In South Korea, octopus is a commonly consumed cephalopod, with an output of 11,398 metric tons in 2010 (Agriculture and Fishery Statistics Department, 2010). The cephalopod has a short life cycle and is characterized by a high growth rate (Hurtado et al., 1999). Because such rapid growth involves a high turnover of proteins, intense proteolytic activity has been found in cephalopod muscle and viscera (Sakai and Matsumoto, 1981). The octopus processing industry generates large amounts of by-products, such as the viscera, skin, and bone, which can have adverse effects on the environment (Souissi et al., 2008). Among these solid by-products, the viscera accounts for 20–40% of the original material. Therefore, advantages exist in converting such viscera waste into resources to extract useful enzymes.
Proteases are the most important industrial enzymes used worldwide, accounting for about 50% of the total industrial enzyme market (Rao et al., 1998; Bougatef et al., 2007). These proteases are characterized by their mechanism of hydrolysis into endoproteinases or exopeptidases (Guerard, 2007). The breakdown of proteins into peptides is catalyzed primarily by endoproteases such as trypsin, chymotrypsin, pepsin, papain, and elastase, which cleave peptide bonds in protein molecules, usually at specific residues, to produce relatively large peptides (Kristinasson and Rasco, 2000). Exopeptidases such as aminopeptidase and carboxypeptidase then systematically detach the C- or N-terminal amino acids from the protein or peptide chain. In the food industry, proteases are also used most extensively to improve quality, stability, or solubility in the baking, brewing, and cheese-making industries, as well as meat processing (Haard, 1990; Gupta et al., 2002). However, the enzyme hydrolysates of various proteins frequently exhibit a bitter taste caused by specific peptides (Matoba et al., 1970; Clegg et al., 1974; Bumberger and Belitz, 1993; Izawa et al., 1997), which limits their potential utility in the food industry. Because this bitterness is closely correlated with peptide hydrophobicity (Clegg et al., 1974), several attempts have been made to reduce bitterness by hydrolyzing bitter peptides using exopeptidases (Umetsu and Ichishima, 1988; Minagawa et al., 1989). Although many industrial proteases have been developed, only a limited number of commercial peptidases exist, including aminopeptidase and carboxypeptidase (Deejing et al., 2005).
For the efficient use of exopeptidases from the cephalopod hepatopancreas (HP), extraction, fractionation, and purification procedures must be used. The fractionation process can affect enzyme activity, and in recent years, many studies have been performed to efficiently use cephalopod HPs as a resource for extracting endoproteases (Ezquerra-Brauer et al., 2002; Kim et al., 2007, 2008a) and their inhibitors (Kishimura et al., 2001), as well as exopeptidases (Raksakulthai and Haard, 1999; Raksakulthai et al., 2002; Kim et al., 2008b). However, few studies have assessed the efficient use of octopuses as a resource for extracting endopeptidases and exopeptidases.
In the present study, fractions obtained using various fractionation methods, such as solid ammonium sulfate fractionation, PEG fractionation, anion exchange chromatography, and gel filtration chromatography, were enzymatically characterized to identify the most efficient fractionation method to obtain exopeptidase-active fractions from octopus HPs.
The cephalopod HP contains several enzymes. Frozen octopus (
Chemicals used to fractionate and collect exopeptidases were purchased from Katayama Chemical Co. (Osaka, Japan) for ammonium sulfate fractionation, Yacuri Pure Chemical Co. Ltd (Kyoto, Japan) for PEG fractionation. Leu-pNA (Sigma-Aldrich Co. St. Louis, MO, USA) and azocasein (Sigma-Aldrich Co.) were used as the substrates for measuring exopeptidase and endopeptidase activities, respectively. All other chemicals were of analytical grade.
The buffer solutions (0.1 M sodium phosphate buffer, pH 6.0) used to fractionate the crude extracts (CEs) and enzyme reactions were prepared as described previously (Dawson et al., 1986).
>
Preparation of the crude extracts
To generate the octopus HP CEs, the frozen octopus HP was partially thawed, and then homogenized with three volumes (v/w) of deionized water. For enzyme activation, the homogenates were incubated at 20°C for 4 h with stirring for every 30 min and then centrifuged at 1,200
The protein concentrations of the CEs were measured according to the method of Lowry (Lowry et al., 1951) using bovine serum albumin (Sigma-Aldrich Co.) as the standard. Protein concentration of chromatography for fractionating enzymes was measured at 280 nm.
>
Endoprotease and exopeptidase activity
Endoprotease activity was assayed using azocasein as the substrate following the method described by Starky (1977), with some modifications. One-hundred microliters of CE was mixed with 300 μL of 1% azocasein (Sigma-Aldrich Co.) in 1.6 mL of 0.1 M sodium phosphate (pH 6.0), followed by incubation at 40°C for 1 h. The reaction was stopped by the addition of 2 mL of 5% trichloroacetic acid (TCA), and the mixture was centrifuged at 146
Exopeptidase activities were determined using Leu-pNA (Sigma-Aldrich Co.) as the substrate using a modified version of the method reported by Garcia-Carreno and Haard (1993). One hundred microliters of CE was mixed with 100 μL Leu-pNA in 2 mL of 0.1 M sodium phosphate buffer (pH 6.0) and then incubated at 40°C for 1 h. The reaction was stopped by the addition of 0.3 mL of 33% acetic acid, and the mixture was centrifuged at 146
One unit of endoprotease and exopeptidase activity was defined as the amount of enzyme that increased the absorbance by 0.1 unit per min. Specific activity (U/mg) was defined as the units of activity per mg of protein.
>
Fractionation of crude extracts using salt
The CEs were fractionated using solid ammonium sulfate ranging from 0−20%, 20−40%, 40−60%, and 60−80% saturation, and the fractions were collected by centrifugation (12,000
>
Fractionation of crude extracts using polyhydric alcohols
The CEs were fractionated using PEG concentrations of 0–5%, 5–10%, 10–20%, and 20–40%, and the fractions were collected by centrifugation at 12,000
>
Fractionation of crude extracts by a difference of ion strength
A Toyopearl DEAE 650M column (1.6 × 20 cm; Tosoh Co. Ltd., Pyungtaek, South Korea) was equilibrated with 10 mM sodium phosphate buffer (pH 7.0), and then washed with the same buffer until the conductivity of the eluent and buffer were equal. To obtain exopeptidase-active fractions, the CEs were applied to a column and eluted, and then separated with 110 mL of an increasing linear NaCl gradient (0, 0.5, and 1.0 M) at a flow rate of 30 mL/h. The protein content (absorbance 280 nm) and protease activity (absorbance 410 nm) were measured. The exopeptidase-active fractions (4 mL/tube) were treated as described above using different ionic strengths for concentrating and dialysis. The supernatants were stored at –25°C until characterization.
>
Fractionation of crude extracts according to molecular weight
Sephacryl S-300 column (1.6 × 100 cm; Pharmacia, Uppsala, Sweden) was equilibrated with 10 mM sodium phosphate buffer (pH 7.0), and then washed with the same buffer until the conductivity of the eluent and buffer were equal. To obtain exopeptidase-active fractions, the CEs were applied to the column and eluted, and separated at a flow rate of 12 mL/h. The protein content (absorbance at 280 nm) and protease activity (absorbance at 410 nm) were then measured. The exopeptidase-active fractions (4 mL/tube) were collected and centrifuged at 3,000 rpm for 30 min using an Amicon Bioseparations Centriplus (centrifugal filter devices for volumes up to 15 mL; Millipore, Billerica, MA, USA) for concentration and dialysis. The supernatants were stored at –25°C until characterization.
>
Comparison of the endoprotease and exopeptidase activities of the crude extracts
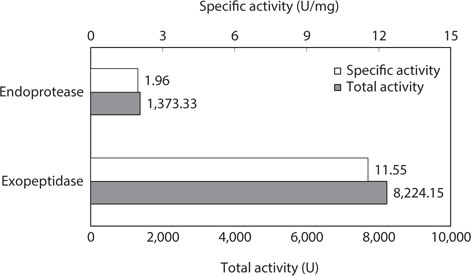
To examine the potential of octopus HP as a resource for extracting endoprotease- and/or exopeptidase-active fractions, the specific and total endoprotease and exopeptidase activities of the CE fractions were measured and compared (Fig. 1). The specific and total endoprotease activity of the CE was 1.96 U/mg and 1,373.33 U, respectively, which constituted about 16.7% of the total exopeptidase activity. Conversely, the exopeptidase activities were 11.55 U/mg and 8,224.15 U, respectively. These results suggest that octopus HP is a potential superior resource for extracting exopeptidases compared to endoproteases. Kim et al. (2008a, 2008b) reported that the specific exoprotease activity of CEs from Argentinian shortfin squid HPs using Leu-pNA as a substrate was 4.66 U/mg, which is 2.86-times higher than that of endoprotease using azocasein as a substrate of 1.63 U/mg. They concluded that Argentinian shortfin squid HP could be a potential resource for extracting exopeptidases. Taken together, these data suggest that CEs might be useful as a resource for extracting exopeptidases because octopus HP CEs had higher exopeptidase than endoprotease activity.
>
Optimum fractionation method for obtaining exopeptidase-active fractions
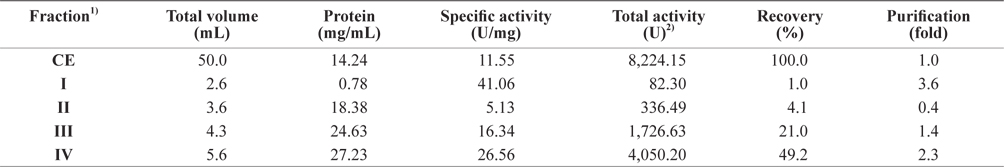
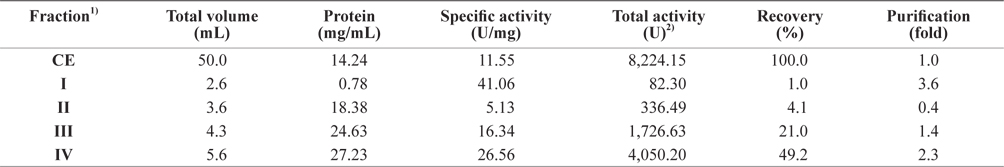
To assess the optimum fractionation method to obtain exopeptidase-active fractions from octopus HP, the CEs were fractionated consecutively using varying ammonium sulfate concentrations; the fractions were designated fraction I (obtained using 0–20% ammonium sulfate), II (20–40%), III (40–60%), and IV (60–80%). The exopeptidase activity and the yield of octopus HP CE fractions obtained using various ammonium sulfate concentrations are shown in Table 1. The specific exopeptidase activity and purity of fractions from CEs of octopus HP were the highest in fraction I (41.06 U/mg and 3.6-fold, respectively), followed by fractions IV (26.56 U/mg and 2.3-fold, respectively), III (16.34 U/mg and 1.4-fold, respectively), and II (5.13 U/mg and 0.4-fold, respectively). This suggests that fractions I and IV contained more purified exopeptidase than the CE, but fractions II and III did not. The purification of exoprotease from the CE was affected by the presence of ammonium sulfate, but was independent of concentration.
Exopeptidase activities of fractions obtained from the crude extracts of octopus heptapancreas by the ammonium sulfate fractionation toward LeuPNA as a substrate
Raksakulthai and Haard (1999) reported that the purity and recovery of exopeptidase activity in squid CE (
The total recovery of exopeptidase activity from CE fractions of the octopus HP was 75.3%. The recovery of exopeptidase activity from fractions III and IV was 21.0% and 49.2%, respectively, which constituted about 28% and 65% of the total recovery, respectively. The total exopeptidase activity of the CE fractions from octopus HP was the highest in fraction IV (4,050.20 U), followed by fraction III (1,726.63 U); fractions II and I had low activities of <350 U. These results suggest that the exoprotease active fraction from the CE of octopus HP could be fractionated effectively using 60–80% ammonium sulfate
Kim et al. (2008b) reported that the exoprotease-active fraction from CEs of octopus HP could be isolated effectively using 30–40% ammonium sulfate, with a total activity of 2,071.10 U.
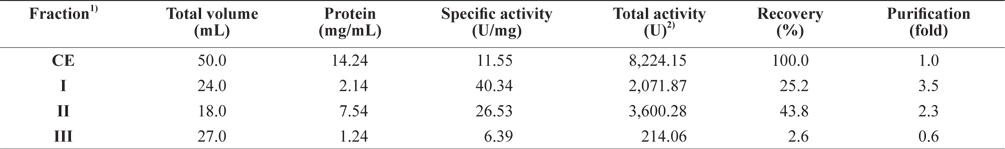
To assess the optimum conditions to isolate the exopeptidase-active fraction from octopus HP, the CEs were fractionated using increasing concentrations of PEG. The fractions were designated fraction I (obtained using 0–5% ammonium sulfate), II (5–10%), III (10–20%), and IV (20–40%).
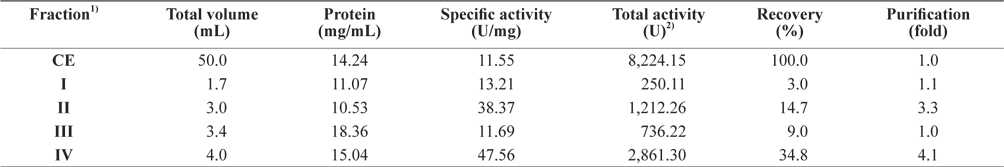
Exopeptidase activity and the yield of the fractions from CEs of octopus HP obtained using various concentrations of PEG are shown in Table 2. The specific activity and purity of exopeptidase activity was highest in fraction IV (47.56 U/mg and 4.1-fold, respectively), followed by fraction II (38.37 U/mg and 3.3-fold, respectively), I (13.21 U/mg and 1.1-fold, respectively), and III (11.69 U/mg and 1.0-fold, respectively). These results suggest that fractions II and IV contained purified exopeptidase activity compared with the CE, whereas the other fractions, such I and III, did not. According to analysis of the specific activity and purity of the fractions, the exoprotease purity from CEs was affected by PEG, but was independent of concentration.
Exopeptidase activities of fractions obtained from the crude extracts of octopus heptapancreas by the polyethylene glycol (PEG) fractionation toward LeuPNA as a substrate
The total recovery of exopeptidase activity in CE fractions of octopus HP was 61.5%; the activity of fraction IV was 34.8%, which constituted about 57% of the total recovery. The total exopeptidase activity was the highest in the fraction IV (2861.30 U), followed by fraction II (1,212.26 U); other fractions, such as I and III, had low activities of <750 U. These results suggest that the exoprotease active fraction from CEs of octopus HP could be obtained effectively using 10–20% PEG-based fractionation.
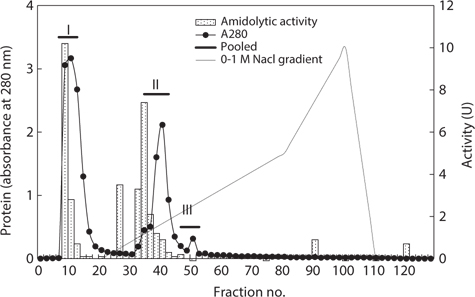
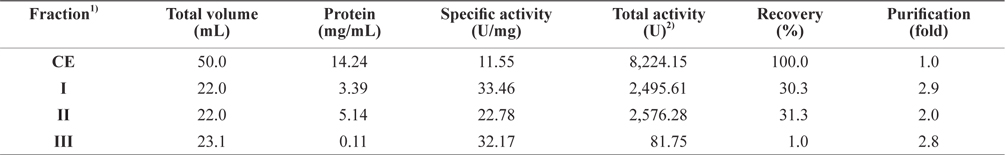
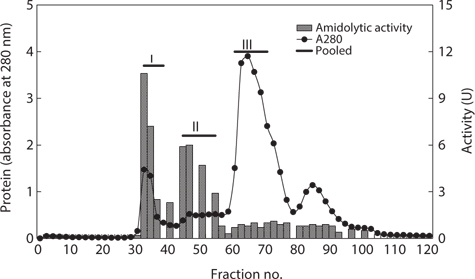
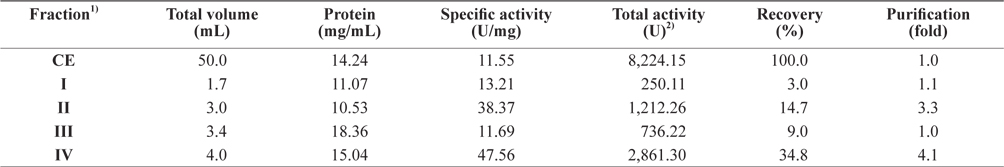
Next, the optimum fractionation conditions using DEAE- 650M anion-exchange chromatography were assessed, and the resulting chromatogram is shown in Fig. 2. Based on the protein concentration and exopeptidase activity shown in the ion exchange chromatogram, these fractions were designated fraction I (a concentrate of fractions 7–12), II (fractions 34– 42), and III (fractions 46–52). The exopeptidase activity and yield of the CE fractions of octopus HP obtained using ionexchange chromatography are shown in Table 3. The specific activity and purity was highest in fraction I (33.46 U/mg and 2.9-fold, respectively), followed by fractions III (32.17 U/mg and 2.8-fold, respectively) and II (22.78 U/mg and 2.0-fold, respectively). These results suggest that all fractions obtained using ion-exchange chromatography contained purified exopeptidase activity compared with the CE. The purification of exoprotease from the CE was affected by ionic strength, but was independent of ion strength.
Exopeptidase activities of fractions obtained from the crude extracts of octopus heptapancreas by DEAE-650M ion exchange chromatography toward LeuPNA as a substrate
Kim et al. (2008b) reported that an exopeptidase active fraction could be effectively obtained from CEs of Argentinian shortfin squid (
The total recovery of exopeptidase activity in the fractions was 62.6%; the activity in fractions I and II was 30.3% and 31.3%, respectively, accounting for about 48% and 50%, respectively, of the total recovery. The total exopeptidase activity of the fractions was highest in fraction II (2,576.28 U) followed by fraction I (2,495.61 U); fraction III had a very low activity of 81.75 U. These results suggest that the exopeptidase- active fraction from CEs of octopus HP could be fractionated effectively by concentrating fractions 7–12 and 34–42 of ion exchange chromatography
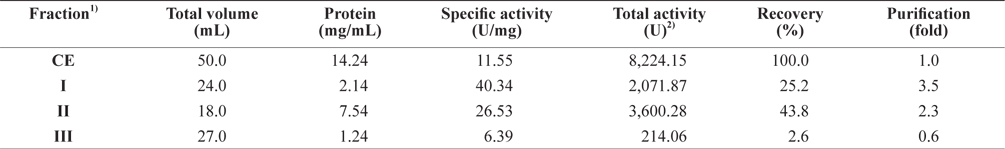
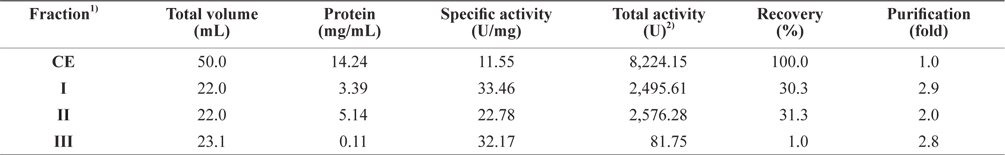
The optimum fractionation conditions to obtain exopeptidase- active fractions from CEs of octopus HP using gel filtration chromatography were assessed using a Sephacryl S-300 resin (Tosoh Co. Ltd). The resulting gel filtration chromatogram is shown in Fig. 3. Based on the protein concentrations and exopeptidase activity, the fractions were designated fraction I (a concentrate of fractions 32–38), II (fractions 44–54), and III (fractions 60–70).
The exopeptidase activity and yield of CE fractions of octopus HP obtained using gel filtration chromatography are shown in Table 4. The specific exopeptidase activity and purity was highest in fraction I (40.34 U/mg and 3.5-fold, respectively), followed by fractions II (26.53 U/mg and 2.3-fold, respectively) and III (6.39 U/mg and 0.6-fold, respectively). These results suggest that fractions I and II obtained using gel filtration chromatography contained purified exopeptidase activity, but that fraction III did not. The purity of exoprotease activity was affected by molecular weight and specifically increased with increasing molecular weight.
Exopeptidase activity of fractions obtained from the crude extracts of octopus heptapancreas by the DEAE-650M anion exchange chromatography toward LeuPNA as a substrate
The total recovery of exopeptidase activity in the fractions obtained using gel filtration chromatography was 71.6%. The recovery in fractions I and II was 25.2 and 43.8%, respectively, which constituted about 35% and 61% of the total recovery, respectively. The total exopeptidase activity was highest in fraction II (3,600.28 U), followed by fraction I (2,071.87 U); fraction III had very low activity of only 214.06 U. These results suggest that exopeptidase activity could be fractionated from CEs of octopus HP by obtaining concentrates of fractions 32–38 and 44–54 using gel filtration chromatography.
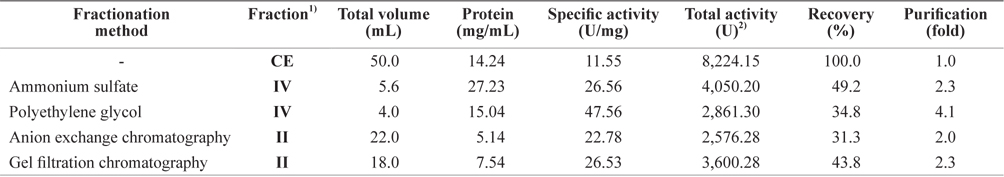
Exopeptidase activity and yield of the fractions obtained using the different fractionation methods are shown in Table 5. The fractions with the highest total activity toward Leu-pNA were fraction IV from the ammonium sulfate and PEG fractionations, and fraction II from ion exchange and gel filtration chromatography. The specific exopeptidase activity and purity was highest in fraction IV from PEG fractionation (47.56 U/mg and 4.1-fold, respectively), followed by fraction IV from ammonium sulfate fractionation (26.56 U/mg and 2.3-fold, respectively), fraction II from gel filtration chromatography (26.53 U/mg and 2.3-fold, respectively), and fraction II from ion exchange chromatography (22.78 U/mg and 2.0-fold, respectively). The total exoprotease activity was highest in fraction IV of ammonium sulfate fractionation (4,050.20 U), followed by fraction II of gel filtration chromatography (3,600.28 U), fraction IV of PEG fractionation (2,861.30 U), and fraction II of anion exchange chromatography (2,576.28 U).
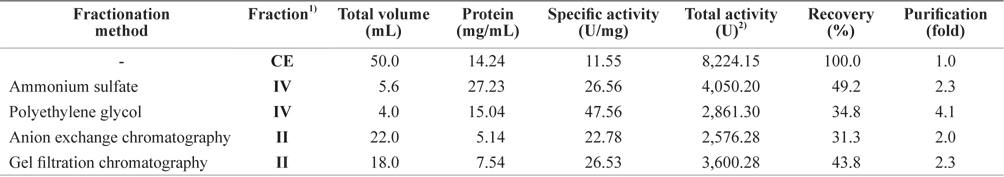
Comparison of exopeptidase activity of fractions from the crude extracts of octopus hepatopancreas obtained by different fractionation methods toward LeuPNA as a substrate
These results suggest that ammonium sulfate fractionation using 60–80% ammonium sulfate is the most efficient method for separating the exoprotease active fractions from CEs of octopus HP.