There were various efforts to use sericin in polymeric fields (Kundu
The aim of present study is to find a way to delay the gelation time of sericin solution. It would be enough to maintain the sol state before fabrication is finished. We added NaCl, KCl and CaCl2 to the sericin solution because these salts have been known to alter the gelation time of silk fibroin (Kim
Silkworm cocoons were kindly provided by Heung Jin Co. Ltd. All chemicals were purchased from Sigma-Aldrich (USA).
>
Preparation of sericin solution
SS was extracted by boiling 25 g of Bombyx mori silkworm cocoons with 1 L of distilled water using an autoclave for 1 h at 120°C. The extracted solution was filtered with a nonwoven filter in order to remove the remaining cocoons. The solution was concentrated with a rotary evaporator, and the final concentration was 2.91 % (w/v)
>
Measurement of gelation time of sericin solution
In each 20 mL vial, 10 mL of sericin solution were added. Predetermined amounts of salts (NaCl, KCl and CaCl2) were added into the sericin solution, and stood in the laboratory at room temperature. The vial was tilt to 45 degree after predetermined intervals, and gelation time was determined when the surface line of solution did not change any longer.
>
Secondary structure analysis
The circular dichroism (CD, J-700, JASCO, Japan) spectra were obtained at the wavelength of 190-240 nm. The sericin solution was diluted with distilled water until a clear spectrum was obtained. In the case of sericin gel, the gel was spread gently on the surface of the quartz cell.
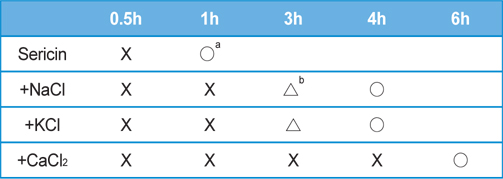
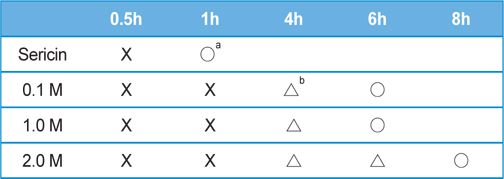
In general, the gelation time of sericin is affected by the initial concentration. When the concentration of sericin is below 1% (w/v), sericin tends to agglomerate rather than forming a gel. If the sericin concentration is higher than 1% (w/v), gel will be formed, and the gelation time will be shorten with the increase of concentration. In the present study, the concentration of sericin was adjusted to 2.91% (w/v) by concentrating the original solution. Table 1 shows the gelation time of sericin solution in the presence of 1 M of various salts. The gelation time of neat sericin solution was 1 h, but it took 4 h in the presence of NaCl and KCl. When CaCl2 was added, the gelation time was 6 h.
[Table 1.] Gelation time of sericin solution (2.91 wt%) in the presence various salts (1.0 M)
Gelation time of sericin solution (2.91 wt%) in the presence various salts (1.0 M)
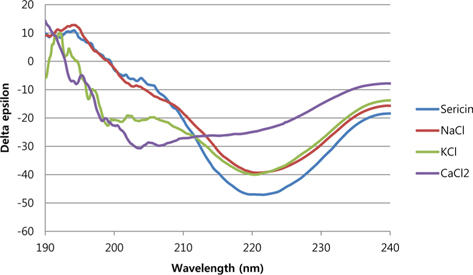
It has been known that the gelation of sericin occurs by the structural transition of sericin secondary structure from random coil to β-sheet structure (Padamwar and Pawar, 2004). Fig. 1 shows the CD spectra of sericin solution or gel after 2 h of incubation. In the CD spectra, random coil structure exhibits negative peak around 195 nm and very low ellipticity above 210 nm. On the other hand, positive peak around 195 nm and negative around 210-220 nm will be assigned as β-sheet structure (Greenfield, 2006). It could be clearly seen in Fig. 2 that the addition of salt affected the structural transition of sericin. While the neat sericin solution exhibited a typical β-sheet structure, the β-sheet structure was not fully developed in the salt added sericin solutions. When CaCl2 was added, the negative peak at 220 nm was not fully developed, indicating low β-sheet content compare to others. Indeed, after 2 h of incubation, the neat sericin solution was in a gel state, while NaCl and KCl added solutions were a gel-like state. The gel-like state means that the surface of solution was maintained at the beginning of tilting the vial but finally corrupted at the end of tilting. Conversely, the CaCl2 added solution was still in sol state after 2 h of incubation.
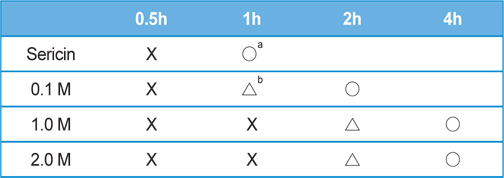
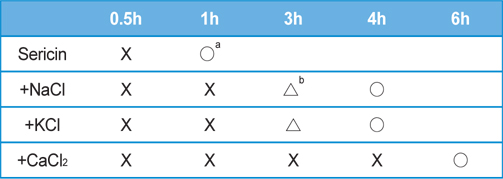
Table 2 and 3 shows the effect of salt concentration on the gelation time of sericin. The gelation time of sericin solution was delayed with the increase of salt concentration. The gelation times of KCl added sericin solution were as same as NaCl added one. In the case of NaCl or KCl added sericin solution, the gelation time was 4 h when 2 M of each salt were added. However, when 2 M of CaCl2 was added to sericin solution, the gelation could be prevented up to 8 h.
[Table 2.] Effect of NaCl concentration on the gelation time of sericin solution (2.91 wt%)
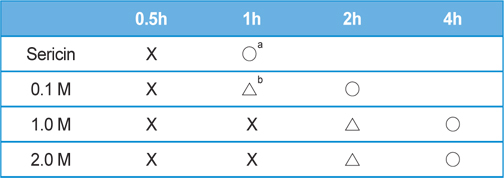
Effect of NaCl concentration on the gelation time of sericin solution (2.91 wt%)
[Table 3.] Effect of CaCl2 concentration on the gelation time of sericin solution (2.91 wt%)
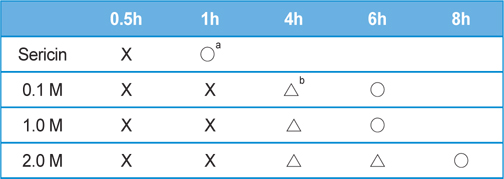
Effect of CaCl2 concentration on the gelation time of sericin solution (2.91 wt%)
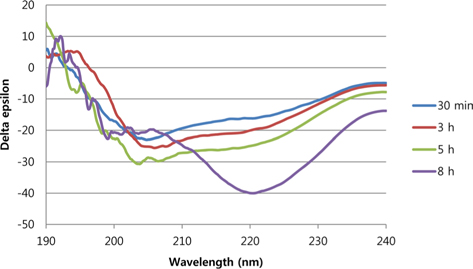
Fig. 2 shows the time course change of secondary structure of sericin solution when 1 M of CaCl2 was added. As the incubation time increased, the intensity of negative peak at 220 nm was increased indicating the development of β-sheet structure.
It could be summarized that the gelation time of sericin could be prevented by adding salt to the solution. The CaCl2 was most effective for the delay of gelation time of sericin solution. This is unexpected result when we consider the effect of CaCl2 in fibroin gelation. In the case of fibroin, gelation is promoted in the presence of CaCl2 due to divalent character of Ca2+ which could act as an ion bridge (Kim