Bee venom pharmacopuncture (BVP) is a new type of treatment combining the efficacy of acupuncture and the pharmacological actions of the venom that is artificially extracted and refined from live honey bees (
Until now BVP has been administered intramuscularly, intracutaneously or into acupoints. However, in our opinion, intravenous injection should be possible if SBV is a safe medium. Thus, we conducted a single-dose toxicity test of SBV administered intravenously in Sprague-Dawley (SD) rats, and we report the results here.
Twenty-four SD rats of each gender were obtained from a specific pathogen-free facility (ORIENTBIO Inc., Gyeong- gi, Korea) at 5-weeks of age and were used after a week of quarantine and acclimatization. The animals were housed in a room maintained at 20.0 ─ 23.0℃ under a relative humidity of 42.8% ─ 68.9%. The room was illuminated with artificial lighting from 07:00 to 19:00 hours and 10 ─ 15 air changes per hour. Three animals were housed in suspended stainless-steel wire-mesh cages and were allowed sterilized tap water and commercial rodent chow (Teklad Certified Irradiated Global 18% Protein Rodent Diet 2918C, Harlan Laboratories, Inc., U.S.A.). The protocol of this study was approved by the institutional Animal Care Committee of Biotoxtech, Co. (Oh Chang, Korea).
SBV was prepared using a freeze dryer (FD 8508, Ilshin Lab Co. Ltd., Korea) by Ginseng & Venom (G & V) Co. (Wonju, Korea) and was stored at ─ 20℃. A high-performance liquid chromatography (HPLC, Agilent 1100 series, Agilent Technologies Ltd., U.S.A.) analysis was performed to confirm that SBV was pure melittin.
Twenty healthy male and 20 healthy female rats were selected by average weights, and 5 rats of each gender were assigned to 1 of 4 groups: control (normal saline), low dosage (0.1 mL/animal), medium dosage (0.5 mL/animal), and high dosage (1.0 mL/animal). In a pilot study, no mortalities had been observed at a dosage of 1.0 mL/animal of SBV in male and female SD rats; based on that results, we set the high dose at 1.0 mL/animal. SBV was administered to the rats by intravenous injection into the caudal vein. The control group was administered 1.0 mL/animal of normal saline (Lot No. 12115, Choongwae Pharma Corp., Korea).
All animals were observed for clinical signs at 30 minutes, 1 hour, 2 hours, 4 hours and 6 hours after the injection of SBV. Clinical signs were observed daily from the injection day to 14 day after the first injection. The body weight of each rat was measured at the initiation of treatment and at 3 days, 7 days and 14 days after the injection.
The animals were fasted for 18 hours prior to necropsy and blood collection. Blood samples were drawn from the abdominal aorta by using a syringe needle under ether anesthesia. Blood samples were collected into tubes containing ethylenediaminetetraacetic acid (EDTA) and were analyzed by using a blood counting analyzer (ADVIA 120, SIEMENS, Germany) to determine the red blood cell count (RBC), hemoglobin concentration (HGB), hematocrits (HCT), mean corpuscular cell volume (MCV), mean corpuscular hemoglobin (MCH), mean corpuscular cell hemoglobin concentration (MCHC), platelet count (PLT), white blood cell count (WBC), WBC differential count, reticulocyte (Reti) count, prothrombin time (PT) and active partial thromboplastin time (APTT).
For the serum biochemistry analysis, blood samples were centrifuged at 3,000 rpm for 10 minutes and analyzed using an auto-analyzer (7180, HITACHI, Japan). Serum biochemistry parameters, including alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), gamma glutamyl transpeptidase (GGT), blood urea nitrogen (BUN), creatinine, total bilirubin, total protein, albumin, albumin/globulin ratio (A/G ratio), total cholesterol, triglycerides (TG), phosphorus, glucose, calcium, chloride, sodium and potassium, were examined.
Tissue was obtained from the injection site on all animals and was then fixed with 10% neutral buffered formalin solution. The tissue was routinely processed, embedded in paraffin, and sectioned. The sections were stained with hematoxylin & eosin (H&E) stain, and bone tissue was decalcified with Calci-Clear-RapidTM (National diagnostics, U.S.A.) for microscopic examination.
Data on the weights and from the hematology and serum biochemistry results were tested by using a statistical analysis system (SAS, version 9.3, SAS Institute Inc., U.S.A.). The variance was checked by using the Bartlett test (
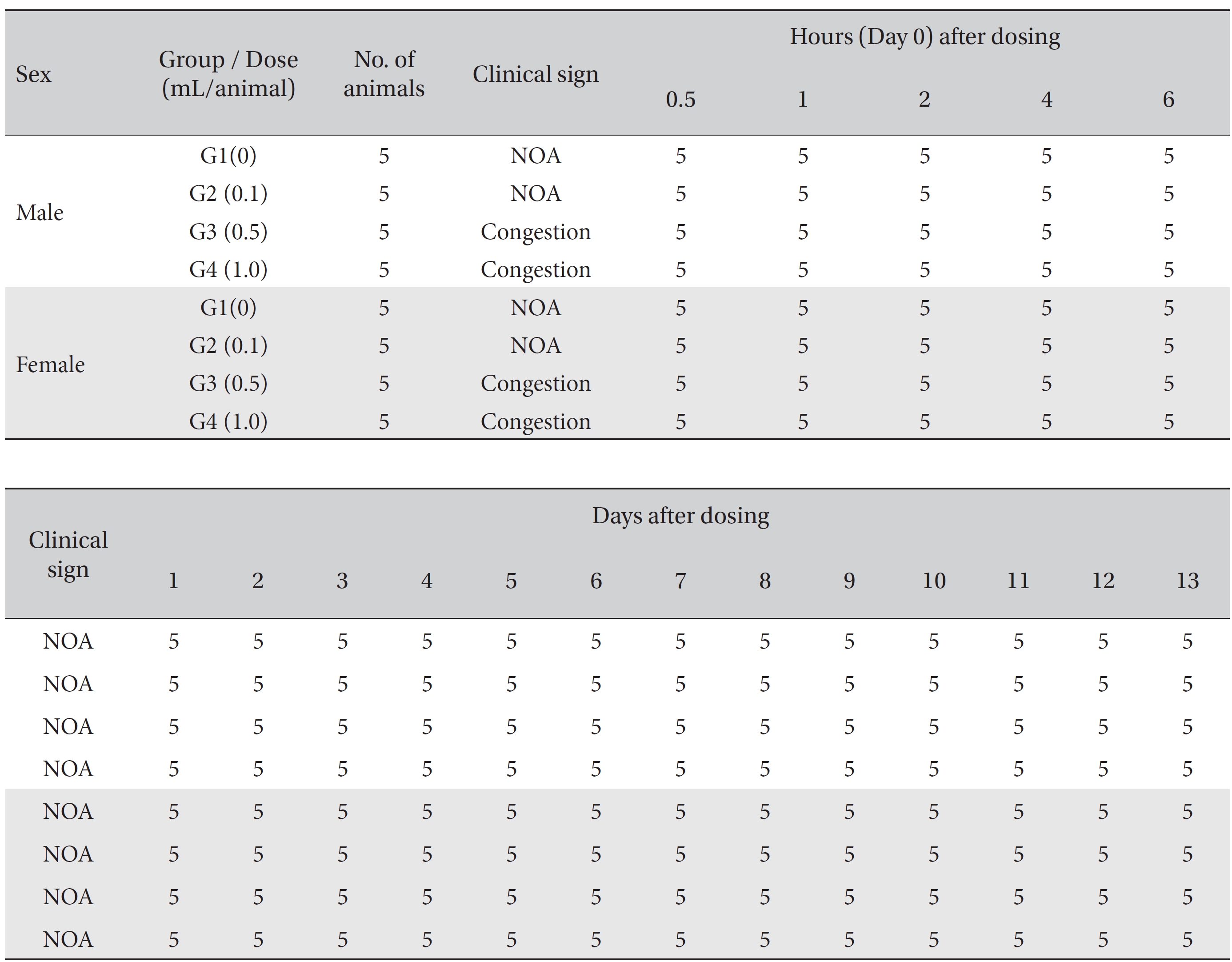
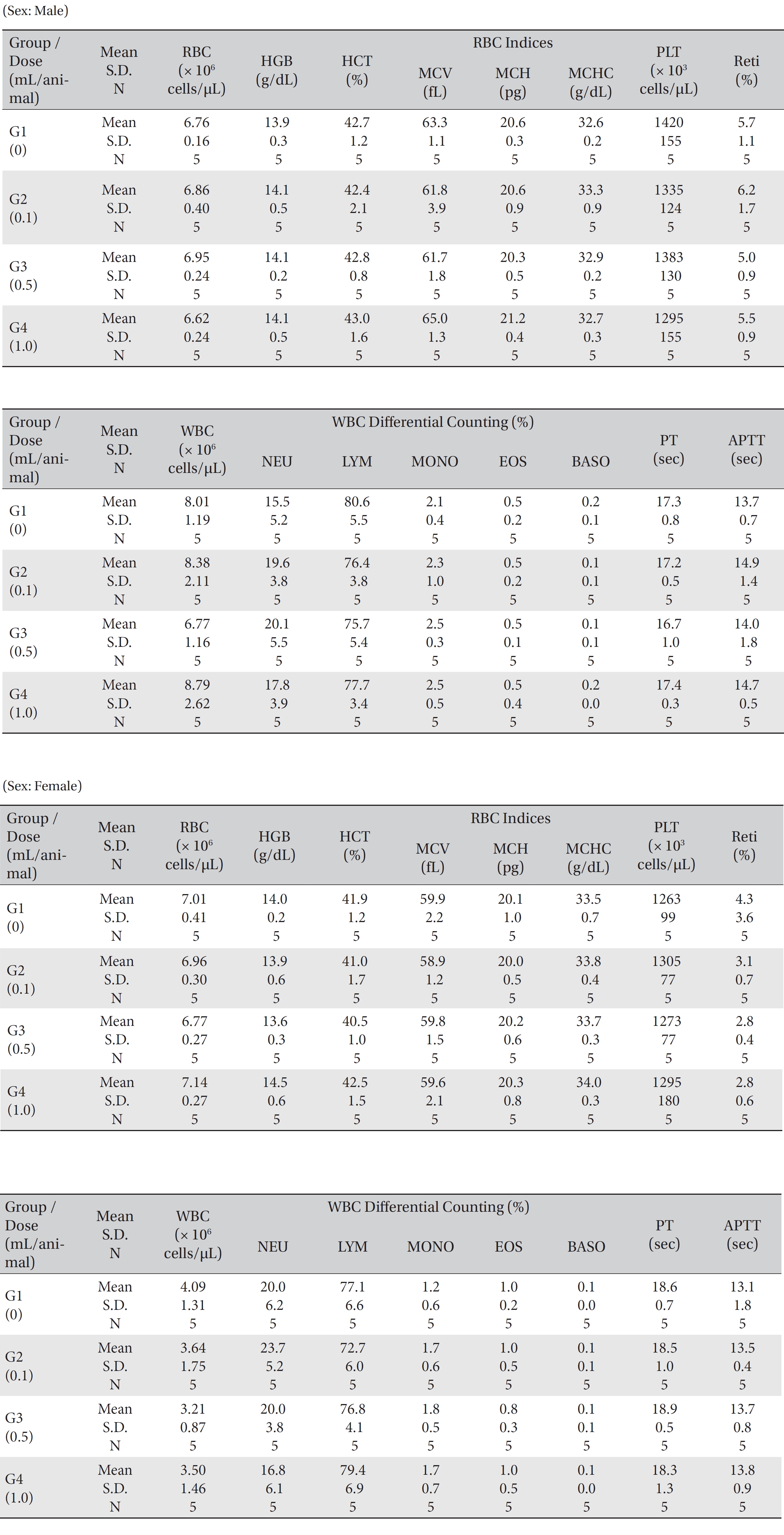
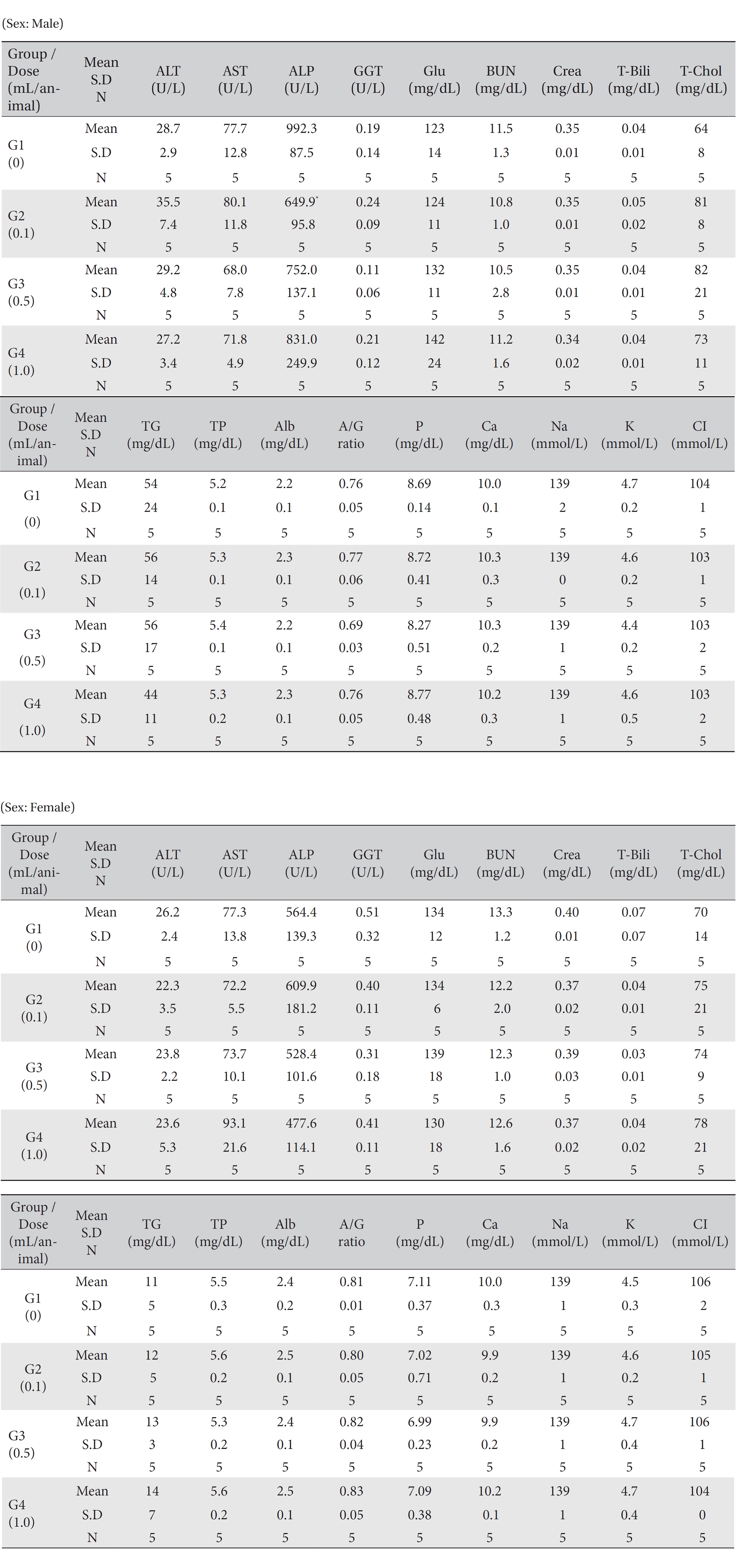
No mortality occurred in either the control group or any of the experimental groups. No clinical signs were observed in male and female SD rats during the observation period. Caudal congestion was observed in male and female rats in the experimental groups from 30 minutes to 6 hours after injection, but it was not observed the next day or after. This symptom seemed to have occurred at the administration of SBV (Table 1). Compared to the control group, the weights of the experimental groups showed no significant changes. No significant change in hematology was observed in any of the groups (Table 2), and no meaningful change in serum biochemistry was observed in any of the groups (Table 3). Significant changes were minimal and were found not to be dose dependent.
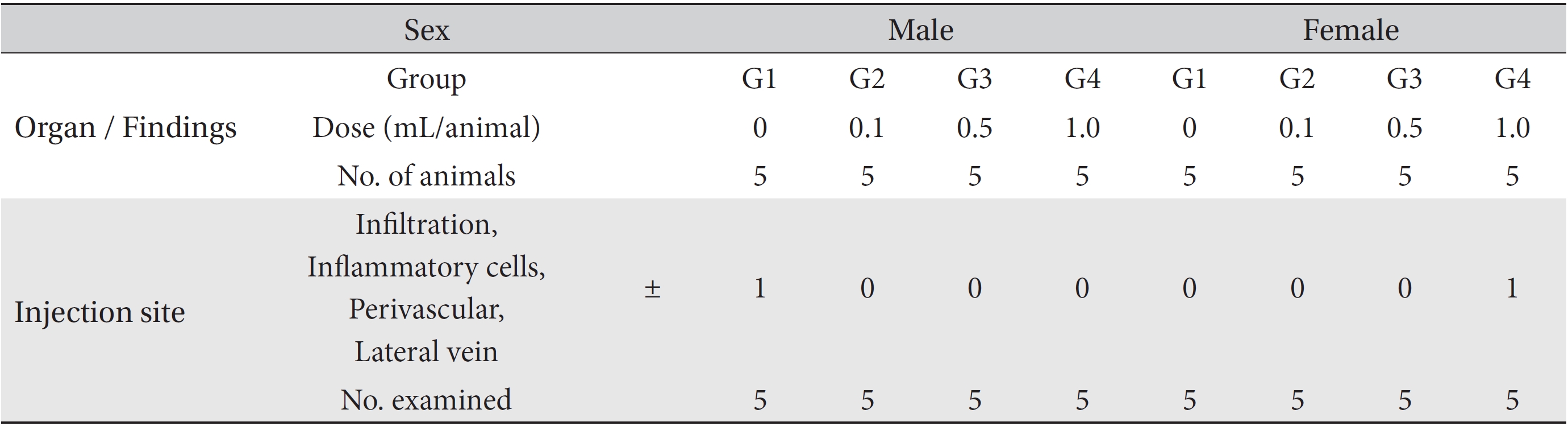
During necropsy, no abnormal macroscopic appearances were observed in either the control experimental groups. Perivascular infiltration of inflammatory cells into the lateral vein at the injection site was observed in the male control group and in the female high dosage group, but those changes were minimal and seemed to be due to the injection (Table 4).
The concept of blood vessels has been studied in Oriental medicine [20, 21], and the intravenous injection of mountain ginseng pharmacopuncture is currently being used by Korean medical doctors. BVP possesses strong analgesic and anti-inflammatory actions by stimulation of the hypothalamic-pituitary-adrenal axis to increase the secretion of adrenocortical hormone. BVP shows superior efficacy in the treatment of most pain related conditions and musculoskeletal diseases. However, bee venom contains enzymes with molecular weights greater than 10,000 such as phospholipase A2 (PLA2) and hyaluronidase. These proteins act as antigens, causing allergic reactions, which presently limits its clinical use. Administration of BVP without a full understanding of its allergic responses, especially anaphylactic shock, puts practitioners and patients at great risk [1]. SBV was developed to replace dangerous bee venom with a safe material and to guarantee its effects [22, 23] because SBV is pure melittin obtained through a protein separation technique using gel filtration [12, 24-26].
In previous studies, the LD50 of SBV in SD rats was over 30 mg/kg [14], and the maximum dose of SBV in beagle dogs was found to be over 9 mg/kg [15] through single-dose toxicity tests. Also, no significant changes were observed at doses below 0.14 mg/kg in SD rats, and no direct side effects of SBV were observed at a dose of 0.56 mg/kg in beagle dogs through a 4-week repeated-dose toxicity test, and the ‘No Observed Adverse Effect Level’ of SBV was found to be approximately 0.07 mg/kg in male and female SD rats based on a 13-week repeated-dose toxicity test with a 4-week recovery period [16-19].
Whether intravenous injection of SBV is possible is controversial. However, in our opinion, the use of SBV should be safe, so we conducted an intravenous single-dose toxicity test of SBV in SD rats. Our objective was to investigate the influences of intravenous injection of SBV in SD rats. In the present study, no significant changes were observed in the weights or in the results of the hematology, biochemistry, necropsy and histopathological examinations in SD rats. These results show that intravenous injection of SBV in SD rats is safe. Because no mortalities were observed in this study, the lethal dose of MGP is judged to be above 1.0 mL/animal in SD rats. This study is the first attempt to evaluate the safety of intravenous SBV injection, and further studies on this subject are needed.
The results of this study suggest that the lethal dose of SBV is above 1.0 mL/animal in SD rats and that intravenous injection of SBV in SD rats is safe.
[Table. 1] Summary of clinical signs
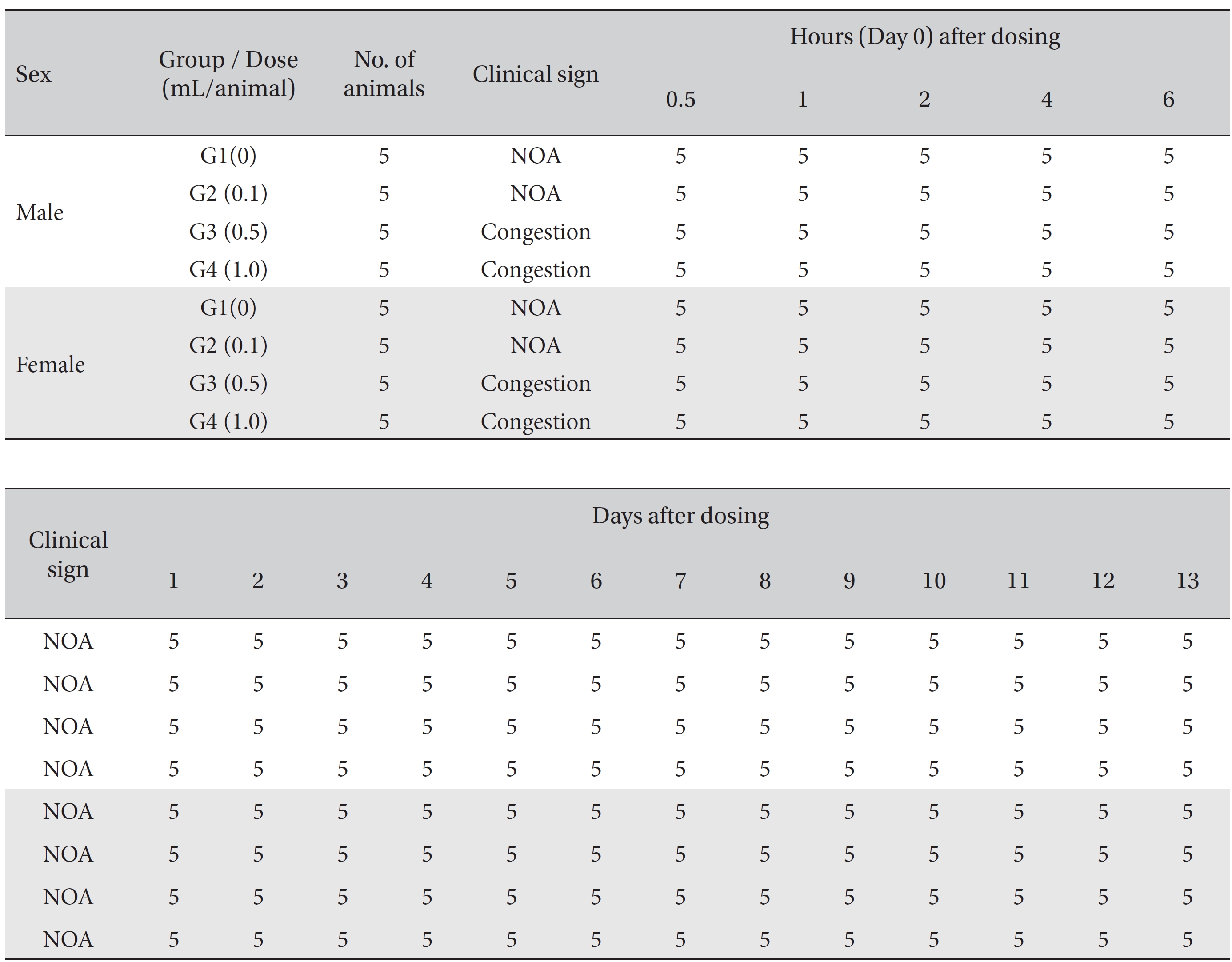
Summary of clinical signs
[Table. 2] Mean hematology parameters
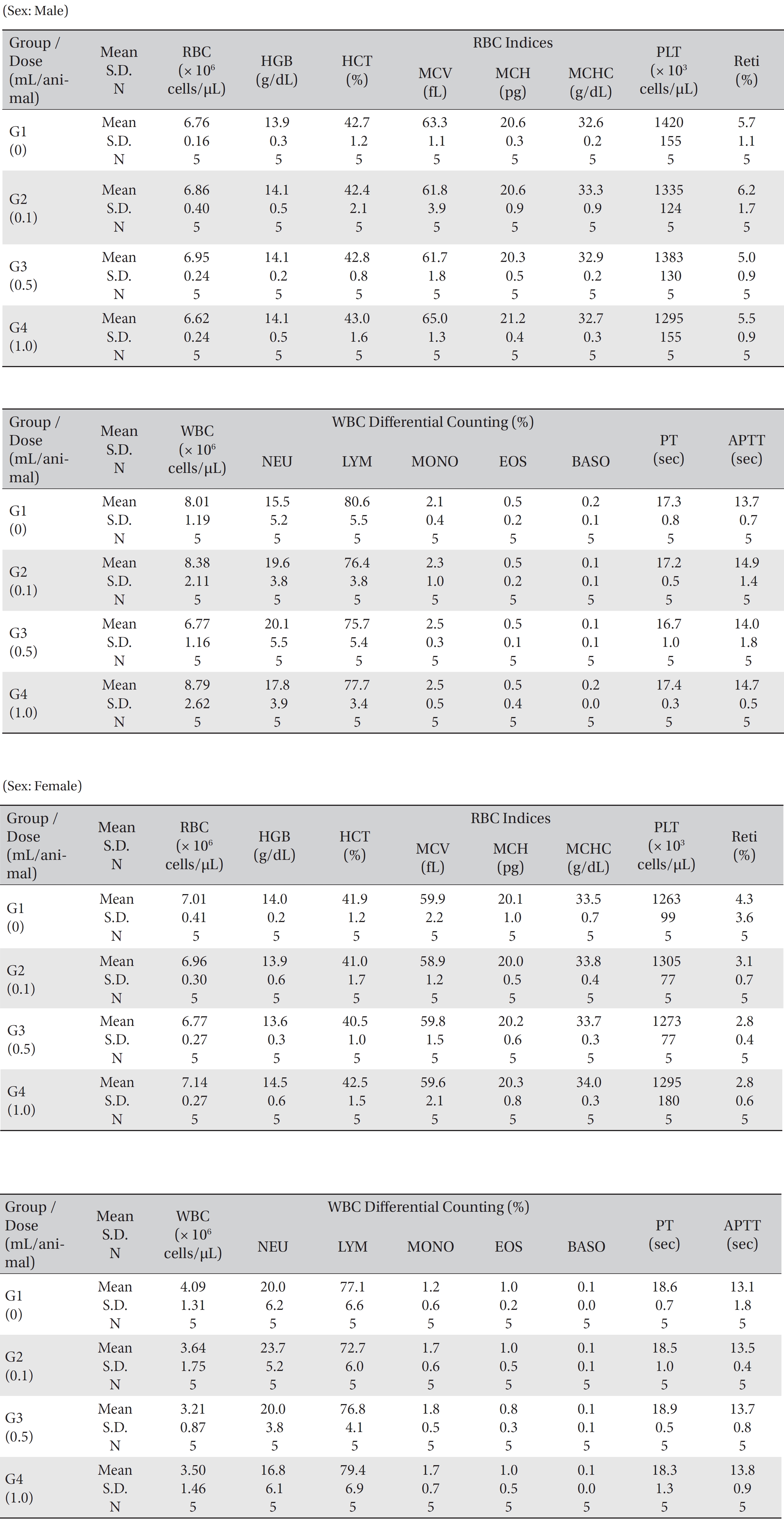
Mean hematology parameters
[Table. 3] Mean clinical chemistry
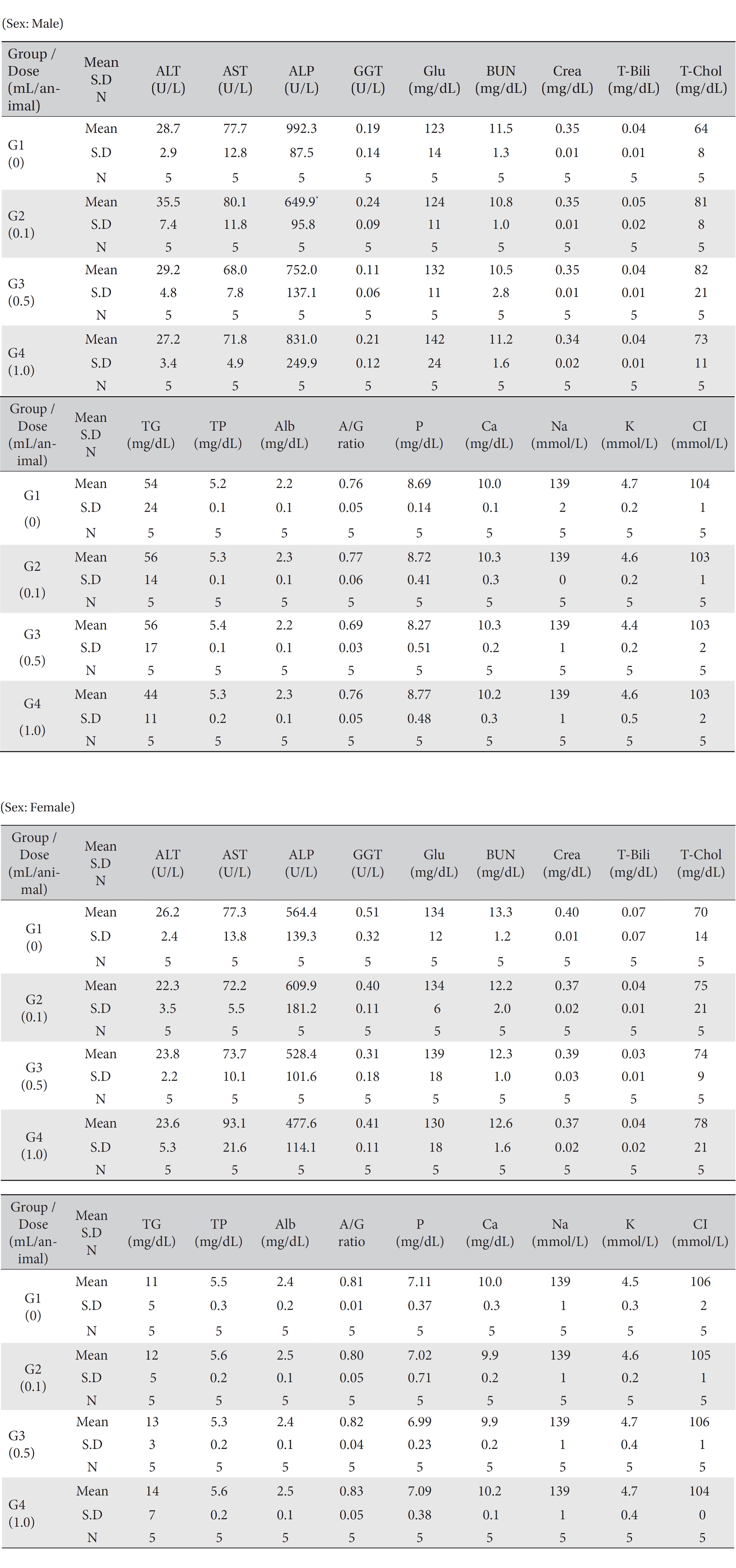
Mean clinical chemistry
[Table. 4] Summary of histopathological findings
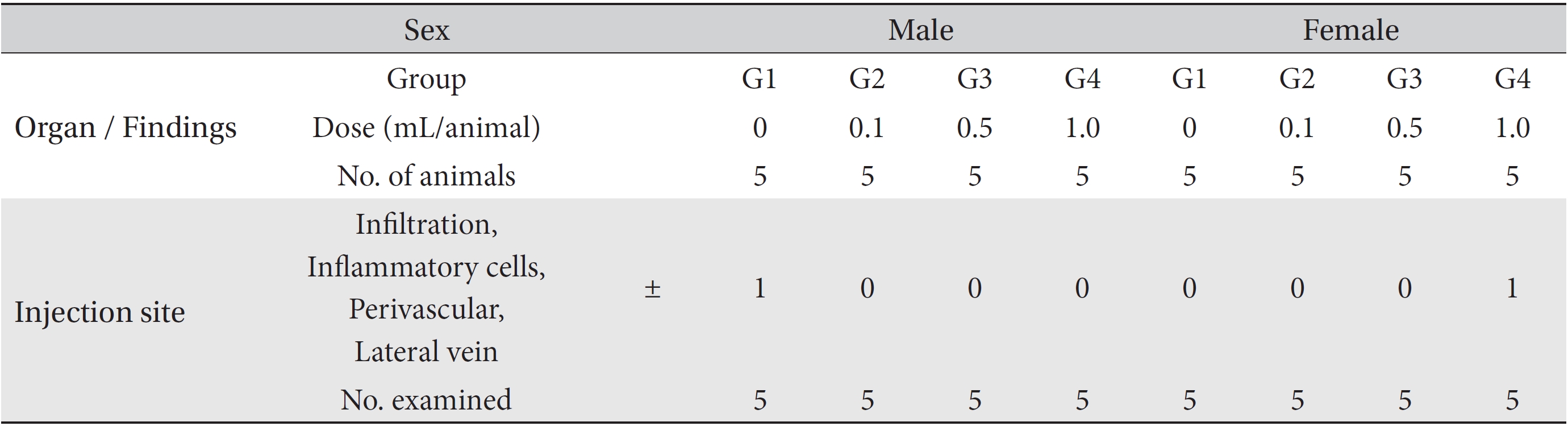
Summary of histopathological findings