Pollination is an ecosystem service, given that wild pollinators, particularly wild bees, contribute significantly to the pollination of a wide range of crops (Morandin and Winston, 2005; Greenleaf and Kremen, 2006; Winfree et al., 2007). Bees are critical for the pollination of natural vegetation and agricultural plants, including fruits, vegetables, seed plants, edible oil crops, garden flowers, and major forage crops. Bees are both widely diverse and abundant, with 16,325 species currently identified worldwide (Michener, 2000). Commercially managed bees are available for pollination services and they are used in large commercial fields, small gardens, and enclosures, such as greenhouses and screen houses (Free, 1993; Dag and Kammer, 2001). The use of bumblebees, one of commercially managed bees, for pollination has become widespread, and the demand for bumblebees increases every year.
Generally, Bombus species are eusocial insects with short-lived colonies that are found primarily in the temperate regions of the world. Queens are the only caste of Bombus to overwinter (enter diapause), whereas the workers and males perish in the late summer and early autumn, respectively. In the early spring, queens that have overwintered depart from their hibernation sites. The queen builds up a store of pollen and then lays her first batch of eggs into the pollen mass after finding a suitable site for the foundation of a colony. As soon as the workers of the first brood have emerged, they assume the foraging actives of the queen, after which she spends her time predominantly laying eggs. In the late summer, many males and new queens are produced. Only the mated queens hibernate and emerge in the spring (Heinrich, 1979; Duchateau and Velthusis, 1988).
Bumblebees generally produce one generation per year. One of the key steps in year-round rearing of bumblebees is breaking diapause. Diapause is defined as a stage in the development of certain animals during which morphological development may be suspended or markedly retarded (Andrewartha, 1952; Mansigh, 1971). The programming of diapause involves the development of specific behavioral, morphological, and physiological design features that uniquely prepare the diapause-destined insect for a period of developmental arrest (Denlinger, 2002). With the goal of inducing diapause break, several researchers attempted to first induce hibernation in bumblebee queens under controlled conditions, despite their lengthy ovarian diapause (Horber, 1961; Alford, 1969, 1975; Hoem, 1972; Beekman et al., 1998), or by CO2 narcosis (Röseler and Röseler, 1984; van den Eijnde et al., 1991). Under natural conditions, the duration of the hibernation period ranges from 6 to 9 mo (Alford, 1969). Hoem (1972) maintained hibernating B. terrestris queens in mounds of soil in unheated greenhouses or in plastic containers with perlite as bedding; the bees were subsequently maintained in a refrigerator at 4-5℃ for 8-9 mo. Röseler and Röseler (1984) demonstrated that prediapausing B. terrestris queens that were narcotized using carbon dioxide (a 30-min narcosis treatment repeated twice) would start laying eggs within a week. However, this method produced many side effects. Pormeroy and Plowright (1979) found that this treatment induced the ejection of larvae by bumblebee workers in narcotized colonies. Röseler (1985) reported the emergence of some males among the first batch of workers in such colonies. Carbon dioxide-treated bumblebee queens sometimes produced males instead of workers and their nests could be of smaller size than those of overwintered queens (Tasei, 1994; Yoon et al., 2003). Although increased survival rates have been reported in some studies, few studies have attempted to evaluate the effects of different diapause methods, including a combination method of CO2-narcosis and cold treatment, on the survival rates of diapaused queens and their subsequent ability to establish a colony.
To evaluate the effects of a combination method of CO2-narcosis and cold treatment for diapause break in B. ignitus and B. terrestris queens, we determined whether this method affected their ability to establish a colony after diapause break. This is the first study describing a combination method of CO2-narcosis and cold-treatment application for breaking the diapause of B. ignitus and B. terrestris queens.
The insects used in the experiment were second and sixth-generation queens acquired from B. ignitus and B. terrestris colonies that were reared year-round in a climate-controlled room (27℃, 65% relative humidity, and continuous darkness) at the Division of Applied Entomology, Department of Agricultural Biology, National Academy of Agricultural Science, Republic of Korea.
The basic colony-rearing technique used in the present study was described previously by Yoon et al. (2002). The queens were reared in three different types of cardboard and plastic boxes for nest initiation (10.5 × 14.5 × 6.5 cm), colony foundation (21.0 × 21.0 × 15.0 cm), and colony maturation (24.0 × 27.0 × 18.0 cm). Queens were first confined individually in small boxes for colony initiation and remained there until oviposition. After the adults from the first brood had emerged, the nest was transferred to a medium-sized box for colony foundation and was stored there until the number of workers reached 50. The nest was subsequently moved to a large box for further colony development. A 50% sugar solution and pollen dough were provided ad libitum. The pollen dough was composed of a sugar solution and pollen (v:v 1:1).
To evaluate the effects of different diapause-breaking methods on the colony development of B. ignitus queens, the following diapause-breaking treatments were used: CO2 (CO2–narcosis), CT-1M (cold treatment at 5℃ for 1 mo), CT-1M-CO2 (CO2–narcosis after cold treatment at 5℃ for 1 mo), CT-2M (cold treatment at 5℃ for 2 mo), or CT-2M-CO2 (CO2–narcosis after cold treatment at 5℃ for 2 mo). CO2-narcosis involved exposure to 99% CO2 for 30 min daily during two consecutive days beginning the second day after mating (Yoon et al., 2003). The cold-temperature-treated queens were individually maintained in bottles filled with perlite in a perforated plastic box containing perlite to prevent mold growth under a constant temperature of 5℃ and 80% humidity. At the end of treatment, the queens were transferred to flight cages for three days (Yoon et al., 2004b). Each queen was reared in a climate-controlled room (27±1℃, 65% R.H. and continuous darkness). To stimulate egg-laying, two narcotized B. ignitus workers aged 10-20 d (after emergence) were placed with each queen (Yoon and Kim, 2002). The number of B. ignitus queens tested in this study using CO2, CT-1M, CT-1M-CO2, CT-2M, or CT-2M-CO2 was 32, 18, 23, 48 and 50, respectively.
The ability of a queen to develop a colony was based on the preoviposition period, the rates of oviposition, colony foundation, and progeny-queen production, the period until the emergence of the first adult, and the number of adults. The colony foundation period was defined as the period necessary for more than 50 workers to emerge from a colony. The period until the emergence of the first adult represents the duration from the first oviposition to the emergence of the first adult. The queens that did not evidence oviposition within 60 d were excluded from the analysis of the number of oviposited colonies.
To examine the effects of different diapause-breaking treatments on the colony development of B. terrestris queens, the following treatments were used: CO2 (CO2–narcosis), CT-1M (cold treatment at 5℃ for 1 mo), CT-1M-CO2 (CO2–narcosis after cold treatment at 5℃ for 1 mo), CT-2M-CO2 (CO2–narcosis after cold treatment at 5℃ for 2 mo), CT-2M (cold treatment at 5℃ for 2 mo), CT-2.5M-CO2 (CO2–narcosis after cold treatment at 5℃ for 2.5 mo), and CT-2.5M (cold treatment at 5℃ for 2.5 mo). The conditions used for CO2-narcosis and cold treatment of B. terrestris queens were identical to those used for B. ignitus queens. Each queen was reared in a climate-controlled room (28±1℃, 65% R.H. and continuous darkness). To stimulate egg-laying, two narcotized B. terrestris workers aged 10-20 d (after emergence) were placed with each queen. The number of B. terrestris queens used in the, CO2, CT-1M, CT-1M-CO2, CT-2M, CT-2M-CO2, CT-2.5M, and CT-2.5M-CO2 experiments was 35, 44, 54, 44, 45, 100 and 72, respectively.
The ability of a queen to develop a colony was based on the preoviposition period, the rates of oviposition, colony foundation, and progeny-queen production, the period until the emergence of the first adult, and the number of adults. The colony foundation period was defined as the period necessary for more than 50 workers to emerge from a colony. The period until the emergence of the first adult represents the duration from the first oviposition to the emergence of the first adult. The queens that did not evidence oviposition within 60 d were excluded from the analysis of the number of oviposited colonies.
The statistical analyses were conducted using the Chi-squared test and Tukey’s pairwise comparison test (MINITAB Release 13 for Windows, 2000). The Chi-squared test was used to compare the colony development of B. ignitus and B. terrestris queens. Tukey’s pairwise comparison test was used to examine the durations until colony initiation and first adult emergence, as well as the number of adults produced.
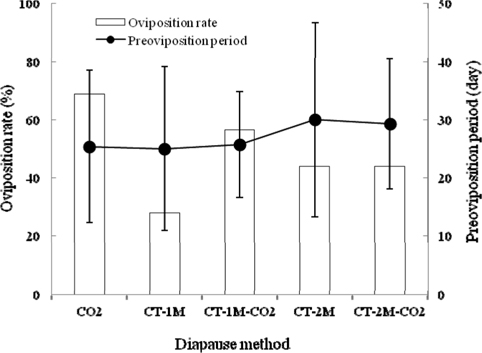
To evaluate the effects of different methods for breaking diapause on the egg-laying characteristics of B. ignitus queens, we investigated their oviposition rates (Fig. 1). Among the queens given these diapause treatments, the oviposition rate of CO2–narcotized queens showed the best performance, at 68.8%, which was 1.2-2.5 times higher than that of queens given the other diapause methods, which were 56.5%, 44.0%, 43.8% and 27.8% for the CT-1 M-CO2-, CT-2 M-CO2-, CT-2 M-, and CT-1 M-treated queens, respectively. There was no significant difference among the values for the treatment groups (Chi-squared test: x2=9.450, df=4, p=0.051) (Fig. 1). The preoviposition periods of queens given different diapause treatments, except for CT-2M-CO2 and CT-2M queens, were 25.0-25.7 d. The average preoviposition period of queens that were treated using CT-1M was 25.0±14.1 d, followed by the CO2-, CT-1M-CO2-, CT-2M-CO2- and CT-2M-treated queens. There were no significant differences in the preoviposition period among the groups given different diapause treatments (Tukey’s pairwise comparison test: F=0.56, df=4, 81, p=0.693) (Fig. 1). Among the diapause-treatment groups, B. ignitus queens given the CO2 treatment exhibited the best egg-laying characteristics. Insect metabolism is influenced in various ways by carbon-dioxide exposure and other environmental factors. Röseler and Röseler (1984) demonstrated that narcotizing prediapausing queens using carbon dioxide (a 30-min narcosis repeated twice) inhibited the formation of fat reserves, increased the content of juvenile hormone in vitro, and induced oogenesis. Histological studies of the honeybee revealed evidence of CO2 effects on the neurosecretory cells of brain, the corpora allata; additionally, the titer of juvenile hormone was increased (Nicolas, 1989).
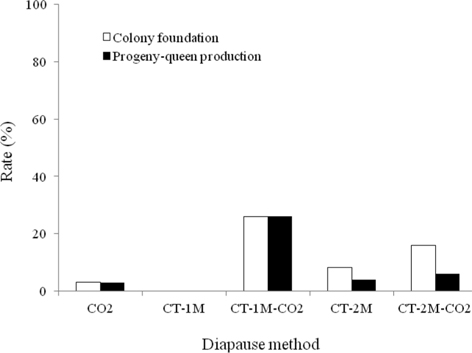
The colony development of B. ignitus queens given different diapause treatments was investigated (Fig. 2). Regarding the rate of colony foundation, which is one of the main criteria of colony quality in commercial rearing, the CT-1M-CO2-treated queens showed the best performance, 26.1%, which was 1.6-26.1 times higher than that of queens given other diapause treatments, followed by CT-2M-CO2, CT-2M, CO2 and CT-1M queens, at 16.0%, 8.6%, 3.1% and 0%, respectively. The CT-1M (cold treatment for 1 mo) queens did not form a colony. There were significant differences in the colony foundation rate of B. ignitus queens given different diapause treatments (Chi-squared test: x2=12.032, df=4, p=0.017). As shown in Fig. 2, the rate of progeny-queen production of the CT-1M-CO2 queens was 26.1%; this value was 4.4-26.1-fold higher than that of the CT-2M-CO2, CT-2M, CO2, or CT-1M queens. The progeny-queen production rate of B. ignitus queens was significantly affected by the diapause treatment at the level of p < 0.05, determined using the Chi-squared test (x2=15.597, df=4, p=0.004).
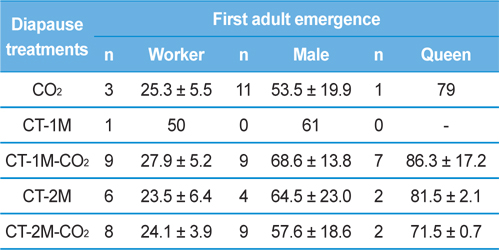
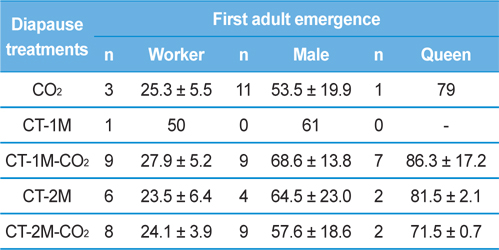
Table 1 shows period to adult emergence in the colonies of B. ignitus queens given different diapause treatments. The period to the emergence of the first worker for the CT-2M queens was the shortest among the queens given any of the diapause treatments (23.5±6.4 d), followed by the CT-2M-CO2, CT-2M-CO2, CO2, CT-1M-CO2 and CT-1M queens. No significant difference in the period to the emergence of the first worker was detected among the diapause treatment groups (Tukey’s pairwise comparison test, F = 1.08, df = 4, 37, p = 0.379). The period to the emergence of first the male, which was 53.5-68.6 d, was not significantly different between the diapause treatment groups (F = 0.15, df = 4, 41, p = 0.960). Similarly, the period to the emergence of the first queen, which was 71.5-86.3 d, was also not affected by the diapause treatment (F = 0.29, df = 4, 23, p = 0.884). The period until the first emergence of workers, males and queens of post-hibernating B. ignitus queens was 25.7-31.2 d, 67.7-79.0 d and 78.9-103.0 d, respectively and those of the CO2-treated B. ignitus queens was 23.7-25.0 d, 39.1-44.9 d and 65.3-82.7 d, respectively (Yoon et al., 2002, Yoon et al., 2003).
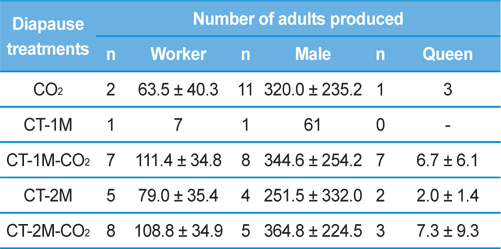
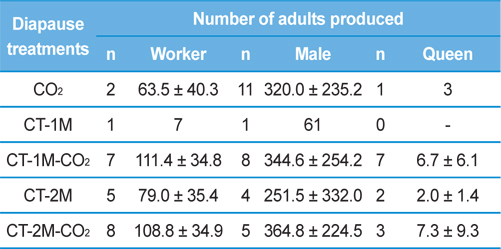
The relationship between the number of adults produced and the diapause treatment was investigated (Table 2). The numbers of workers reared in the colonies of the queens given CT-1M-CO2 or CT-2M-CO2 treatment was 111.4±34.8 and 108.8±34.9, which was 1.7-1.8 times that of the queens given CO2 or CT-2M treatments; nevertheless, there was no significant difference among the diapause treatment groups (Tukey’s pairwise comparison test, F = 2.82, df = 4, 18, p = 0.056). Worker production is important in evaluating the potential pollination efficiency of an insect pollinator. Asada and Ono (2002) found that the first oviposition day and the date of the emergence of the first worker had significant effects on the number of workers and queen progeny produced in laboratory-reared B. ignitus and B. hypocrite colonies because earlier oviposition by foundresses would result in earlier colony development. The number of males produced by queens given different diapause treatments, which was 251.5-364.8, was not significantly different among the diapause treatment groups (F = 0.35, df = 4, 21, p = 0.841). Regarding the number of queens produced, which is an important factor in the year-round rearing of bumblebees, the CT-1M-CO2 and CT-2M-CO2 treatment groups produced 6.7±6.1 and 7.3±9.3, respectively. These numbers were 2.2-7.3-fold higher than those for queens that were given the other treatments. However, the number of queens produced by B. ignitus queens was not significantly different among the diapause treatment groups (F = 0.38, df = 3, 9, p = 0.773). Diapause most likely underlies the variation among queens that may lead to differences in their ability to produce offspring (Beekmand and Vanstratum, 2000).
Considering the above-described colony developmental characteristics, the most favorable diapause treatment was CT-1M-CO2 (CO2–narcosis after cold treatment for 1 mo). A combination method of CO2- and cold-temperature treatment provided better results than did single CO2–narcosis or cold temperature treatment in terms of the colony development of diapause-broken B. ignitus queens. Although the B. ignitus queens given the CO2–narcosis treatment demonstrated the best egg-laying characteristics, the colony developmental ability of these queens was 1.4-8.4 times lower than that of CT-1M-CO2, CT-2M-CO2 and CT-2M queens. Carbon-dioxide-treated bumblebee queens sometimes produced males instead of workers and their nests could be of smaller size than those of overwintered queens Tasei, 1994; Yoon et al., 2003). Among the cold-treated B. ignitus queens, the CT-1M queens did not form a colony. Therefore, the necessary cold period for B. ignitus queens was determined to be at least 2 mo. Yoon et al. (2013) reported that a cold period of at least 2 mo applied 12 d after emergence was found to be the most favorable condition for diapause break in B. ignitus queens.
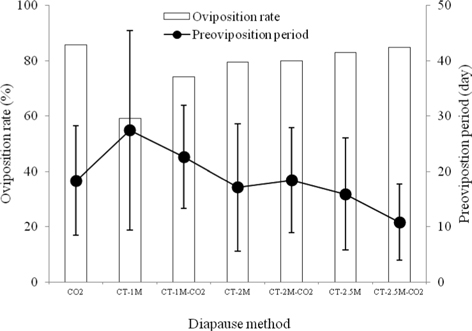
We investigated the oviposition rate of B. ignitus queens to determine the effects of different methods for diapause break on their egg-laying characteristics (Fig. 3). Among the those given the seven diapauses treatments, the oviposition rate of CO2–narcotized queens was highest, at 85.7%, which was 1.0-1.5 times higher than that of queens given the other diapause methods, which decreased in the order of CT-2.5M-CO2 (84.7%), CT-2.5M (83.0%), CT-2M-CO2 (80.0%), CT-2M (79.5%), CT-1M-CO2 (74.1%), and CT-1M (59.1%). There were significant differences among the treatment groups (Chi-squared test: x2=15.751, df=6, p=0.015) (Fig. 3). The preoviposition period of the CT-2.5M-CO2 queens was the shortest among the diapause treatment groups, at 10.8±6.9 d. This value was 1.5-2.5 times shorter than that of the other diapause treated queens, which followed of order of CT-2.5M (15.9±0.1 d), CT-2M (17.1±11.5 d), CO2 (18.3±9.9 d), CT-2M-CO2 (18.4±9.5 d), CT-1M-CO2 (22.6±9.3 d) and CT-1M (27.4±18.0 d). There were significant differences in the preoviposition period of the different diapause treatment groups (Tukey’s pairwise comparison test: F=9.95, df=6, 296, p=0.0001) (Fig. 3). In the case of cold treatment, the longer the cold duration is, the shorter the preoviposition period of the queen appears to be. Considering the egg-laying characteristics of the diapause treatment groups, the B. terrestris queens given the CO2 treatment had the best rate of oviposition and those given the CT-2.5 M-CO2 treatment exhibited the best (shortest) preoviposition period. In B. terrestris, Larrere et al. (1993) observed that the neurosecretory cells of diapausing queens resumed activity at 72 h after CO2–narcosis. Although egg-laying is only one small step in the process of successfully rearing bumblebees, it is a key factor in the large-scale production of commercial colonies and can be manipulated to reduce production costs. Yoon et al. (2004a) reported that the queen that had the earliest first oviposition day could make a stronger colony and could shorten the colony-formation period.
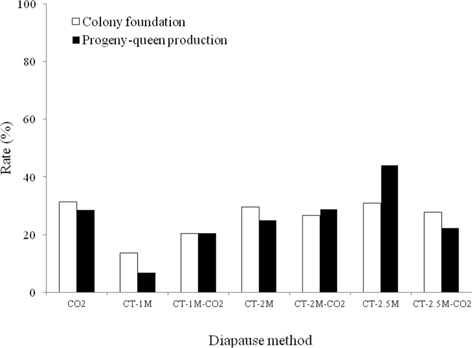
The colony development of B. terrestris queens given different diapause treatments was investigated (Fig. 4). Regarding the rate of colony foundation, the CO2-treated queens showed the best performance, 31.4%, which was 1.0-2.3 times higher than that of queens given the other diapause treatments, which followed the order of CT-2.5M, CT-2M, CT-2.5M-CO2, CT-1M-CO2 and CT-1M queens, at 31.0%, 29.5%, 27.8%, 26.7%, 20.4% and 13.6%, respectively. The CT-1M B. ignitus queens did not form a colony, whereas the CT-1M B. terrestris queens did form a colony. There were no significant differences in the colony foundation rate of B. terrestris queens given different diapause treatments (Chi-squared test: x2=6.541, df=6, p=0.365). As shown in Fig. 4, the rate of progeny-queen production of the CT-2.5M queens was 44.0%; this value was 1.5-6.4-fold higher than that of the CT-2M-CO2 (28.9%), CO2 (28.6%), CT-2M (25.0%), CT-2.5M-CO2 (22.2%), CT-1M-CO2 (20.4%) and CT-1M (6.9%) queens. The progeny-queen production rate of B. terrestris queens was affected by diapause treatment at the level of p < 0.05, as determined using the chi-squared test (x2=20.714, df=5, p=0.001). In the case of the B. terrestris queens, the colony development ability of the CT-2.5M-CO2 queens was lower than that of the CT-2.5M-CO2 queens. In view of the above-described colony developmental characteristics, we concluded that a combination method of CO2- and cold-temperature treatment provided better results for the colony development of diapause-broken B. terrestris queens than did only cold-temperature (of at least 2 mo) treatment. Diapausing bumblebee queens require sizeable fat reserves, which are consumed during the diapause period, and the amount of metabolic reserves remaining after diapause completion depends primarily on the length of diapause (Hoem, 1972).
In conclusion, the results of the present study demonstrated the most favorable method of inducing diapause break in B. ignitus and B. terrestris queens. Collectively, the findings of the present study indicated that the combined application of CO2 and cold temperature was a better method than only CO2–narcosis or cold-temperature treatment in terms of the colony development of diapause-broken B. ignitus and B. terrestris queens. The combination method of CO2 and cold treatment reduced the side effects of CO2–narcosis and shortened the duration of cold treatment by at least 1 mo.