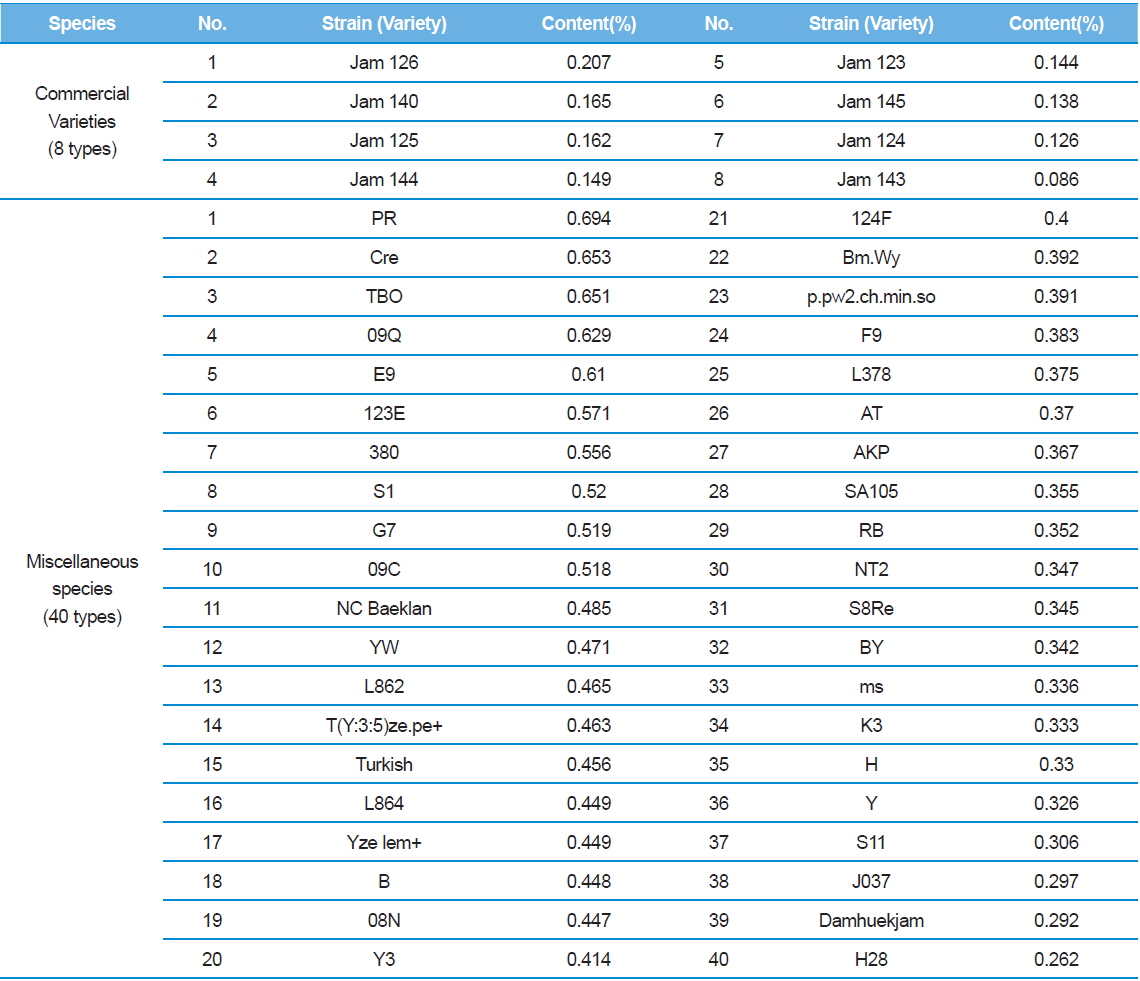
In Table 2, C48 had the highest DNJ content (0.646%) while Shinjoong 103 had the lowest of all Chinese species with 0.159%. Of the European breeds, Rok 191 had the highest DNJ content (Table 3). In addition, there was no discernable difference in DNJ content with regard to blood color, cocoon color, or egg color of the silkworm. DNJ content of the Korean breeds was relatively lower than the other varieties (Table 3). On the other hand, the highest Tropics varieties were HM (0.732%) and PR (0.694%) within the miscellaneous speicies (Table 4). Ryu
[Table 2.] 1-DNJ contents for Chinese silkworm varieties
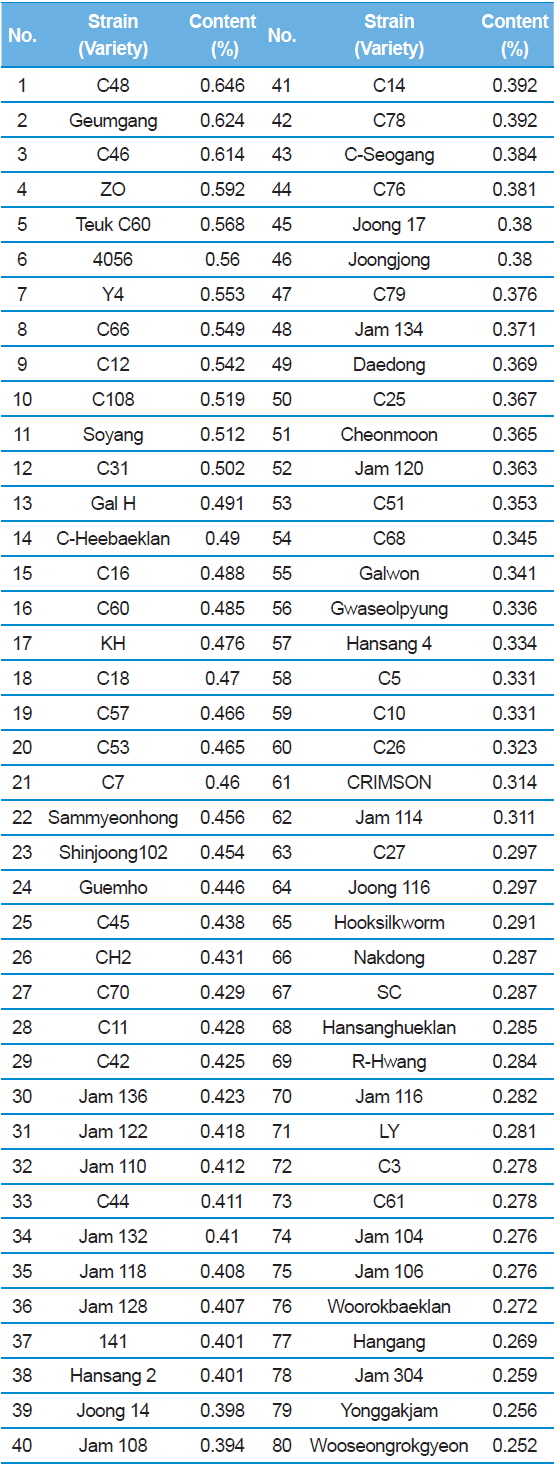
1-DNJ contents for Chinese silkworm varieties
[Table 3.] 1-DNJ contents for Europe, Tropics, Korean silkworm varieties

1-DNJ contents for Europe, Tropics, Korean silkworm varieties
[Table 4.] 1-DNJ contents for Practical application and ETC silkworm varieties
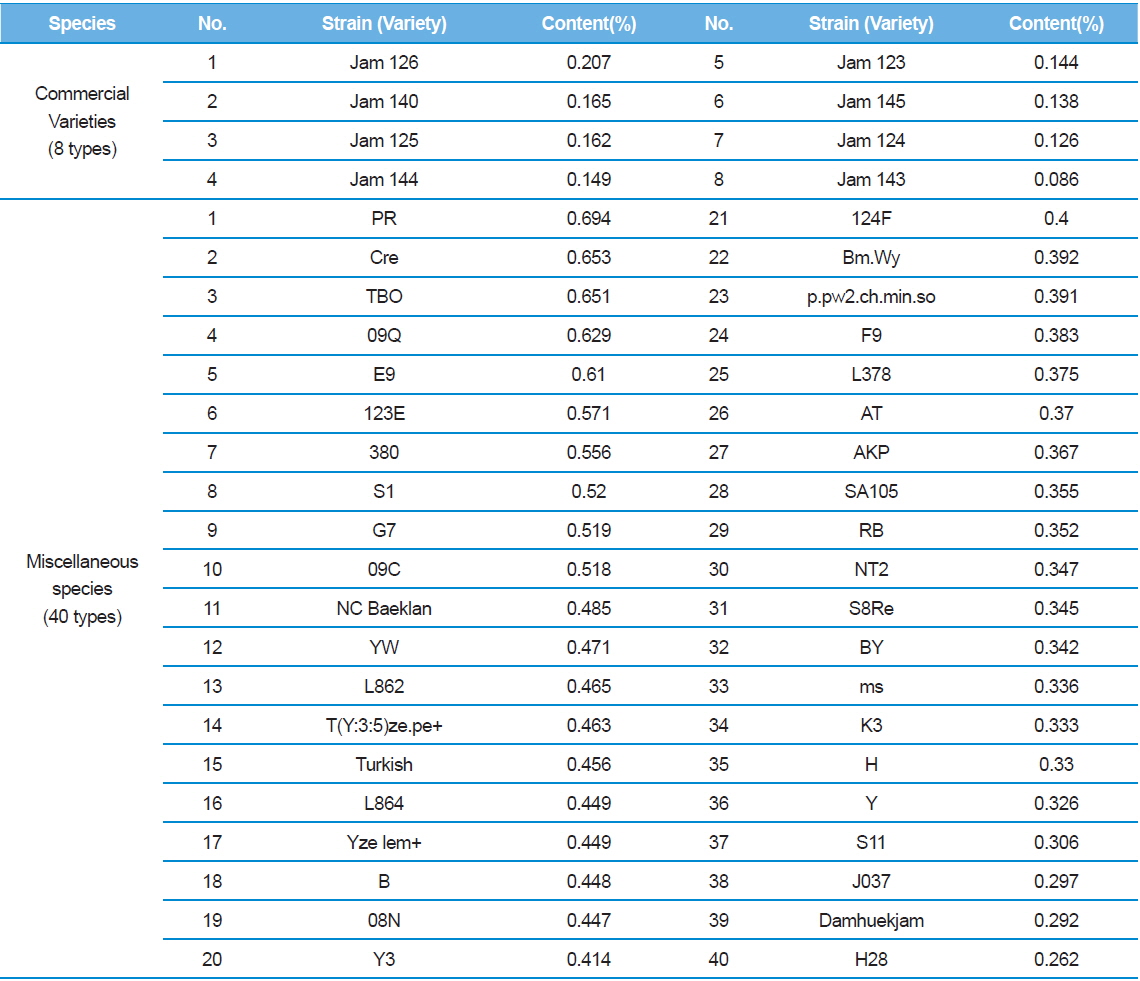
1-DNJ contents for Practical application and ETC silkworm varieties
>
DNJ content about larvae organs and between male and female silkworm larvae
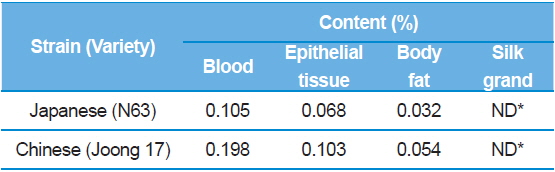
The results showed that the DNJ content in each of organs was the highest in the silkworm blood, then epithelial tissue, and finally body fat (Table 5). The distribution of DNJ in the larval body was investigated in all organs and, except for the silk glands, all organs and tissues contained DNJ. In addition, among the tissues, there was a significant increase in DNJ content in blood, followed by the intestinal juices and alimentary canals (Yin
[Table 5.] 1-DNJ contents for each of larval organs.
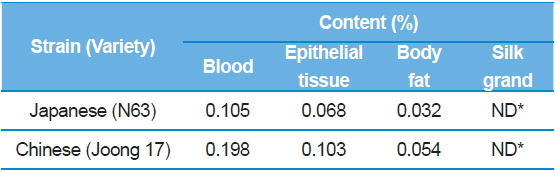
1-DNJ contents for each of larval organs.
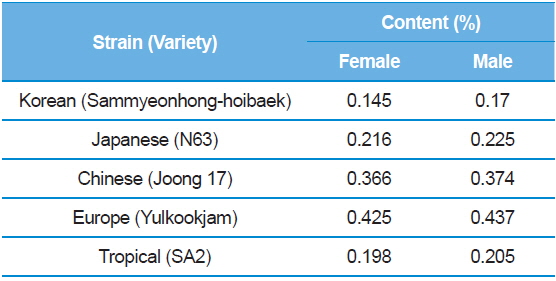
[Table 6.] 1-DNJ contents for female and male larvae.
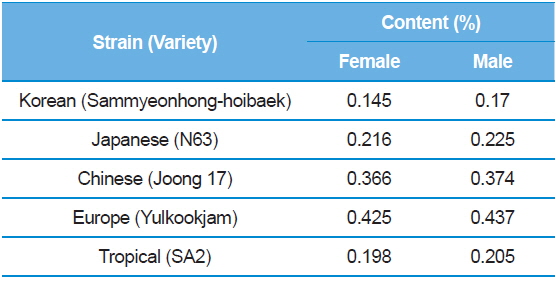
1-DNJ contents for female and male larvae.
>
DNJ accumulation among silkworm varieties
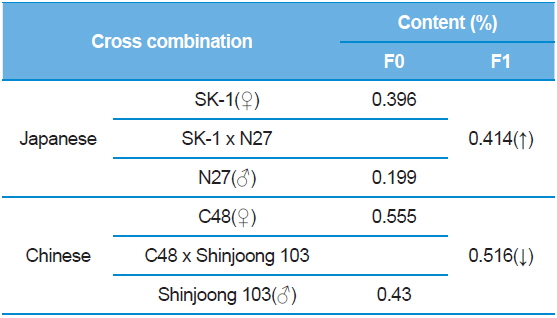
The purpose of combining tests was to optimize hybrid combinations that yielded higher DNJ contents than basic silkworm larvae. The DNJ contents among various silkworm varieties were investigated. Results indicated that there was a difference among various silkworm varieties. When the SK-1 and N27 were crossbreed, the DNJ content was a higher than the parent SK-1. In contrast, between C48 and Shinjoong 103 in Chinese breeds, the hybrid was a lower than Shinjoong 103 (Table 7). The variation difference of DNJ content by a crossbreeding test could be utilized in a silkworm breeding database.
[Table 7.] 1-DNJ contents among silkworm varieties.
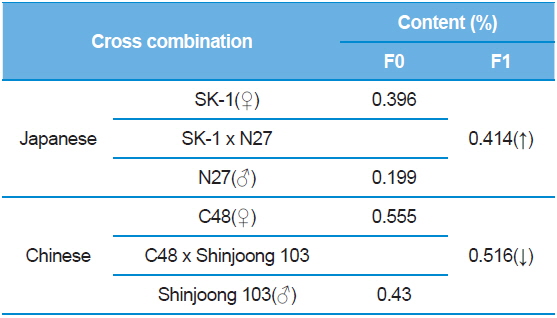
1-DNJ contents among silkworm varieties.